Advanced Synthesis of Dibenzothiophene-Coumarin Chalcones for High-Performance Luminescent Applications
The field of organic optoelectronics is continuously evolving, driven by the demand for materials with superior thermal stability and enhanced photophysical properties. Patent CN102516235B introduces a significant advancement in this domain by disclosing a novel class of dibenzothiophene chalcones containing a coumarin skeleton. These compounds are engineered to function as high-performance organic luminescent materials, specifically designed to overcome the limitations of existing fluorophores. The core innovation lies in the construction of an A-π-A (Acceptor-π-Acceptor) system, which integrates the robust electron-accepting capabilities of both the dibenzothiophene and coumarin moieties. This structural motif results in materials that exhibit remarkable thermal decomposition temperatures exceeding 300°C, alongside strong ultraviolet absorption and fluorescence emission. For R&D directors and procurement specialists seeking reliable organic luminescent material intermediates, this patent outlines a synthesis pathway that balances molecular complexity with practical manufacturability.
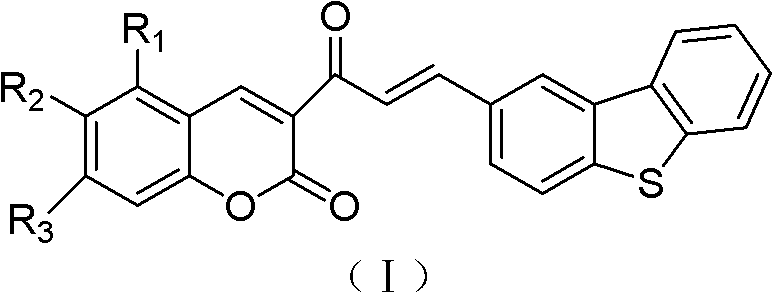
The strategic design of these molecules allows for tunable photophysical properties through the modification of substituents on the coumarin ring, such as methoxy, hydroxy, or diethylamino groups. This versatility makes them highly attractive candidates for applications ranging from two-photon microscopy imaging to organic light-emitting diodes (OLEDs). Furthermore, the patent emphasizes that the molecular size of these compounds reaches the nanometer level, enabling their utilization as nanomaterials in advanced device architectures. By leveraging the inherent stability of the dibenzothiophene core and the well-established fluorescence of the coumarin scaffold, this technology provides a robust platform for developing next-generation display and sensing materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing conjugated chalcone derivatives often suffer from significant drawbacks regarding thermal stability and purification complexity. Many conventional D-π-A (Donor-π-Acceptor) systems, while effective for certain applications, frequently exhibit lower thermal decomposition thresholds, which can limit their processing windows in vacuum deposition or high-temperature curing processes common in semiconductor manufacturing. Additionally, older synthetic routes may rely on harsh reaction conditions or expensive transition metal catalysts that introduce difficult-to-remove impurities, thereby compromising the purity profile required for electronic grade materials. The presence of residual metals can act as quenching sites for fluorescence, drastically reducing the efficiency of the final luminescent device. Moreover, multi-step syntheses involving unstable intermediates often lead to lower overall yields and increased waste generation, posing challenges for both cost reduction in organic luminescent material manufacturing and environmental compliance.
The Novel Approach
The methodology presented in CN102516235B addresses these challenges through a streamlined three-step synthesis that prioritizes both yield and purity. By constructing an A-π-A system, the inventors have created a molecule with inherently higher thermal stability, verified by thermogravimetric analysis showing decomposition temperatures above 300°C. The synthetic route avoids the use of precious metal catalysts in the final coupling step, relying instead on organocatalysis with piperidine, which simplifies the downstream purification process significantly. The final products can be isolated via simple filtration and recrystallization, eliminating the need for complex column chromatography on a large scale. This approach not only enhances the chemical purity of the high-purity organic luminescent material intermediates but also drastically reduces the operational expenditure associated with solvent consumption and waste treatment. The simplicity of the separation method ensures that the supply chain remains robust and capable of meeting the stringent quality standards of the optoelectronics industry.
Mechanistic Insights into TiCl4-Catalyzed Formylation and Condensation
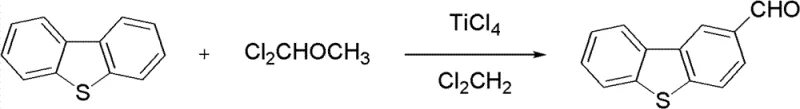
The synthesis begins with the functionalization of the dibenzothiophene core, a critical step that sets the stage for the subsequent conjugation. The first stage involves a Vilsmeier-Haack type formylation where dibenzothiophene reacts with 1,1-dichloromethyl methyl ether in the presence of titanium tetrachloride (TiCl4). In this electrophilic aromatic substitution, TiCl4 acts as a potent Lewis acid, activating the chloromethyl ether to generate a highly reactive iminium-like electrophile. This species attacks the electron-rich dibenzothiophene ring, preferentially at the 3-position due to steric and electronic factors, to install the crucial aldehyde functionality. The reaction is conducted in anhydrous dichloromethane at controlled temperatures between 20-30°C to prevent over-reaction or polymerization. Following the reaction, the mixture is quenched in ice water, and the product is extracted and purified via column chromatography to yield 3-dibenzothiophene aldehyde. This intermediate serves as the electrophilic partner for the final assembly of the chalcone skeleton.
The second phase of the mechanism involves the construction of the coumarin acceptor unit. Salicylaldehyde derivatives undergo a Knoevenagel-type condensation with ethyl acetoacetate, catalyzed by a base such as piperidine. This reaction proceeds through the formation of an enolate from the beta-keto ester, which attacks the aldehyde carbonyl carbon. Subsequent intramolecular transesterification and dehydration lead to the formation of the 3-acetylcoumarin derivative. This step is highly efficient, often yielding the desired product simply upon filtration of the precipitate from the reaction mixture. The final assembly utilizes a Claisen-Schmidt condensation between the 3-dibenzothiophene aldehyde and the 3-acetylcoumarin derivative. Under basic conditions and reflux, the methyl ketone of the coumarin forms an enolate that attacks the aldehyde of the dibenzothiophene. The resulting beta-hydroxy ketone spontaneously dehydrates to form the conjugated alpha,beta-unsaturated ketone linkage, completing the A-π-A system. This final conjugation extends the pi-system across the entire molecule, which is responsible for the red-shifted absorption and enhanced two-photon absorption properties observed in the final material.
How to Synthesize Dibenzothiophene Chalcone Efficiently
The preparation of these advanced luminescent materials follows a logical progression of functional group transformations that are well-suited for batch processing. The process starts with the activation of the dibenzothiophene core, followed by the independent synthesis of the functionalized coumarin fragment, and concludes with their convergence. Each step has been optimized to maximize yield while minimizing the formation of side products that could complicate purification. The use of common solvents like dichloromethane and ethanol, along with inexpensive catalysts like TiCl4 and piperidine, ensures that the process remains economically viable. Detailed standardized synthetic steps for replicating this high-efficiency route are provided in the guide below, ensuring reproducibility for process chemists aiming to scale this technology.
- Synthesize 3-dibenzothiophene aldehyde via TiCl4-catalyzed formylation of dibenzothiophene using 1,1-dichloromethyl methyl ether.
- Prepare 3-acetylcoumarin derivatives by condensing salicylaldehyde derivatives with ethyl acetoacetate using a basic catalyst like piperidine.
- Perform the final Claisen-Schmidt condensation between the aldehyde and acetylcoumarin under reflux conditions to yield the target chalcone.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the synthesis route described in this patent offers distinct advantages that translate directly into operational efficiency and cost optimization. The reliance on commodity chemicals such as dibenzothiophene, salicylaldehyde, and ethyl acetoacetate means that raw material sourcing is not constrained by geopolitical risks or limited supplier bases. These starting materials are produced on a multi-ton scale globally, ensuring a continuous and reliable supply chain for the manufacturing of these specialized intermediates. Furthermore, the reaction conditions are relatively mild, avoiding the need for cryogenic temperatures or ultra-high pressure equipment, which reduces the capital expenditure required for plant retrofitting. The ability to isolate intermediates and final products through simple crystallization rather than energy-intensive distillation or preparative HPLC significantly lowers the utility costs associated with production.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in the final coupling step is a major driver for cost savings. Traditional cross-coupling reactions often require palladium or nickel catalysts, which are not only expensive but also require rigorous removal steps to meet ppm-level specifications for electronic materials. By utilizing an organocatalytic aldol condensation, this process removes the need for expensive metal scavengers and the associated loss of product during purification. Additionally, the high thermal stability of the final product reduces the risk of degradation during processing, leading to higher throughput and less material waste. The simplified workup procedures, involving filtration and washing, further reduce labor hours and solvent consumption, contributing to substantial cost savings in organic luminescent material manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent batch-to-batch quality, which is critical for long-term supply agreements with display manufacturers. Since the reaction does not depend on sensitive reagents that degrade rapidly upon exposure to air or moisture (beyond standard anhydrous requirements for the first step), inventory management is simplified. The intermediates, particularly the 3-acetylcoumarin derivatives, are stable solids that can be stored for extended periods, allowing for flexible production scheduling. This stability mitigates the risk of supply disruptions caused by the spoilage of sensitive reagents. Consequently, partners can maintain leaner inventory levels while still guaranteeing the reducing lead time for high-purity organic luminescent material intermediates required by their downstream clients.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot runs without significant re-optimization. The solvents used, primarily ethanol and dichloromethane, are widely recycled in industrial settings, and the absence of heavy metal waste simplifies effluent treatment protocols. This aligns with increasingly stringent environmental regulations governing the production of fine chemicals. The high atom economy of the condensation steps means that less waste is generated per kilogram of product, supporting sustainability goals. The commercial scale-up of complex organic luminescent material intermediates is thus facilitated by a green chemistry approach that minimizes environmental impact while maximizing production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these dibenzothiophene-coumarin hybrids. The answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on performance metrics and process feasibility. Understanding these details is essential for stakeholders evaluating the integration of these materials into existing product lines or R&D pipelines.
Q: What are the thermal stability characteristics of these dibenzothiophene chalcones?
A: According to patent CN102516235B, the synthesized compounds exhibit exceptional thermal stability with decomposition temperatures exceeding 300°C, making them suitable for high-temperature processing in electronic device fabrication.
Q: Why is the A-π-A system significant in this molecular design?
A: The A-π-A (Acceptor-π-Acceptor) architecture formed by coupling the electron-deficient coumarin and dibenzothiophene units enhances two-photon absorption cross-sections and improves fluorescence emission properties compared to traditional D-π-A systems.
Q: Is the synthesis scalable for industrial production?
A: Yes, the process utilizes readily available raw materials like dibenzothiophene and salicylaldehyde derivatives, and relies on standard unit operations such as reflux and recrystallization, facilitating straightforward commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dibenzothiophene Chalcone Supplier
The technological potential of the dibenzothiophene-coumarin chalcone system is immense, offering a pathway to higher performance in OLEDs and bio-imaging applications. NINGBO INNO PHARMCHEM stands ready to support your development needs as a premier CDMO partner with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific anhydrous and reflux conditions required for this synthesis, ensuring that the stringent purity specifications demanded by the optoelectronics industry are consistently met. With our rigorous QC labs and state-of-the-art analytical instrumentation, we guarantee that every batch delivered meets the highest standards of quality and consistency, providing you with a secure foundation for your product development.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and purity requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that identifies opportunities to optimize your supply chain further. We encourage you to request specific COA data and route feasibility assessments to validate the compatibility of these intermediates with your downstream processes. Let us help you accelerate your time-to-market with reliable, high-quality materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
