Scalable Taxol C13 Side Chain Synthesis via Immobilized Biscinconyl Alkaloid Ligands
The pharmaceutical industry's relentless pursuit of effective oncology treatments has placed Paclitaxel, commonly known as Taxol, at the forefront of anticancer drug development, creating an unprecedented demand for its key synthetic intermediates. Patent CN1239441C introduces a groundbreaking methodology for catalyzing asymmetric hydroxylamination and dihydroxylation reactions using synthesized polymer-immobilized biscinconyl alkaloid ligands, specifically addressing the critical bottlenecks in producing the Taxol C13 side chain. This innovation represents a paradigm shift from traditional homogeneous catalysis, offering a robust solution that combines high enantioselectivity with the operational ease of heterogeneous systems. By anchoring chiral ligands onto a polyethylene glycol backbone, the technology ensures that the catalyst can be repeatedly recovered and reused without compromising the stereochemical integrity of the final product. For R&D directors and supply chain leaders, this patent data signifies a viable pathway to secure a reliable pharmaceutical intermediates supplier capable of meeting the rigorous purity and volume requirements of modern oncology drug manufacturing. The strategic implementation of this technology not only enhances the feasibility of complex chiral synthesis but also aligns with global sustainability goals by minimizing hazardous waste generation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the cis-dihydroxylation and asymmetric hydroxylamination of alkenes have relied heavily on stoichiometric amounts of oxidants like osmium tetroxide or potassium permanganate, which present severe safety and environmental challenges in an industrial setting. Although the Sharpless asymmetric dihydroxylation reaction introduced catalytic amounts of osmium tetroxide paired with cinchonine alkaloids, the homogeneous nature of these catalysts makes their recovery from the reaction mixture exceptionally difficult and economically prohibitive. The high toxicity and volatility of osmium tetroxide necessitate stringent safety protocols and specialized waste treatment facilities, driving up the operational costs significantly for any manufacturer attempting large-scale production. Furthermore, the residual presence of heavy metals in the final active pharmaceutical ingredient is a critical quality concern that requires additional purification steps, thereby extending the production timeline and reducing overall process efficiency. These inherent limitations have prevented many promising laboratory-scale asymmetric synthesis routes from being successfully translated into commercially viable manufacturing processes for high-value fine chemicals.
The Novel Approach
The novel approach detailed in the patent data overcomes these historical barriers by utilizing a homogeneous polymer-immobilized biscinconyl alkaloid ligand that retains the high selectivity of homogeneous catalysis while offering the recoverability of heterogeneous systems. This method employs a polyethylene glycol-supported ligand structure that facilitates the asymmetric transformation of olefins into chiral diols or amino alcohols with exceptional yield and enantiomeric excess. 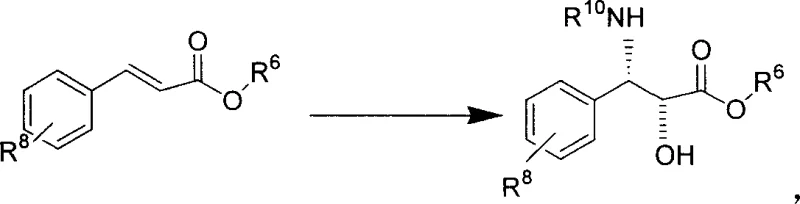 . By enabling the catalyst to be recycled repeatedly, the process drastically reduces the consumption of expensive chiral ligands and toxic osmium reagents, leading to substantial cost reduction in pharmaceutical intermediates manufacturing. The operational simplicity of this system allows for easier scale-up from laboratory benchtop to commercial production vessels, ensuring a consistent supply of high-purity OLED material or API precursors without the logistical nightmares associated with hazardous reagent handling. This technological advancement provides a competitive edge for procurement managers seeking to optimize their supply chain for complex polymer additives or specialty chemical production.
. By enabling the catalyst to be recycled repeatedly, the process drastically reduces the consumption of expensive chiral ligands and toxic osmium reagents, leading to substantial cost reduction in pharmaceutical intermediates manufacturing. The operational simplicity of this system allows for easier scale-up from laboratory benchtop to commercial production vessels, ensuring a consistent supply of high-purity OLED material or API precursors without the logistical nightmares associated with hazardous reagent handling. This technological advancement provides a competitive edge for procurement managers seeking to optimize their supply chain for complex polymer additives or specialty chemical production.
Mechanistic Insights into PEG-Immobilized Biscinconyl Alkaloid Catalysis
The core of this technological breakthrough lies in the unique structural design of the polymer-immobilized ligand, which chemically bonds chiral cinchonine alkaloids to a polyethylene glycol chain to create a soluble yet recoverable catalytic species.  . This molecular architecture ensures that the chiral environment surrounding the osmium center is maintained with high fidelity, directing the stereochemical outcome of the hydroxylamination or dihydroxylation reaction with precision. The polyethylene glycol moiety not only enhances the solubility of the catalyst in water-miscible organic solvents but also provides a steric framework that prevents the aggregation of active sites, thereby maintaining high catalytic turnover numbers over multiple cycles. For technical teams, understanding this mechanism is crucial as it explains how the system achieves high enantioselectivity comparable to free ligands while avoiding the contamination issues typical of homogeneous processes. The ability to fine-tune the molecular weight of the PEG support allows for further optimization of the catalyst's physical properties, ensuring compatibility with various downstream processing equipment and filtration methods used in commercial scale-up of complex pharmaceutical intermediates.
. This molecular architecture ensures that the chiral environment surrounding the osmium center is maintained with high fidelity, directing the stereochemical outcome of the hydroxylamination or dihydroxylation reaction with precision. The polyethylene glycol moiety not only enhances the solubility of the catalyst in water-miscible organic solvents but also provides a steric framework that prevents the aggregation of active sites, thereby maintaining high catalytic turnover numbers over multiple cycles. For technical teams, understanding this mechanism is crucial as it explains how the system achieves high enantioselectivity comparable to free ligands while avoiding the contamination issues typical of homogeneous processes. The ability to fine-tune the molecular weight of the PEG support allows for further optimization of the catalyst's physical properties, ensuring compatibility with various downstream processing equipment and filtration methods used in commercial scale-up of complex pharmaceutical intermediates.
Impurity control is another critical aspect where this immobilized ligand system excels, as the physical separation of the catalyst prevents the leaching of osmium and chiral ligands into the final product stream. In traditional homogeneous systems, trace amounts of heavy metals often persist despite rigorous purification, posing risks to patient safety and requiring costly additional scavenging steps that lower overall yield. The immobilized nature of the ligand ensures that the osmium complex remains associated with the polymer backbone throughout the reaction, allowing for simple separation techniques such as precipitation or extraction to remove the catalyst completely. This results in a cleaner crude product profile, reducing the burden on purification teams and ensuring that the final high-purity API intermediate meets stringent regulatory specifications for residual solvents and metals. Consequently, this mechanism supports a more robust quality assurance framework, giving supply chain heads confidence in the consistency and reliability of the manufacturing process for critical oncology drug components.
How to Synthesize Taxol C13 Side Chain Efficiently
The synthesis of the Taxol C13 side chain using this patented methodology involves a streamlined sequence of steps that leverages the unique properties of the immobilized ligand to achieve high efficiency and selectivity. The process begins with the preparation of the reaction mixture in a water-miscible organic solvent, where the immobilized ligand, osmium source, co-oxidant, and base are combined under controlled temperature conditions to initiate the catalytic cycle. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations. . This approach minimizes the exposure of personnel to hazardous reagents while maximizing the throughput of the reaction vessel, making it an ideal candidate for continuous manufacturing environments. By adhering to the specified molar ratios and reaction times outlined in the patent, manufacturers can consistently produce the chiral amino alcohol precursors required for Taxol semi-synthesis with minimal batch-to-batch variation. This level of process control is essential for maintaining the supply continuity required by global pharmaceutical partners who depend on timely delivery of critical raw materials.
- Prepare the homogeneous polymer-immobilized biscinconyl alkaloid ligand by connecting chiral cinchonine ligands to polyethylene glycol via chemical bonds.
- Conduct the reaction in a water-miscible organic solvent at -5°C to 10°C using K2OsO2(OH)4, co-oxidant, and base for 5 to 15 hours.
- Recover the catalyst through filtration or extraction, allowing for repeated use while isolating the high-purity chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this immobilized ligand technology offers profound advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for high-value chemical intermediates. The ability to recycle the catalyst multiple times translates directly into significant cost savings by reducing the need for frequent purchases of expensive chiral ligands and osmium reagents, which are subject to volatile market pricing and supply constraints. . Furthermore, the reduced generation of hazardous waste lowers the environmental compliance costs associated with disposal and treatment, contributing to a more sustainable and economically viable production model. For supply chain leaders, the robustness of this process ensures enhanced supply chain reliability, as the simplified workflow reduces the risk of production delays caused by complex purification or waste management issues. The scalability of the method means that production volumes can be increased to meet surging demand without the need for proportional increases in raw material consumption, providing a buffer against market fluctuations and ensuring a steady flow of materials to downstream drug manufacturers.
- Cost Reduction in Manufacturing: The elimination of stoichiometric oxidants and the reusability of the chiral catalyst fundamentally alter the cost structure of asymmetric synthesis, removing the need for expensive metal scavenging resins and reducing raw material expenditure. By avoiding the loss of valuable chiral ligands in the waste stream, the process ensures that the cost per kilogram of the final product remains competitive even at large production scales. This economic efficiency is further amplified by the reduced energy consumption associated with simpler workup procedures, allowing manufacturers to allocate resources to other critical areas of their operation. Ultimately, this leads to a more resilient cost base that can withstand external economic pressures while maintaining high profit margins on specialty chemical products.
- Enhanced Supply Chain Reliability: The simplified reaction workflow and the stability of the immobilized catalyst contribute to a more predictable production schedule, minimizing the risk of unplanned downtime due to catalyst deactivation or contamination issues. Since the catalyst can be recovered and reused, the dependency on external suppliers for fresh batches of chiral ligands is significantly reduced, mitigating the risk of supply disruptions caused by geopolitical or logistical challenges. This self-sufficiency in catalyst management ensures that production lines can operate continuously for extended periods, guaranteeing on-time delivery to customers who rely on just-in-time inventory systems. For procurement teams, this reliability translates into stronger supplier relationships and more favorable contract terms, as the risk of default is substantially lowered.
- Scalability and Environmental Compliance: The process is inherently designed for scale, with reaction conditions that are easily manageable in large-scale reactors without the need for exotic equipment or extreme operating parameters. The reduction in toxic osmium waste aligns with increasingly stringent environmental regulations, reducing the regulatory burden and potential liability associated with hazardous chemical handling. This environmental stewardship not only protects the company's reputation but also facilitates smoother approvals from regulatory bodies, accelerating the time to market for new drug formulations. Additionally, the lower waste volume reduces the logistical complexity of waste transport and disposal, further streamlining the overall supply chain operation and reducing the carbon footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydroxylamination technology in industrial settings. These answers are derived directly from the patent specifications to provide accurate and actionable insights for decision-makers evaluating this process for their supply chain. . Understanding these details is crucial for assessing the feasibility of integrating this method into existing manufacturing workflows and for calculating the potential return on investment. Technical teams should review these points closely to ensure that all operational parameters are aligned with the patent's recommendations for optimal performance.
Q: How does the immobilized ligand improve catalyst recovery compared to homogeneous systems?
A: The polymer-immobilized ligand allows for physical separation of the catalyst from the reaction mixture, enabling repeated reuse without significant loss of enantioselectivity or activity.
Q: What are the environmental benefits of this asymmetric hydroxylamination method?
A: By reducing the reliance on stoichiometric osmium tetroxide and enabling catalyst recycling, the process significantly minimizes toxic heavy metal waste and environmental contamination.
Q: Is this method suitable for large-scale production of Taxol intermediates?
A: Yes, the patent explicitly highlights the method's suitability for the mass production of Taxol C13 side chains due to its high yield, selectivity, and operational simplicity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Taxol C13 Side Chain Supplier
NINGBO INNO PHARMCHEM stands ready to leverage this advanced catalytic technology to support your production needs for high-purity Taxol C13 side chain intermediates with unmatched expertise and capacity. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for oncology drug synthesis. Our commitment to technical excellence means that we can adapt this patented immobilized ligand method to your specific process requirements, delivering consistent results that enhance your overall supply chain efficiency.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your current sourcing strategy and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements and operational constraints. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this technology for your specific application. Let us partner with you to secure a sustainable and cost-effective supply of critical pharmaceutical intermediates for the future.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
