Scalable Production of 7-Methylindole: A Technical Breakthrough for API Manufacturing
Scalable Production of 7-Methylindole: A Technical Breakthrough for API Manufacturing
The pharmaceutical industry constantly seeks robust, scalable pathways for critical heterocyclic building blocks, and the recent disclosure in patent CN112457235A represents a significant leap forward in the synthesis of 7-methylindole. This specific indole derivative serves as a pivotal scaffold in the development of various bioactive molecules, including potential treatments for hypertension, antiviral agents, and antitumor compounds. Historically, the manufacturing of substituted indoles has been plagued by low yields, harsh reaction conditions, and significant scalability bottlenecks that hinder commercial viability. The technical solution presented in this patent addresses these chronic pain points by introducing a streamlined two-step sequence that operates under atmospheric pressure and utilizes readily available reagents. For R&D directors and procurement specialists alike, this methodology offers a compelling alternative to legacy processes, promising not only improved chemical efficiency but also a more favorable economic profile for large-scale production. By leveraging a novel condensation strategy followed by a straightforward catalytic reduction, this approach effectively bypasses the complex purification hurdles associated with traditional indole synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 7-methylindole was largely dependent on methodologies that were fundamentally flawed for industrial application. One prominent historical route, documented in J. Med. Chem. (1980), relied on the reaction of aniline derivatives with chloroacetonitrile in the presence of titanium tetrachloride. As illustrated in the reaction scheme below, this pathway suffered from catastrophic yield losses, with the first step yielding merely 26 percent and the overall process capping at a dismal 22.3 percent. Furthermore, the use of titanium tetrachloride introduces severe handling hazards and corrosion issues, while the heating of chloroacetonitrile often leads to uncontrolled polymerization, making the reaction mixture difficult to stir and impossible to scale beyond laboratory glassware.
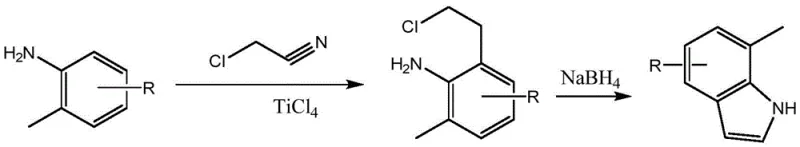
Another widely cited approach, the Bartoli indole synthesis, presents its own set of formidable logistical challenges. While chemically elegant, this method typically necessitates cryogenic reaction conditions, often requiring temperatures as low as -50°C to control regioselectivity and prevent side reactions. Maintaining such extreme低温 on a multi-kilogram or ton scale demands specialized refrigeration infrastructure and incurs prohibitive energy costs. Additionally, the Bartoli reaction is notoriously dilute, frequently requiring solvent volumes up to 100 times that of the reactants to achieve acceptable conversion. This massive solvent burden not only inflates raw material costs but also creates a bottleneck in reactor occupancy time, as the subsequent concentration and solvent recovery steps become excessively time-consuming. The combination of low yields (typically around 50 percent) and high operational complexity renders these conventional methods economically unviable for modern supply chains.
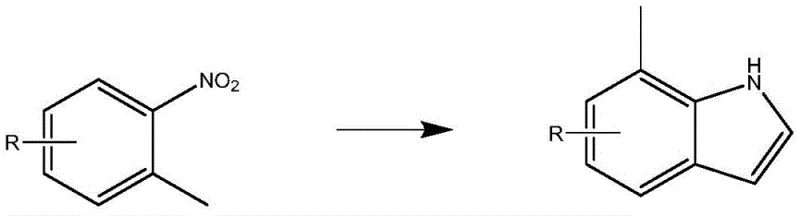
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN112457235A utilizes a thermodynamically favorable condensation between a substituted nitrobenzene derivative and pyrrole, mediated by N,N-dimethylformamide dimethyl acetal (DMFDMA). This innovative route eliminates the need for hazardous Lewis acids like TiCl4 and avoids the energy-intensive cryogenic requirements of the Bartoli synthesis. The process operates at elevated temperatures between 120°C and 150°C, specifically optimized at 140°C, which facilitates rapid reaction kinetics without the risk of thermal runaway or polymerization. The reaction medium employs a mixed solvent system of DMF and toluene, which allows for the efficient removal of byproducts via azeotropic distillation, driving the equilibrium toward the desired intermediate. This shift from low-temperature, dilute conditions to high-temperature, concentrated reflux represents a paradigm shift in process chemistry, enabling the use of standard multipurpose reactors commonly found in fine chemical manufacturing facilities.
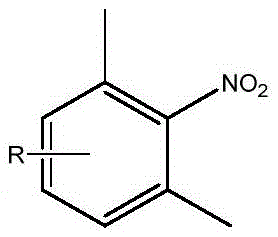
Mechanistic Insights into DMFDMA-Mediated Cyclization
The core of this technological advancement lies in the mechanistic role of DMFDMA as a versatile formylating and condensing agent. In the first step, DMFDMA reacts with the methyl group ortho to the nitro group on the aromatic ring, likely forming an enamine or imine intermediate that activates the benzylic position for nucleophilic attack by pyrrole. This activation is crucial because it lowers the energy barrier for the C-C bond formation that constructs the indole skeleton. Unlike electrophilic aromatic substitutions that often suffer from poor regiocontrol, this mechanism leverages the strong electron-withdrawing nature of the nitro group to direct the cyclization precisely, ensuring the formation of the 7-substituted isomer with high fidelity. The subsequent elimination of dimethylamine drives the reaction forward, resulting in a stable nitro-indole intermediate that can be isolated in high purity. This mechanistic pathway is inherently cleaner than radical-based or metal-catalyzed couplings, as it generates fewer heavy metal impurities and avoids the formation of complex tar-like byproducts.
Following the construction of the carbon framework, the second step involves a catalytic hydrogenation that serves a dual purpose: reducing the nitro group to an amine and facilitating the final aromatization or stabilization of the indole ring. The use of 5% Palladium on Carbon (Pd/C) as a heterogeneous catalyst is a strategic choice for process safety and ease of workup. The reduction proceeds smoothly in ethanol under atmospheric hydrogen pressure, avoiding the need for high-pressure autoclaves which require stringent safety certifications. The mechanism likely involves the adsorption of the nitro-intermediate onto the palladium surface, where sequential hydrogen transfer reduces the nitrogen-oxygen bonds. The resulting amine spontaneously cyclizes or rearranges if necessary to form the final 7-methylindole structure. Crucially, because the catalyst is solid, it can be removed entirely by simple filtration through a bed of diatomite, leaving the product solution free of palladium residues. This eliminates the need for expensive metal scavenger resins, which are often a significant cost driver in API manufacturing.
How to Synthesize 7-Methylindole Efficiently
The operational simplicity of this patent makes it highly attractive for technology transfer teams looking to implement new capacity quickly. The procedure begins with the preparation of the reaction vessel by charging a mixture of DMF and toluene, which is then heated to remove moisture and stabilize the temperature. Once the system reaches the target range of 140°C, the starting nitro-compound, pyrrole, and DMFDMA are introduced in a precise molar ratio, typically slightly excessing the pyrrole and DMFDMA to ensure complete conversion of the valuable aromatic starting material. The mixture is maintained under reflux for a period ranging from 24 to 50 hours, allowing sufficient time for the condensation to reach completion. Upon cooling, the reaction mass is quenched with water, and the product is extracted into the organic phase, dried, and concentrated to yield the intermediate. For the detailed standardized synthetic steps and specific workup parameters, please refer to the guide below.
- Condense compound (I) with pyrrole and DMFDMA in a DMF/toluene mixture at 140°C under reflux for 24-50 hours to form the nitro-intermediate.
- Isolate the intermediate via aqueous workup and toluene extraction, followed by drying and solvent removal.
- Perform catalytic hydrogenation of the intermediate using 5% Pd/C in ethanol under reflux to cyclize and reduce the nitro group to the final indole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound benefits for supply chain resilience and cost management. The elimination of cryogenic cooling and the reduction in solvent usage directly translate to lower utility costs and reduced waste disposal fees, which are increasingly significant factors in the total cost of ownership for chemical intermediates. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to batch-to-batch variability caused by minor fluctuations in temperature or mixing efficiency, thereby enhancing supply reliability for downstream customers.
- Cost Reduction in Manufacturing: The transition away from expensive and hazardous reagents like titanium tetrachloride and chloroacetonitrile significantly lowers the raw material bill of materials. Additionally, the ability to operate at atmospheric pressure and standard reflux temperatures removes the capital expenditure burden associated with specialized high-pressure or cryogenic reactors. The high yield of the first step, reported to be over 90 percent in examples, minimizes the loss of expensive starting materials, ensuring that the cost per kilogram of the final active ingredient is drastically optimized compared to legacy routes.
- Enhanced Supply Chain Reliability: The use of commodity chemicals such as ethanol, toluene, and DMF ensures that the supply chain is not vulnerable to shortages of exotic or highly regulated precursors. The simplified workup procedure, which relies on standard liquid-liquid extraction and filtration rather than complex chromatography or crystallization from difficult solvent systems, reduces the cycle time per batch. This increased throughput capability allows manufacturers to respond more agilely to market demand spikes, reducing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to the TiCl4 route, which produces corrosive acidic waste streams requiring neutralization. The heterogeneous nature of the Pd/C catalyst allows for potential recycling or easier disposal, aligning with green chemistry principles. The absence of extreme conditions makes the process inherently safer to scale from pilot plant to multi-ton commercial production, reducing the risk of plant shutdowns due to safety incidents and ensuring continuous availability for global clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for process engineers.
Q: What are the primary advantages of this new 7-methylindole synthesis route over the Bartoli method?
A: Unlike the Bartoli method which requires cryogenic temperatures (-50°C) and excessive solvent volumes (100x reactant volume), this new patent utilizes standard reflux conditions (120-150°C) and significantly reduced solvent ratios, drastically lowering energy consumption and equipment costs.
Q: How does the catalyst removal process impact the purity of the final product?
A: The process employs heterogeneous 5% Pd/C catalyst which can be easily removed via simple suction filtration through diatomite. This eliminates the need for complex metal scavenging resins often required with homogeneous catalysts, ensuring high purity and simplifying the downstream processing workflow.
Q: Is this synthesis method suitable for multi-ton commercial production?
A: Yes, the patent explicitly addresses scalability issues found in prior art. By avoiding polymerization-prone reagents like chloroacetonitrile and eliminating the need for specialized low-temperature reactors, the process is designed for robust operation in standard industrial glass-lined or stainless steel reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Methylindole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex heterocycles like 7-methylindole requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development programs. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a stable, cost-effective supply of this critical intermediate.
