Advanced Beta-Lactam Coupling Strategies for Commercial Taxol Intermediate Production
Advanced Beta-Lactam Coupling Strategies for Commercial Taxol Intermediate Production
The global demand for Paclitaxel, commonly known as Taxol, has driven intense research into efficient semi-synthetic routes that bypass the ecological limitations of natural extraction. Patent CN1086512A introduces a groundbreaking methodology for preparing Taxol intermediates utilizing specific beta-lactam derivatives as side-chain precursors. This technical insight report analyzes the chemical innovation disclosed in the patent, focusing on the reaction of beta-lactams with alcohols in the presence of activators to generate high-purity Taxol intermediates. The disclosed method addresses the critical bottleneck of attaching the C13 beta-amido ester side chain to the taxane nucleus, a step that has historically plagued manufacturers with low yields and complex purification requirements. By leveraging this technology, pharmaceutical manufacturers can achieve a more robust and economically viable supply chain for this essential anticancer agent.
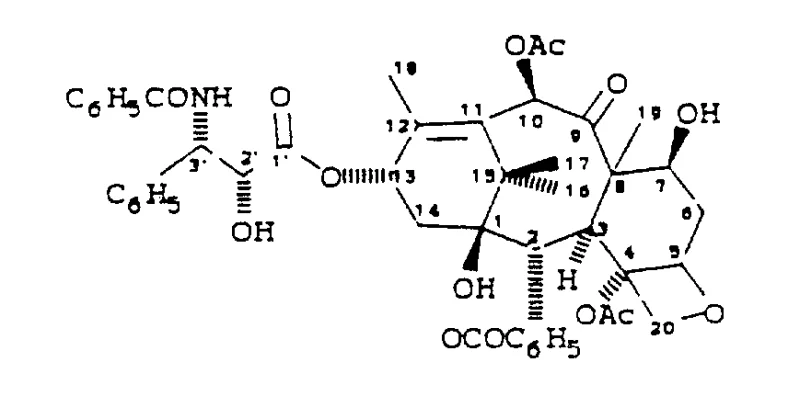
The structural complexity of Taxol, as illustrated in the provided chemical diagram, necessitates a synthesis strategy that preserves the integrity of the sensitive taxane ring system while efficiently installing the functional side chain. The patent highlights that traditional methods often struggle with the steric hindrance and reactivity issues associated with the C13 hydroxyl group. The innovation lies in the use of activated beta-lactams, which serve as highly reactive electrophiles capable of acylating the C13 position under mild conditions. This approach not only simplifies the synthetic route but also enhances the overall yield of the final product, making it a preferred choice for industrial applications where consistency and purity are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semi-synthesis of Taxol from 10-deacetylbaccatin III has faced significant challenges, particularly in the esterification step at the C13 position. Conventional methods often involve multi-step sequences to activate the side chain acid, which can lead to racemization and loss of optical purity, critically affecting the biological activity of the final drug. Furthermore, traditional coupling reagents may require harsh conditions that degrade the sensitive baccatin core, resulting in substantial material loss and increased production costs. The low atom economy and the generation of difficult-to-remove byproducts in these older processes create a burden on downstream purification, extending lead times and reducing the overall throughput of manufacturing facilities. These inefficiencies highlight the urgent need for a more streamlined and chemically elegant solution to this specific synthetic transformation.
The Novel Approach
The method disclosed in the patent offers a transformative solution by utilizing beta-lactams as activated side-chain donors that react directly with the taxane alcohol. This novel approach eliminates the need for pre-activating the carboxylic acid side chain, thereby reducing the number of synthetic steps and minimizing the exposure of the intermediate to potentially damaging conditions. The reaction proceeds efficiently in the presence of tertiary amine activators such as DMAP, facilitating the ring-opening of the beta-lactam and the subsequent formation of the desired ester bond with high regioselectivity. This strategy not only improves the chemical yield but also simplifies the work-up procedure, as the byproducts are generally easier to separate from the desired Taxol intermediate. The result is a more sustainable and cost-effective process that aligns with modern green chemistry principles.
Mechanistic Insights into Beta-Lactam Catalyzed Esterification
The core of this technology relies on the unique reactivity of the four-membered beta-lactam ring, which possesses significant ring strain that drives the acylation reaction. When exposed to a nucleophilic alcohol like 7-O-triethylsilyl baccatin III, the beta-lactam ring undergoes nucleophilic attack, facilitated by the activator which enhances the electrophilicity of the carbonyl carbon. This mechanism ensures that the side chain is attached with the correct stereochemistry, preserving the crucial chiral centers that define the drug's efficacy. The use of protecting groups, such as the ethoxyethyl group on the beta-lactam nitrogen, further controls the reactivity and prevents unwanted side reactions, ensuring that the coupling occurs exclusively at the desired position. Understanding this mechanistic pathway is essential for R&D teams aiming to optimize reaction parameters for maximum efficiency.
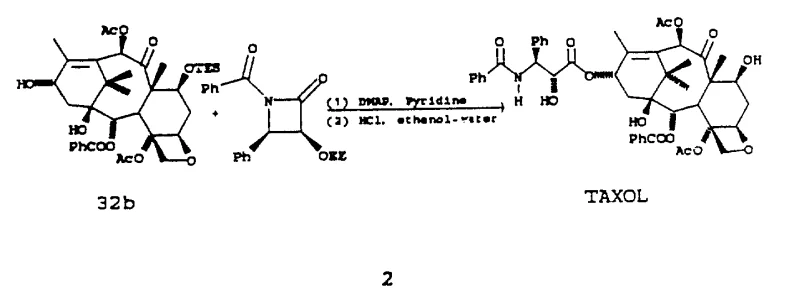
Impurity control is another critical aspect where this mechanism excels, as the mild conditions prevent the formation of common degradation products associated with harsher esterification methods. The patent details how the choice of activator and solvent system can be tuned to minimize the formation of diastereomers, which are difficult to separate and can compromise the quality of the final API. By maintaining a strict control over the reaction environment, manufacturers can ensure that the impurity profile of the Taxol intermediate remains within acceptable limits, reducing the need for extensive chromatographic purification. This level of control is vital for meeting the stringent regulatory requirements of global health authorities and ensuring patient safety.
How to Synthesize Taxol Intermediates Efficiently
The synthesis of Taxol intermediates using this beta-lactam methodology involves a sequence of well-defined steps that begin with the preparation of the activated side chain precursor. The process requires careful attention to stoichiometry and temperature control to ensure optimal conversion rates and product quality. Detailed standardized synthesis steps are provided in the guide below, which outlines the specific reagents and conditions required to replicate the high yields reported in the patent literature. Following these protocols allows production teams to achieve consistent results across different batches, ensuring reliability in the supply of this critical pharmaceutical intermediate.
- Prepare the activated beta-lactam side chain precursor, such as 1-benzoyl-3-(1-ethoxyethyl)-4-phenylazetidin-2-one, ensuring high stereochemical purity through resolution.
- React the beta-lactam with 7-O-triethylsilyl baccatin III in the presence of a tertiary amine activator like DMAP to form the protected Taxol intermediate.
- Perform mild hydrolysis to remove protecting groups without affecting the sensitive ester bonds, yielding the final Taxol intermediate ready for purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this beta-lactam coupling technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The streamlined nature of the process reduces the consumption of raw materials and solvents, leading to significant cost reduction in API manufacturing without compromising on quality. By minimizing the number of unit operations, facilities can increase their production capacity and respond more agilely to market demand fluctuations, thereby enhancing supply chain reliability. Furthermore, the use of readily available starting materials mitigates the risk of supply disruptions, ensuring a continuous flow of intermediates necessary for uninterrupted drug production. These factors collectively contribute to a more resilient and cost-effective supply chain for oncology medications.
- Cost Reduction in Manufacturing: The elimination of multiple activation steps and the use of milder reaction conditions significantly lower the operational costs associated with energy consumption and waste disposal. By avoiding expensive coupling reagents and reducing the need for complex purification processes, manufacturers can achieve substantial cost savings that improve the overall margin of the final product. This economic efficiency makes the process highly attractive for large-scale production where even small percentage improvements in yield translate to significant financial gains.
- Enhanced Supply Chain Reliability: The reliance on stable and commercially available beta-lactam precursors ensures that the supply chain is less vulnerable to the volatility often seen with specialized reagents. This stability allows procurement managers to secure long-term contracts with suppliers, guaranteeing the availability of key materials and reducing the risk of production delays. Additionally, the robustness of the reaction means that it can be performed in a wider range of manufacturing environments, further diversifying the supply base and enhancing overall security.
- Scalability and Environmental Compliance: The process is inherently scalable, allowing for seamless transition from pilot plant to commercial scale-up of complex pharmaceutical intermediates without the need for major equipment modifications. The reduced generation of hazardous waste and the use of less toxic solvents align with increasingly strict environmental regulations, minimizing the compliance burden on manufacturing sites. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing organization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this beta-lactam coupling technology in industrial settings. These answers are derived from the detailed experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and benefits of the method. Understanding these aspects is crucial for decision-makers evaluating the potential integration of this process into their existing manufacturing workflows.
Q: What are the advantages of using beta-lactam for Taxol side chain attachment?
A: The beta-lactam method offers significantly higher yields and milder reaction conditions compared to traditional coupling methods, reducing the risk of epimerization and degradation of the sensitive baccatin core.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the process utilizes readily available starting materials and avoids extreme conditions, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: How is stereochemical purity maintained during the coupling reaction?
A: Stereochemical integrity is preserved by using enantiomerically pure beta-lactam precursors and mild activators like DMAP, which prevent racemization at the chiral centers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Taxol Intermediate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging deep expertise in complex organic transformations to deliver high-quality pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Taxol intermediate meets the highest global standards. Our commitment to technical excellence allows us to navigate the complexities of beta-lactam chemistry with precision, delivering products that empower our clients to bring life-saving medications to market faster.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through advanced chemical solutions. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your strategic goals. Let us be your partner in achieving operational excellence and securing a reliable supply of critical anticancer intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
