Industrial Scale Synthesis of 1,3-Bis(2,4-Diaminophenoxy)Naphthalene for Advanced Electronic Materials
Industrial Scale Synthesis of 1,3-Bis(2,4-Diaminophenoxy)Naphthalene for Advanced Electronic Materials
The rapid evolution of the global electronics industry demands materials that can withstand extreme thermal and mechanical stresses while maintaining superior electrical insulation properties. At the forefront of this technological shift is the development of advanced polyimide resins, which rely heavily on the availability of high-purity aromatic diamine monomers. A pivotal advancement in this domain is documented in patent CN101245025A, which outlines a robust and efficient preparation method for 1,3-bis(2,4-diaminophenoxy)naphthalene. This specific compound serves as a critical building block for next-generation flexible copper clad laminates (FCCL) and high-performance composite materials. For R&D Directors and Procurement Managers seeking a reliable electronic chemical supplier, understanding the nuances of this synthesis route is essential for securing a competitive edge in the supply of specialty chemical intermediates. The methodology described offers a significant departure from traditional, cumbersome processes, presenting a pathway that aligns perfectly with modern manufacturing requirements for efficiency, purity, and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex aromatic diamines like 1,3-bis(2,4-diaminophenoxy)naphthalene has been plagued by significant operational challenges that hinder large-scale commercial viability. Traditional reduction methods often rely on catalytic hydrogenation using high-pressure hydrogen gas, which necessitates expensive, specialized reactor equipment and rigorous safety protocols to manage explosion risks. Furthermore, alternative chemical reduction pathways, such as those utilizing iron powder or sulfide reagents, frequently generate substantial amounts of hazardous waste sludge, creating severe environmental disposal burdens and increasing the overall cost reduction in electronic chemical manufacturing. These conventional routes often suffer from inconsistent reaction kinetics, leading to variable product quality and the formation of difficult-to-remove impurities that compromise the thermal stability of the final polyimide resin. The need for extensive post-reaction purification steps not only extends the production cycle but also drastically reduces the overall yield, making the cost of goods sold prohibitively high for mass-market applications in the aerospace and microelectronics sectors.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the reference patent introduces a streamlined catalytic system that fundamentally reshapes the production landscape for this high-value intermediate. By employing a palladium on carbon (Pd/C) catalyst in conjunction with hydrazine hydrate within a saturated fatty alcohol solvent system, the process achieves highly selective reduction under mild thermal conditions ranging from 70°C to 85°C. This methodological shift eliminates the dependency on high-pressure infrastructure, thereby drastically simplifying the reactor setup and enhancing operational safety for the supply chain head. The use of hydrazine hydrate as a hydrogen donor ensures a clean reaction profile where the primary byproduct is nitrogen gas, significantly minimizing the generation of toxic waste streams compared to metal-based reductions. Moreover, the specific solvent system facilitates an elegant work-up procedure where the product can be isolated through simple hot filtration and water precipitation, bypassing the need for energy-intensive distillation or chromatographic purification. This innovation represents a paradigm shift towards greener chemistry, offering substantial cost savings and improved process reliability for manufacturers of complex polymer additives.
Mechanistic Insights into Pd/C-Catalyzed Hydrazine Reduction
The core of this technological breakthrough lies in the precise interaction between the palladium catalyst and the hydrazine reducing agent, which facilitates the efficient conversion of nitro groups to amino groups without affecting the sensitive ether linkages in the naphthalene backbone. The reaction mechanism involves the adsorption of hydrazine onto the palladium surface, where it decomposes to generate active hydrogen species in situ. These active species then attack the nitro groups of the 1,3-bis(2,4-dinitrophenoxy)naphthalene substrate in a stepwise manner, progressing through nitroso and hydroxylamine intermediates before finally yielding the stable diamine product. The choice of a saturated fatty alcohol solvent is critical, as it not only dissolves the organic substrate effectively but also stabilizes the catalyst dispersion, preventing agglomeration and ensuring consistent reaction rates throughout the batch. This controlled environment minimizes side reactions such as over-reduction or dehalogenation, which are common pitfalls in less optimized systems, thereby ensuring the structural integrity required for high-performance polyimide applications.
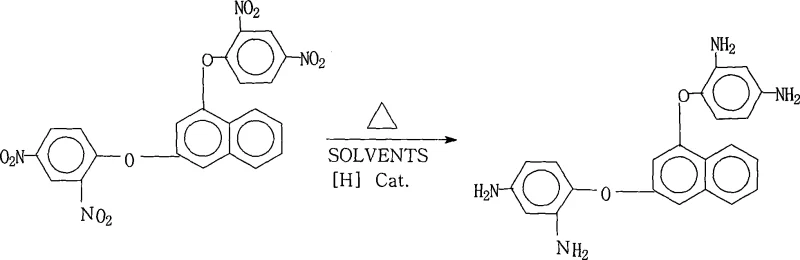
Furthermore, the impurity control mechanism inherent in this process is designed to meet the stringent purity specifications demanded by the semiconductor and display industries. The reaction conditions, specifically the temperature window of 70°C to 85°C and the reaction duration of 1 to 5 hours, are finely tuned to maximize conversion while preventing the degradation of the product. The subsequent purification step, which involves cooling the mother liquor and adding pure water, leverages the solubility differences between the desired diamine and any unreacted starting materials or intermediate byproducts. This crystallization process effectively traps impurities in the mother liquor, allowing the high-purity crystals to be isolated with a purity exceeding 99%. For the R&D Director, this level of control over the impurity profile is paramount, as even trace amounts of residual nitro compounds can act as defects in the final polymer matrix, compromising the dielectric properties and thermal resistance of the flexible copper clad laminate. The robustness of this mechanism ensures batch-to-batch consistency, a critical factor for maintaining quality standards in high-purity OLED material and electronic chemical manufacturing.
How to Synthesize 1,3-Bis(2,4-Diaminophenoxy)Naphthalene Efficiently
Implementing this synthesis route in a commercial setting requires a clear understanding of the operational parameters that drive efficiency and yield. The process is designed to be scalable, moving seamlessly from laboratory benchtop experiments to multi-ton production runs without significant re-engineering of the workflow. The key to success lies in the precise management of the catalyst loading, the rate of hydrazine addition, and the thermal profile of the reaction vessel. Operators must ensure that the Pd/C catalyst is evenly dispersed to maximize surface area contact, and the hydrazine hydrate must be added dropwise to control the exotherm and prevent local hot spots that could degrade the product. The patent data suggests that a weight ratio of substrate to catalyst between 100:1 and 100:20 provides an optimal balance between reaction speed and cost efficiency. By adhering to these standardized protocols, manufacturers can achieve high-purity 1,3-bis(2,4-diaminophenoxy)naphthalene with minimal operational friction. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction system by mixing 1,3-bis(2,4-dinitrophenoxy)naphthalene with Pd/C catalyst in a saturated fatty alcohol solvent.
- Add hydrazine hydrate solution dropwise while maintaining the temperature between 70°C and 85°C for 1 to 5 hours.
- Filter the mixture while hot, cool the mother liquor, add pure water to precipitate crystals, and vacuum dry to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis method translates directly into tangible strategic advantages that extend beyond mere technical performance. The elimination of high-pressure hydrogenation equipment represents a significant reduction in capital expenditure (CAPEX), allowing facilities to be established or retrofitted with lower financial barriers. Additionally, the simplified work-up procedure, which avoids complex distillation or extraction steps, drastically reduces energy consumption and labor hours per batch, leading to substantial cost savings in the overall manufacturing overhead. The use of readily available reagents like hydrazine hydrate and common fatty alcohol solvents ensures that the supply chain remains resilient against raw material shortages, reducing lead time for high-purity electronic chemical intermediates. This reliability is crucial for maintaining continuous production schedules in the fast-paced electronics sector, where delays can result in significant market share losses. By optimizing the process for yield and purity without the need for extensive downstream purification, the method enhances the overall throughput of the facility, enabling suppliers to meet increasing global demand more effectively.
- Cost Reduction in Manufacturing: The transition to atmospheric pressure conditions removes the necessity for expensive high-pressure reactors and the associated safety infrastructure, which significantly lowers the initial investment and ongoing maintenance costs for production facilities. Furthermore, the high selectivity of the Pd/C catalyst minimizes the formation of byproducts, reducing the waste disposal costs and the consumption of raw materials per unit of finished product. The ability to isolate the product through simple crystallization rather than energy-intensive distillation further contributes to a leaner cost structure, allowing for more competitive pricing in the global market for specialty chemical intermediates. This economic efficiency is compounded by the high yield reported in the patent examples, ensuring that maximum value is extracted from every kilogram of starting material processed.
- Enhanced Supply Chain Reliability: The reliance on common, commercially available solvents and reagents mitigates the risk of supply chain disruptions that often plague processes dependent on exotic or regulated chemicals. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up, ensuring that suppliers can respond quickly to fluctuations in market demand without compromising quality. The simplified process flow also reduces the potential for operational bottlenecks, enhancing the overall agility of the manufacturing unit. For the supply chain head, this means a more predictable and stable flow of goods, which is essential for long-term planning and contract fulfillment with major electronics manufacturers who require just-in-time delivery of critical raw materials.
- Scalability and Environmental Compliance: The process is inherently scalable, as the reaction kinetics and heat transfer characteristics remain consistent when moving from pilot to production scale, facilitating the commercial scale-up of complex polymer additives. From an environmental perspective, the generation of nitrogen gas as the primary byproduct significantly reduces the chemical oxygen demand (COD) of the waste stream compared to traditional metal reduction methods. This aligns with increasingly stringent global environmental regulations, reducing the compliance burden and the risk of regulatory penalties. The minimal waste generation also supports corporate sustainability goals, making the supply chain more attractive to environmentally conscious partners and investors who prioritize green chemistry initiatives in their sourcing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized diamine monomer. These insights are derived directly from the patented technology and are intended to clarify the operational benefits and performance characteristics for potential partners. Understanding these details is crucial for making informed decisions about integrating this material into your supply chain or product development pipeline. The answers reflect the consensus of technical data regarding the efficiency, safety, and applicability of the described synthesis route.
Q: What is the purity level achievable with this synthesis method?
A: The patented process consistently achieves product purity exceeding 99%, eliminating the need for complex subsequent refining or purification steps.
Q: Does this method require high-pressure hydrogenation equipment?
A: No, the process utilizes hydrazine hydrate as the reducing agent under atmospheric pressure conditions, significantly reducing equipment investment and safety risks.
Q: What are the primary applications for this diamine monomer?
A: It is a critical raw material for synthesizing highly branched polyimide resins used in flexible copper clad laminates (FCCL) and high-temperature advanced composites.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Bis(2,4-Diaminophenoxy)Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced electronic materials hinges on the availability of high-quality intermediates produced via robust and scalable pathways. The synthesis method for 1,3-bis(2,4-diaminophenoxy)naphthalene discussed herein exemplifies the type of innovative chemistry we specialize in bringing to market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from R&D to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for high-performance polyimide applications. We are committed to delivering not just a chemical product, but a comprehensive solution that enhances your competitive position in the global electronics market.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this superior synthesis technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower you to make data-driven decisions for your next product launch. Contact us today to discuss how we can support your growth with reliable, high-purity chemical solutions that drive innovation in the electronic materials sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
