Advanced Synthesis of 2-Methyl-3-Methylmercaptofuran for Industrial Flavor Manufacturing
The landscape of synthetic flavor and fragrance manufacturing is constantly evolving, driven by the dual imperatives of cost efficiency and regulatory compliance. A pivotal advancement in this domain is detailed in patent CN115340513B, which discloses a robust preparation method for 2-methyl-3-methylmercaptofuran, a high-value compound known by its FEMA number 3949. This molecule is critically important in the food industry, imparting desirable meaty and nutty notes to savory formulations, yet its historical production has been fraught with logistical and safety challenges. The patented methodology fundamentally reimagines the synthetic pathway, shifting away from complex, multi-step derivations of furfural towards a streamlined construction using readily available commodity chemicals. By leveraging a strategic combination of nucleophilic substitution and cyclization, this innovation not only simplifies the molecular architecture but also addresses the severe safety concerns associated with traditional methylating agents. For global procurement and R&D teams, this represents a significant opportunity to optimize the supply chain for high-purity flavor intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
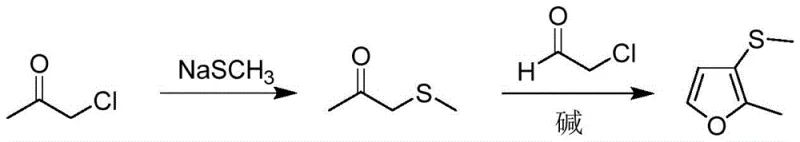
Historically, the industrial synthesis of 2-methyl-3-methylmercaptofuran has relied heavily on a methylation strategy that begins with 2-methyl-3-mercaptofuran. This precursor is itself a specialized perfume ingredient that is not commonly stocked as a bulk chemical raw material, typically requiring a lengthy synthesis starting from furfural. This elongated value chain introduces multiple points of failure, including yield losses at each isolation step and significant energy consumption during purification. Furthermore, the methylation step traditionally employs dimethyl sulfate, a reagent notorious for its extreme toxicity and carcinogenicity. The handling of dimethyl sulfate necessitates specialized containment equipment, rigorous personal protective protocols, and complex waste treatment procedures to neutralize hazardous byproducts. These factors collectively inflate the operational expenditure and create substantial regulatory hurdles for manufacturers aiming to scale production. Consequently, the reliance on such a convoluted and hazardous pathway has long been a bottleneck for reliable flavor and fragrance intermediate suppliers seeking to meet growing market demand efficiently.
The Novel Approach
In stark contrast, the methodology outlined in patent CN115340513B deconstructs the molecule to its fundamental building blocks, utilizing chloroacetone and sodium methyl mercaptide as the primary feedstocks. This approach bypasses the need for the expensive and scarce 2-methyl-3-mercaptofuran entirely, constructing the carbon-sulfur framework directly through a highly efficient nucleophilic substitution reaction. The subsequent cyclization with chloroacetaldehyde allows for the rapid assembly of the furan ring under mild alkaline conditions. By eliminating the use of dimethyl sulfate, the process inherently reduces the toxicological burden on the manufacturing facility, aligning perfectly with modern green chemistry principles. The use of chloroacetone and chloroacetaldehyde, which are produced on a massive industrial scale for various applications, ensures a stable and cost-effective supply of raw materials. This strategic pivot from functionalizing an existing furan ring to building it from acyclic precursors demonstrates a profound understanding of process chemistry, offering a pathway that is not only chemically elegant but also commercially superior for cost reduction in synthetic flavors manufacturing.
Mechanistic Insights into Nucleophilic Substitution and Cyclization
The core of this synthetic breakthrough lies in the precise orchestration of two distinct reaction mechanisms: nucleophilic substitution and base-catalyzed cyclization. In the first stage, the sulfur atom of the sodium methyl mercaptide acts as a potent nucleophile, attacking the electrophilic alpha-carbon of chloroacetone. This displacement of the chloride ion generates methylthio acetone, a key intermediate that retains the necessary functionality for ring closure. The patent data indicates that maintaining the reaction temperature between 45°C and 55°C is critical; temperatures that are too low result in sluggish kinetics, while excessive heat promotes side reactions that degrade the yield. The stoichiometry is carefully balanced, often employing a slight excess of the thiolate species to ensure complete conversion of the chloroacetone, thereby minimizing the presence of unreacted starting materials that could complicate downstream purification. This step exemplifies how controlling reaction parameters at the molecular level can drastically improve the purity profile of the intermediate before it even enters the cyclization phase.
Following the formation of methylthio acetone, the system transitions to the cyclization phase, which is governed by enolate chemistry. The intermediate is dispersed into an alkaline medium, such as potassium hydroxide or pyridine, which abstracts an acidic proton to generate a reactive carbanion or enolate species. The subsequent dropwise addition of chloroacetaldehyde at low temperatures (0-5°C) is a masterstroke of process control. Chloroacetaldehyde is highly reactive and prone to self-polymerization or rapid exothermic reactions if introduced too quickly or at elevated temperatures. By adding it slowly to the pre-cooled enolate solution, the reaction favors the desired intramolecular nucleophilic attack that closes the furan ring, rather than intermolecular polymerization. The alkaline environment also serves to scavenge the hydrogen chloride byproduct generated during the ring closure, driving the equilibrium towards the formation of the final 2-methyl-3-methylmercaptofuran product. This mechanistic understanding allows for the production of high-purity flavor intermediates with minimal impurity generation, a crucial factor for applications in the food and beverage sector.
How to Synthesize 2-Methyl-3-Methylmercaptofuran Efficiently
Implementing this synthesis route requires adherence to specific operational protocols to ensure safety and maximize yield. The process begins with the careful preparation of the nucleophilic substitution mixture, followed by a controlled transition to the cyclization environment. Operators must pay close attention to the order of addition, particularly during the second step, where the sequence of adding methylthio acetone to the base prior to the aldehyde is non-negotiable for success. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are outlined below to guide technical teams in replicating this efficient pathway.
- Perform nucleophilic substitution between chloroacetone and sodium methyl mercaptide at 45-55°C to obtain methylthio acetone.
- Disperse the crude methylthio acetone into an alkaline solution (e.g., KOH or pyridine) at 0-5°C.
- Add chloroacetaldehyde dropwise to effect cyclization, then warm to room temperature to isolate the final furan derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple chemical yield. The primary advantage lies in the radical simplification of the raw material portfolio. By shifting from specialized, low-volume precursors like 2-methyl-3-mercaptofuran to high-volume commodity chemicals like chloroacetone, companies can leverage existing bulk purchasing agreements and reduce exposure to supply volatility. This change fundamentally alters the cost structure of the final product, enabling significant cost savings without compromising on quality. Furthermore, the elimination of dimethyl sulfate removes a major regulatory and safety liability, reducing the costs associated with hazardous waste disposal and specialized worker training. These factors combine to create a more resilient and economically viable supply chain for high-purity flavor intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of expensive, multi-step precursors with cheap, readily available starting materials. Chloroacetone and chloroacetaldehyde are produced globally in vast quantities, ensuring competitive pricing and stable availability compared to niche fragrance ingredients. Additionally, the shorter synthetic route reduces the number of unit operations required, such as isolation and purification steps between stages, which directly lowers energy consumption and labor costs. The avoidance of toxic reagents like dimethyl sulfate also eliminates the need for expensive scrubbing systems and hazardous waste treatment protocols, further contributing to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: Reliance on a single-source supplier for a complex intermediate like 2-methyl-3-mercaptofuran creates a single point of failure in the supply chain. By adopting a synthesis based on commodity chemicals, manufacturers diversify their supply risk, as chloroacetone and sodium methyl mercaptide are available from multiple global vendors. This redundancy ensures continuity of supply even if one vendor faces production issues. Moreover, the simplified logistics of handling less hazardous materials streamline transportation and storage requirements, reducing lead times and facilitating faster response to market demands for this critical flavor component.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is vastly superior. The removal of carcinogenic methylating agents simplifies the permitting process for new production lines and reduces the regulatory burden on existing facilities. The reaction conditions are mild, operating near ambient pressures and moderate temperatures, which makes the process easier to scale from pilot plant to commercial production without requiring exotic reactor materials. The reduced generation of hazardous byproducts aligns with increasingly stringent global environmental regulations, positioning manufacturers as responsible stewards of sustainability while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis pathway. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of this supply source.
Q: Why is the chloroacetone route superior to traditional methylation methods?
A: The traditional method requires 2-methyl-3-mercaptofuran as a starting material, which is expensive and difficult to source, along with toxic dimethyl sulfate. The new route uses commodity chemicals like chloroacetone and chloroacetaldehyde, significantly simplifying the supply chain and improving safety profiles.
Q: What are the critical control points for maximizing yield in this synthesis?
A: Temperature control is vital. The initial substitution should be maintained between 45-55°C to prevent side reactions, while the cyclization step requires strict low-temperature addition (0-5°C) of chloroacetaldehyde to manage exothermicity and minimize polymerization.
Q: Is this process scalable for industrial production of flavor intermediates?
A: Yes, the process utilizes standard unit operations such as dropwise addition, extraction, and rectification. The avoidance of highly toxic reagents like dimethyl sulfate makes it more amenable to large-scale regulatory compliance and environmental safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-3-Methylmercaptofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthetic route requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN115340513B are fully realized in an industrial setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-methyl-3-methylmercaptofuran meets the exacting standards required for food and fragrance applications. Our commitment to quality assurance means that clients can rely on us for consistent product performance, batch after batch.
We invite global partners to engage with our technical procurement team to discuss how this optimized synthesis can drive value for your specific applications. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of this advanced intermediate into your supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
