Scalable Metal-Free Fluoroalkylation Technology for Advanced Pharmaceutical Manufacturing
The landscape of organic fluorine chemistry is undergoing a significant transformation driven by the urgent demand for efficient, sustainable methodologies to synthesize fluorine-containing building blocks. Patent CN110759836B introduces a groundbreaking approach to the fluoroalkylation of unsaturated bonds, specifically targeting olefins and alkynes. This technology leverages the unique properties of acetone, not merely as a solvent but critically as an electron donor compound, to facilitate Atom Transfer Radical Addition (ATRA) under visible light irradiation. For R&D directors and process chemists, this represents a paradigm shift away from the reliance on scarce transition metals and harsh radical initiators. The ability to generate diverse fluoroalkyl-substituted alkenes and alkanes with high stereoselectivity and atom economy addresses a critical bottleneck in the synthesis of bioactive molecules and advanced materials. By utilizing simple, commercially available iodo-fluoroalkyl compounds and inexpensive alkenes, this method streamlines the pathway to high-value intermediates essential for the pharmaceutical and agrochemical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the fluoroalkylation of unsaturated bonds has been dominated by strategies requiring rigorous conditions and expensive reagents. Traditional Atom Transfer Radical Addition (ATRA) often necessitates the use of stoichiometric amounts of radical initiators such as peroxides or sodium dithionite (Na2S2O4), which can lead to poor atom economy and difficult purification profiles due to byproduct formation. Furthermore, transition-metal catalyzed approaches, while effective, frequently rely on precious metals like Palladium, Ruthenium, or Iridium, as well as complex ligand systems that are sensitive to air and moisture. 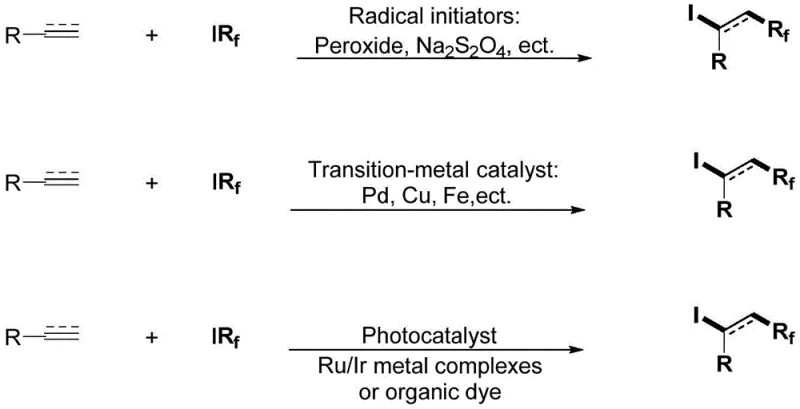 As illustrated in the comparative analysis of prior art, these conventional pathways often suffer from limited substrate scope, particularly when dealing with functionalized olefins that may coordinate with or poison the metal catalyst. The requirement for glovebox handling for certain low-valent metal species further complicates the operational complexity, driving up labor costs and limiting the feasibility of scale-up in standard manufacturing facilities. Additionally, the removal of trace heavy metals from the final API intermediate adds a costly and time-consuming downstream processing step that impacts overall yield and throughput.
As illustrated in the comparative analysis of prior art, these conventional pathways often suffer from limited substrate scope, particularly when dealing with functionalized olefins that may coordinate with or poison the metal catalyst. The requirement for glovebox handling for certain low-valent metal species further complicates the operational complexity, driving up labor costs and limiting the feasibility of scale-up in standard manufacturing facilities. Additionally, the removal of trace heavy metals from the final API intermediate adds a costly and time-consuming downstream processing step that impacts overall yield and throughput.
The Novel Approach
In stark contrast, the methodology disclosed in CN110759836B offers a streamlined, metal-free alternative that utilizes visible light and a simple base in an acetone medium. This novel catalytic system ingeniously exploits the electron-donating capability of acetone under blue light irradiation to generate the necessary fluoroalkyl radicals from iodo-fluoroalkyl precursors. 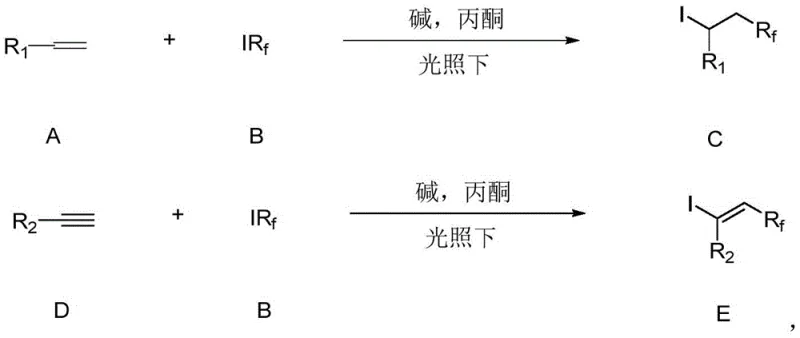 The reaction proceeds under mild conditions, typically at room temperature (25°C), eliminating the need for energy-intensive heating or cryogenic cooling. The use of potassium carbonate (K2CO3) as a benign base further enhances the green chemistry profile of the process. This approach not only simplifies the reaction setup—requiring only a standard LED light source and common laboratory glassware—but also dramatically expands the functional group tolerance. Substrates bearing sensitive moieties such as esters, amides, and halides, which might decompose under traditional radical conditions, are converted efficiently into the desired fluoroalkylated products with high yields, often exceeding 90% in optimized examples. This robustness makes the technology exceptionally attractive for the synthesis of complex pharmaceutical intermediates where structural integrity is paramount.
The reaction proceeds under mild conditions, typically at room temperature (25°C), eliminating the need for energy-intensive heating or cryogenic cooling. The use of potassium carbonate (K2CO3) as a benign base further enhances the green chemistry profile of the process. This approach not only simplifies the reaction setup—requiring only a standard LED light source and common laboratory glassware—but also dramatically expands the functional group tolerance. Substrates bearing sensitive moieties such as esters, amides, and halides, which might decompose under traditional radical conditions, are converted efficiently into the desired fluoroalkylated products with high yields, often exceeding 90% in optimized examples. This robustness makes the technology exceptionally attractive for the synthesis of complex pharmaceutical intermediates where structural integrity is paramount.
Mechanistic Insights into Acetone-Induced Visible Light Fluoroalkylation
The mechanistic elegance of this process lies in the dual functionality of acetone. Under the irradiation of blue LEDs (wavelengths between 430-490 nm), acetone acts as an electron donor, facilitating the single-electron transfer (SET) required to cleave the carbon-iodine bond in the fluoroalkyl iodide (e.g., ethyl iododifluoroacetate). This generates a reactive fluoroalkyl radical species without the need for an external photocatalyst like Ru(bpy)3 or Ir(ppy)3, which are notoriously expensive and difficult to remove from the final product. The radical subsequently adds across the unsaturated bond of the alkene or alkyne substrate to form a carbon-centered radical intermediate. This intermediate then abstracts an iodine atom from another molecule of the starting fluoroalkyl iodide, propagating the chain reaction and yielding the final 1,2-addition product. The presence of the base is crucial for neutralizing any acidic byproducts and potentially stabilizing intermediate species, ensuring the reaction proceeds to completion with minimal side reactions. This mechanism ensures excellent atom economy, as the iodine atom is retained in the product, providing a versatile handle for subsequent synthetic transformations such as cross-coupling or elimination reactions.
From an impurity control perspective, the absence of transition metals eliminates the risk of metal-catalyzed side reactions such as homocoupling or isomerization, which are common pitfalls in Pd or Cu-catalyzed systems. The mild reaction temperature prevents thermal degradation of sensitive functional groups, resulting in a cleaner crude reaction profile. This simplifies the downstream purification process, often allowing for straightforward crystallization or flash chromatography to achieve the stringent purity specifications required for GMP manufacturing. The high stereoselectivity observed, particularly in the formation of E-isomers for alkyne substrates, further reduces the burden of separating diastereomers, thereby enhancing the overall process efficiency and reducing material loss during purification.
How to Synthesize Fluoroalkylated Intermediates Efficiently
To implement this technology effectively, process chemists should focus on optimizing the light source intensity and the concentration of the base to maximize radical generation while minimizing potential side reactions. The protocol is remarkably robust, tolerating a wide range of substrates including terminal alkenes, internal alkenes, and various substituted alkynes. Detailed standardized synthesis steps for replicating this high-yield transformation are provided in the guide below.
- Prepare the reaction mixture by combining the alkene or alkyne substrate with the iodo-fluoroalkyl compound and a carbonate base (e.g., K2CO3) in acetone solvent under an inert atmosphere.
- Irradiate the reaction vessel with blue LEDs (430-490 nm) while stirring at room temperature (approx. 25°C) for approximately 16 hours to facilitate the radical addition.
- Upon completion, purify the resulting fluoroalkyl-substituted product using standard techniques such as column chromatography or recrystallization to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this acetone-induced fluoroalkylation technology offers substantial strategic advantages in terms of cost structure and supply reliability. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with fluctuating prices of metals like Palladium and Iridium. Furthermore, the use of acetone, a commodity chemical produced on a massive global scale, ensures a stable and inexpensive solvent supply chain, unlike specialized fluorinated solvents or high-purity organic solvents that may face logistical bottlenecks.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete removal of expensive transition-metal catalysts and specialized ligands. In traditional processes, these reagents can account for a significant portion of the raw material cost, and their removal requires additional processing steps like scavenging or extensive washing. By replacing these with cheap potassium carbonate and acetone, the direct material cost is drastically reduced. Additionally, the ambient temperature operation significantly lowers energy consumption compared to processes requiring reflux or cryogenic conditions, contributing to lower utility costs per kilogram of product manufactured.
- Enhanced Supply Chain Reliability: The reliance on widely available industrial raw materials enhances the resilience of the supply chain. Acetone, potassium carbonate, and simple alkenes are produced by multiple suppliers globally, reducing the risk of single-source dependency. The simplicity of the reaction setup, which does not require inert atmosphere gloveboxes or specialized high-pressure equipment for many substrates, allows for flexible manufacturing in standard multipurpose reactors. This flexibility ensures that production schedules can be maintained even if specific specialized equipment is unavailable, thereby reducing lead times for high-purity fluoroalkylated intermediates.
- Scalability and Environmental Compliance: The green chemistry profile of this method aligns perfectly with modern environmental regulations and corporate sustainability goals. The use of acetone, which is biodegradable and has a favorable safety profile compared to chlorinated solvents, simplifies waste treatment and disposal. The absence of heavy metal waste streams eliminates the need for complex hazardous waste management protocols, reducing disposal costs and regulatory burdens. The process is inherently scalable; the photochemical reaction can be adapted to flow chemistry setups or large-scale batch reactors with appropriate lighting arrays, facilitating the commercial scale-up of complex fluorine-containing molecules from gram to ton scale without losing efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel fluoroalkylation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What is the primary advantage of using acetone in this fluoroalkylation process?
A: Acetone serves a dual role as both the green solvent and the electron donor compound, eliminating the need for expensive transition-metal catalysts or complex photocatalysts typically required in traditional ATRA reactions.
Q: Does this method tolerate sensitive functional groups on the substrate?
A: Yes, the method exhibits excellent functional group compatibility, successfully processing substrates containing esters, amides, halogens, and hydroxyl groups without significant side reactions or degradation.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The use of cheap industrial raw materials, mild reaction conditions (room temperature), and the absence of air-sensitive catalysts make this protocol highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroalkylated Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the acetone-induced fluoroalkylation technology described in CN110759836B for the next generation of pharmaceutical and agrochemical products. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your innovative molecules can transition smoothly from the laboratory to the marketplace. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of fluoroalkylated intermediate meets the highest quality standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this cost-effective and sustainable synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this metal-free approach can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary custom synthesis projects.
