Revolutionizing 5,6-Indolino[2,3-b]indole Production via Green Photocatalysis
The pharmaceutical and fine chemical industries are constantly seeking more efficient and environmentally benign pathways for constructing complex heterocyclic frameworks. A significant breakthrough in this domain is documented in patent CN113583005A, which discloses a novel synthesis method for 5,6-indolino[2,3-b]indole. This double nitrogen heterocycle serves as a critical scaffold for drug molecules and photoelectric materials due to its enhanced rigidity and electron conjugation properties. Unlike traditional approaches that rely on hazardous reagents and multi-step sequences, this invention utilizes a photocatalytic oxidation strategy to achieve a direct [3+2] cyclization between indole and aniline. By leveraging visible light irradiation and advanced organic photocatalysts, the process bypasses the need for pre-functionalized nitro-compounds, offering a streamlined, one-pot solution that aligns perfectly with modern green chemistry principles and the demands of a reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indoloindole core has been plagued by inefficient and hazardous synthetic routes. Conventional Method I, for instance, relies on palladium-catalyzed coupling followed by a Cadogan reaction using triethyl phosphite, as illustrated in the reaction scheme below. This approach suffers from low overall yields, often below 35%, and generates significant phosphine-containing waste that requires complex treatment. Furthermore, Method III employs toxic carbon monoxide gas under high pressure and temperature, posing severe safety risks and operational challenges for commercial scale-up of complex pharmaceutical intermediates. These legacy methods not only increase the cost reduction in pharmaceutical intermediate manufacturing but also create substantial environmental burdens that modern regulatory frameworks increasingly penalize.
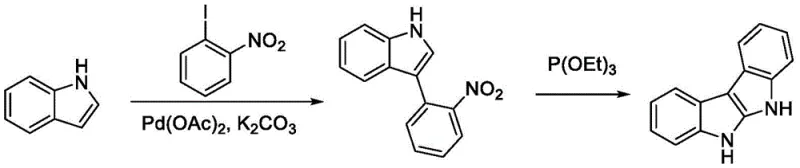
The Novel Approach
In stark contrast, the novel approach described in the patent utilizes a metal-free or low-metal photocatalytic system that operates under mild conditions. By employing commercially available indole and aniline as starting materials, the method eliminates the need for pre-synthesized nitro-precursors. The reaction proceeds via a [3+2] cyclization driven by photocatalytic oxidation, constructing both C-N and C-C bonds in a single operational sequence. This shift from thermal, high-pressure conditions to ambient temperature blue light irradiation drastically simplifies the equipment requirements and enhances process safety. The ability to use air as the terminal oxidant further underscores the sustainability of this route, making it an ideal candidate for high-purity pharmaceutical intermediate production without the baggage of heavy metal residues or toxic gas handling.
Mechanistic Insights into Photocatalytic [3+2] Cyclization
The core of this technological advancement lies in the precise manipulation of radical intermediates through photocatalysis. Under blue light irradiation at 425nm, the photocatalyst, such as the organic acridinium salt Mes-Acr+ BF4-, enters an excited state with high oxidation potential. This excited species oxidizes the electron-rich indole to generate a radical cation, which is the key reactive intermediate. This radical cation then undergoes nucleophilic addition with aniline to form a C-N bond, followed by radical addition and cyclization to construct the C-C bond. The subsequent oxidative dehydrogenation and aromatization steps, facilitated by oxygen from the air, finalize the formation of the rigid 5,6-indolino[2,3-b]indole structure. This mechanism avoids the harsh reduction conditions typical of nitro-group cyclizations, ensuring a cleaner reaction profile.

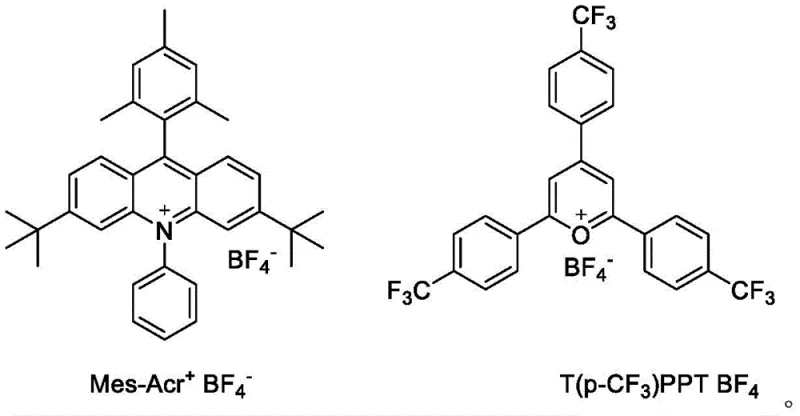
Impurity control is inherently superior in this photocatalytic system due to the specificity of the radical generation and the mildness of the reaction conditions. Traditional methods often produce side products from over-reduction or incomplete cyclization of nitro groups, leading to difficult-to-remove impurities that compromise the purity of the final API intermediate. In this new method, the use of specific organic onium salt photocatalysts, such as the structures shown below, ensures high selectivity. These catalysts possess tailored oxidation potentials that match the substrate, minimizing non-specific oxidation of the solvent or other functional groups. The absence of transition metals like palladium also removes the risk of metal contamination, a critical factor for meeting stringent purity specifications required by global regulatory bodies for drug substance manufacturing.
How to Synthesize 5,6-Indolino[2,3-b]indole Efficiently
Implementing this synthesis route requires careful attention to the choice of photocatalyst, base, and solvent to maximize yield and efficiency. The patent outlines a robust protocol where indole and aniline are mixed in a molar ratio of 1:2 to 1:3 in solvents like acetonitrile or ethyl acetate. The addition of a base, such as 2,6-lutidine or sodium acetate, is crucial for neutralizing protons generated during the reaction and maintaining the solubility of intermediates. Irradiation with a 45W blue LED lamp at 425nm under an air atmosphere drives the reaction to completion within 8 to 14 hours. The detailed standardized synthesis steps, including specific workup procedures like filtration, extraction, and recrystallization from toluene-methanol, are provided in the technical guide below to ensure reproducibility and high yield.
- Mix indole and aniline with a photocatalyst such as Mes-Acr+ BF4- in a solvent like acetonitrile.
- Add a base such as 2,6-lutidine and irradiate the mixture with 425nm blue light under air atmosphere.
- Filter insoluble matter, extract with ethyl acetate, and recrystallize from toluene-methanol to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this photocatalytic methodology offers transformative benefits that directly address cost and reliability concerns. By eliminating the need for expensive palladium catalysts and toxic phosphine reagents, the raw material costs are significantly reduced. The simplification of the process from a multi-step sequence to a one-pot reaction drastically reduces labor hours and energy consumption, leading to substantial cost savings in manufacturing. Furthermore, the use of abundant and cheap starting materials like indole and aniline ensures a stable supply chain, reducing the risk of raw material shortages that often plague specialty chemical production. This stability is crucial for maintaining continuous production schedules and meeting tight delivery windows for downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and phosphine reagents removes the need for expensive metal scavenging steps and complex waste treatment protocols. This simplification of the downstream processing significantly lowers the operational expenditure per kilogram of product. Additionally, the use of air as an oxidant replaces costly chemical oxidants, further driving down the variable costs associated with the synthesis. The overall process efficiency is enhanced by the high yields achieved under mild conditions, minimizing material loss and maximizing the output from each batch.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like indole and aniline mitigates the supply risks associated with specialized, custom-synthesized intermediates. These starting materials are widely available from multiple global suppliers, ensuring that production is not bottlenecked by a single source. The robust nature of the photocatalytic reaction, which tolerates ambient conditions, also reduces the dependency on specialized high-pressure or high-temperature equipment, making the manufacturing process more resilient to infrastructure disruptions. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent supply to partners.
- Scalability and Environmental Compliance: The green nature of this synthesis, characterized by the absence of toxic gases and heavy metals, simplifies regulatory compliance and environmental permitting. Scaling up photochemical reactions has become increasingly feasible with modern flow chemistry and LED technology, allowing for seamless transition from lab to commercial production. The reduction in hazardous waste generation aligns with corporate sustainability goals and reduces the costs associated with waste disposal. This environmental advantage not only protects the company from regulatory fines but also enhances its reputation as a responsible manufacturer in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this technology for their specific product pipelines. The information provided here aims to clarify the operational parameters and benefits to facilitate informed decision-making for procurement and technical leadership.
Q: What are the advantages of this photocatalytic method over traditional Cadogan reactions?
A: This method eliminates the need for nitro-precursors and phosphine compounds, significantly reducing toxic waste and simplifying purification compared to traditional Cadogan cyclizations.
Q: Which photocatalysts are suitable for this synthesis?
A: The patent specifies ruthenium complexes like Ru(bpy)3Cl2 or organic onium salts such as Mes-Acr+ BF4- and T(p-CF3)PPT BF4- as effective catalysts.
Q: Is this process scalable for industrial production?
A: Yes, the use of commercially available raw materials like indole and aniline, combined with mild reaction conditions and air as an oxidant, supports scalable and green manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6-Indolino[2,3-b]indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic technology for the production of high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative lab-scale methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by the global pharmaceutical industry. We are committed to delivering high-purity 5,6-indolino[2,3-b]indole that adheres to the highest quality standards, supporting your drug development and commercialization goals with reliability and precision.
We invite you to collaborate with us to optimize your supply chain and leverage the cost efficiencies of this green synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your production efficiency and reduce your time to market for critical pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
