Advanced Synthesis and Commercial Scalability of Dual-Function Fluorescent Schiff Base Intermediates
Advanced Synthesis and Commercial Scalability of Dual-Function Fluorescent Schiff Base Intermediates
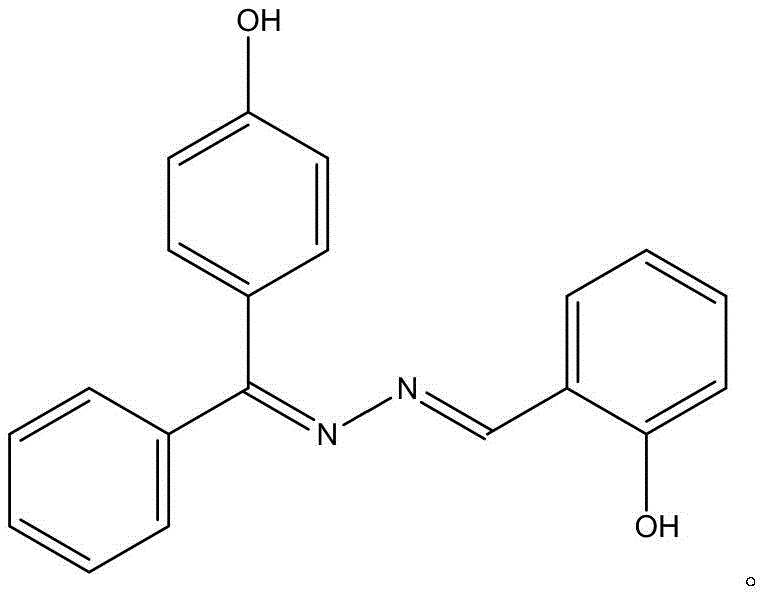
The landscape of functional organic materials is constantly evolving, driven by the demand for compounds that offer multifunctionality in both optical and pharmaceutical applications. Patent CN109912451B introduces a significant breakthrough in this domain by detailing the preparation and application of a novel Schiff base derivative, specifically 2-((E)-(((Z)-(4-hydroxyphenyl)(phenyl)methylene)hydrazono)methyl)phenol. This compound, characterized by its distinct orange bulk crystal appearance and a melting point range of 146.7‑148.1℃, represents a convergence of fluorescent properties and anticancer activity. For R&D directors and procurement specialists seeking reliable sources for high-value intermediates, this patent outlines a robust, one-step synthetic route that eliminates the complexity often associated with traditional heterocyclic synthesis. The molecular formula C20H16N2O2 defines a structure that is not only chemically stable but also exhibits strong absorption peaks near 207.0, 310.0, and 360.0nm, alongside potent fluorescence emission in the 500.0-620.0nm range. This technical insight report analyzes the commercial viability and mechanistic advantages of this synthesis, positioning it as a key candidate for cost reduction in fluorescent material manufacturing and pharmaceutical intermediate supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex Schiff bases and hydrazone derivatives has been plagued by inefficient multi-step protocols that rely heavily on harsh reaction conditions and expensive catalytic systems. Conventional routes often necessitate the use of strong acids or bases to drive the condensation equilibrium, which can lead to the degradation of sensitive functional groups such as phenolic hydroxyls, thereby compromising the overall yield and purity of the final product. Furthermore, many established methods require prolonged reaction times extending over several days, coupled with rigorous anhydrous conditions that increase energy consumption and operational costs significantly. The purification of these intermediates frequently involves column chromatography or multiple recrystallization steps to remove trace metal catalysts or side products, creating a bottleneck in the supply chain for high-purity organic fluorescent intermediates. These inefficiencies result in higher production costs and longer lead times, making it difficult for manufacturers to respond agilely to market demands for specialized optical materials and bioactive compounds.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology described in CN109912451B offers a streamlined, one-step condensation strategy that dramatically simplifies the production workflow. By utilizing 4-hydroxybenzophenone hydrazone and o-hydroxybenzaldehyde as readily available starting materials, the process achieves the target molecular architecture through a direct nucleophilic addition-elimination mechanism without the need for auxiliary catalysts. The flexibility of this approach is highlighted by its compatibility with a wide range of common organic solvents, including methanol, ethanol, acetonitrile, and dichloromethane, or even the option for solvent-free solid-phase grinding. This adaptability allows manufacturers to optimize the process based on available infrastructure and environmental regulations, facilitating the commercial scale-up of complex hydrazone derivatives. The reaction can be completed within a timeframe of 1 to 10 hours at temperatures ranging from ambient to reflux, ensuring rapid turnover and high throughput. The resulting product precipitates as high-quality orange blocky crystals upon natural volatilization of the filtrate, minimizing the need for extensive downstream purification and directly addressing the need for reducing lead time for high-purity anticancer intermediates.
Mechanistic Insights into One-Step Condensation Reaction
The core of this synthetic innovation lies in the efficient formation of the azomethine (-C=N-) linkage, which is the defining characteristic of Schiff bases. The reaction proceeds via a nucleophilic attack of the amino group from the 4-hydroxybenzophenone hydrazone onto the electrophilic carbonyl carbon of the o-hydroxybenzaldehyde. This initial addition forms a tetrahedral intermediate, which subsequently undergoes dehydration to establish the stable imine double bond. The presence of the phenolic hydroxyl groups on both aromatic rings plays a crucial role in stabilizing the final structure through intramolecular hydrogen bonding, which contributes to the compound's high melting point and crystalline nature. From a mechanistic standpoint, the absence of external catalysts suggests that the inherent electronic properties of the reactants are sufficient to drive the equilibrium towards product formation, especially under thermal conditions. This catalyst-free mechanism is particularly advantageous for pharmaceutical applications, as it inherently avoids the introduction of toxic heavy metal residues that would otherwise require costly and time-consuming removal steps to meet stringent regulatory purity specifications.
Regarding impurity control and structural integrity, the patent data indicates that the crystal belongs to a monoclinic system with space group P21/c, confirming a highly ordered lattice structure that facilitates effective purification through simple recrystallization. The specific stoichiometry allows for a molar ratio flexibility between 4:1 and 1:4, providing a buffer against minor weighing errors during large-scale batching without significantly impacting the conversion rate. The structural identification via elemental analysis and NMR spectroscopy confirms the exclusive formation of the desired E/Z isomer configuration, which is critical for maintaining consistent fluorescence properties. The strong fluorescence emission peak observed in the 500.0-620.0nm range is directly attributable to the extended conjugation system formed by the hydrazone bridge connecting the two aromatic systems. Understanding this electronic delocalization is vital for R&D teams aiming to tune the optical properties for specific sensor applications, as slight modifications to the substituents on the phenyl rings could shift the emission wavelength while retaining the core synthetic efficiency demonstrated in this patent.
How to Synthesize 2-((E)-(((Z)-(4-hydroxyphenyl)(phenyl)methylene)hydrazono)methyl)phenol Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it accessible for both laboratory research and industrial manufacturing environments. The process begins with the dissolution of the hydrazone precursor in a selected solvent, followed by the controlled addition of the aldehyde component under continuous stirring. Detailed standard operating procedures regarding specific temperature ramps, agitation speeds, and filtration techniques are critical for maximizing crystal quality and yield. For a comprehensive guide on the exact parameters validated during the patent filing, please refer to the standardized synthesis steps outlined below.
- Dissolve 4-hydroxybenzophenone hydrazone in a suitable organic solvent such as acetonitrile or ethanol within a reaction vessel equipped with stirring capabilities.
- Add o-hydroxybenzaldehyde (salicylaldehyde) to the solution maintaining a molar ratio between 1: 1 and 4:1, then heat under reflux or stir at room temperature for 1 to 10 hours.
- Filter the reaction mixture to remove insoluble impurities and allow the filtrate to stand for natural volatilization, yielding orange blocky crystals of the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route presents a compelling value proposition centered on operational efficiency and risk mitigation. The elimination of transition metal catalysts not only simplifies the chemical process but also removes a significant variable cost associated with precious metal procurement and recovery. Furthermore, the ability to utilize common, commodity-grade solvents like ethanol or acetonitrile ensures that raw material sourcing remains stable and unaffected by the volatility of specialized reagent markets. The robustness of the reaction conditions, which tolerate a wide range of temperatures and ratios, reduces the risk of batch failures, thereby enhancing supply chain reliability and ensuring consistent delivery schedules to downstream customers. This stability is crucial for maintaining the continuity of production lines for optical devices and pharmaceutical formulations that depend on this specific intermediate.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the drastic simplification of the unit operations involved. By consolidating the synthesis into a single step that does not require inert atmospheres or exotic catalysts, the capital expenditure for reactor setup and the operational expenditure for energy and labor are significantly lowered. The absence of a catalyst removal step means that expensive adsorbents or filtration media are not required, and the potential for solvent recycling is high due to the simplicity of the reaction mixture. Additionally, the option for solvent-free mechanical grinding offers a pathway for green manufacturing that could further reduce waste disposal costs and align with increasingly strict environmental regulations, leading to substantial long-term savings in overall production overheads.
- Enhanced Supply Chain Reliability: The reliance on 4-hydroxybenzophenone hydrazone and salicylaldehyde as starting materials leverages a mature global supply chain for bulk chemicals, minimizing the risk of shortages that often plague niche reagents. Since the reaction kinetics are fast, with completion achievable in as little as 1 hour under reflux, the manufacturing cycle time is compressed, allowing for quicker response to urgent orders and fluctuating market demand. The high yield implied by the formation of distinct orange blocky crystals suggests minimal material loss during conversion, optimizing the atom economy and ensuring that every kilogram of raw material translates effectively into sellable product. This efficiency creates a buffer against supply disruptions, enabling manufacturers to maintain safety stock levels more effectively and guarantee on-time delivery to clients in the competitive fine chemical sector.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton industrial production is straightforward due to the lack of exothermic hazards or pressure requirements typically associated with high-energy synthesis. The flexibility to operate in various solvents or under solvent-free conditions allows facilities to adapt the process to their existing equipment without major retrofits, accelerating the time-to-market for commercial quantities. From an environmental perspective, the reduced solvent usage in the grinding method and the avoidance of toxic metal waste streams simplify the wastewater treatment process, lowering the burden on effluent treatment plants. This alignment with green chemistry principles not only reduces compliance costs but also enhances the corporate sustainability profile, which is increasingly a deciding factor for multinational corporations when selecting a reliable Schiff base supplier.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this intermediate into their workflows, we have compiled answers to common inquiries based on the specific technical disclosures within the patent documentation. These responses address critical aspects regarding the compound's biological activity, synthetic requirements, and scalability potential. Understanding these details is essential for assessing the fit of this material within your specific R&D pipeline or manufacturing portfolio.
Q: What are the primary applications of this specific Schiff base derivative?
A: This compound serves a dual purpose as both a fluorescent material for optical devices and a potential anticancer agent, showing inhibition against A549 lung cancer, 4T1 breast cancer, and Hep G2 liver cancer cells with IC50 values ranging from 130.5 to 143.7 μM.
Q: Does the synthesis require expensive transition metal catalysts?
A: No, the patented method utilizes a direct condensation reaction between hydrazones and aldehydes without the need for transition metal catalysts, significantly simplifying the purification process and reducing heavy metal contamination risks.
Q: Is the process scalable for industrial production volumes?
A: Yes, the method supports both solution-phase and solvent-free solid-state grinding reactions, offering excellent flexibility for scaling up from laboratory grams to multi-ton commercial production while maintaining high purity standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-hydroxybenzophenone hydrazone-salicylaldehyde Schiff base Supplier
The technical potential of this dual-function Schiff base is immense, bridging the gap between advanced optical materials and therapeutic agents. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such sophisticated molecules to the global market. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of fluorescent intermediate meets the exacting standards required for DNA diagnosis, photochemical sensors, or anticancer drug development. We understand that consistency is key in fine chemical manufacturing, and our process engineering team is dedicated to optimizing every parameter to maximize yield and minimize impurities.
We invite you to collaborate with us to explore how this innovative synthesis can drive value in your projects. Whether you are looking for cost reduction in fluorescent material manufacturing or need a partner for the commercial scale-up of complex hydrazone derivatives, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis, where we can provide specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a stable, high-quality supply of this critical intermediate.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
