Scalable Green Synthesis of 2,4-Dihydroxybenzophenone for High-Performance UV Stabilizer Production
Scalable Green Synthesis of 2,4-Dihydroxybenzophenone for High-Performance UV Stabilizer Production
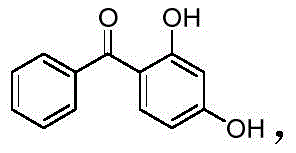
The global demand for high-performance ultraviolet (UV) stabilizers continues to surge, driven by stringent requirements in the aerospace, automotive, and pharmaceutical sectors. At the forefront of this market is 2,4-dihydroxybenzophenone, widely known as UV-0, a critical compound valued for its exceptional ability to absorb harmful UV radiation and protect polymer matrices from degradation. A recent technological breakthrough, detailed in patent CN112142576B, introduces a revolutionary clean production method that fundamentally alters the manufacturing landscape for this essential fine chemical. Unlike traditional routes that rely on harsh Lewis acids and generate substantial hazardous waste, this novel approach utilizes trichlorotoluene and resorcinol in a benign water-ethanol system. This shift not only promises significant environmental benefits through zero wastewater discharge but also offers a robust pathway for producing high-purity intermediates suitable for sensitive applications. For R&D directors and supply chain leaders, understanding the mechanistic nuances and commercial implications of this patent is vital for securing a competitive edge in the specialty chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2,4-dihydroxybenzophenone has been plagued by significant environmental and operational inefficiencies inherent to classical Friedel-Crafts acylation. The most common legacy method involves the reaction of benzoyl chloride with resorcinol in the presence of stoichiometric amounts of Lewis acid catalysts such as aluminum chloride (AlCl3) or zinc chloride. While chemically effective, this process generates massive quantities of acidic sludge and waste salt upon quenching, creating a severe disposal burden and increasing the overall cost of compliance. Furthermore, alternative methods utilizing trichlorotoluene often require aggressive catalytic systems or organic solvents that lead to product discoloration and the release of noxious waste gases. Another existing route employs benzoic acid and resorcinol catalyzed by phosphotungstic acid; however, this method suffers from high catalyst costs, elevated reaction temperatures, and prolonged reaction times, rendering it economically unviable for large-scale continuous production. These conventional pathways collectively represent a bottleneck for modern manufacturers seeking to align with green chemistry principles and reduce their carbon footprint.
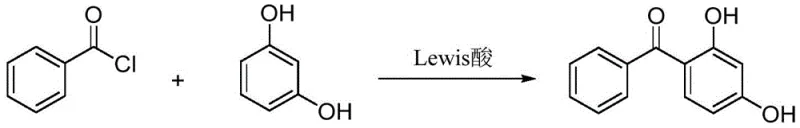
The Novel Approach
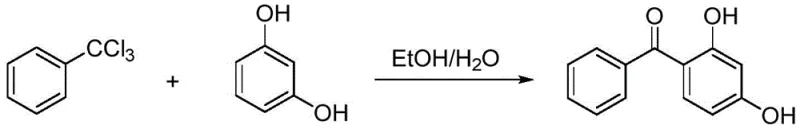
In stark contrast to these legacy processes, the method disclosed in patent CN112142576B leverages a unique reactivity profile of trichlorotoluene in a mixed solvent system of water and ethanol. This innovative route bypasses the need for pre-activated acyl chlorides or expensive solid acid catalysts, instead relying on the in situ generation of reactive intermediates under mild thermal conditions (50-60°C). The use of a water-ethanol mixture is particularly transformative, as it serves both as a reaction medium and a facilitator for the hydrolysis of the trichloromethyl group, driving the acylation forward while maintaining a homogeneous or semi-homogeneous phase that enhances mass transfer. Crucially, this process is designed with a circular economy mindset: the mother liquor is not discarded but is instead concentrated to recover ethanol for reuse, and the filtrate is treated to recover benzoic acid as a valuable secondary product. This holistic approach effectively closes the material loop, eliminating the discharge of wastewater and turning potential waste streams into revenue-generating byproducts, thereby drastically improving the process economics.
Mechanistic Insights into Aqueous-Phase Acylation
The core chemical innovation of this process lies in the controlled hydrolysis and subsequent acylation of the trichloromethyl group attached to the benzene ring. In the presence of water and ethanol, the trichlorotoluene undergoes a partial hydrolysis to form a reactive acylating species, likely an acyl chloride or a mixed orthoester intermediate, which is sufficiently electrophilic to attack the electron-rich resorcinol ring. The presence of ethanol plays a dual role: it solubilizes the organic reactants, ensuring intimate contact between the hydrophobic trichlorotoluene and the hydrophilic resorcinol, and it may also participate in stabilizing the transition state of the reaction. Unlike strong Lewis acid catalysis which activates the carbonyl aggressively but non-selectively, this solvent-mediated activation offers a milder pathway that minimizes side reactions such as poly-acylation or resinification, which are common causes of product darkening in traditional methods. The reaction temperature is carefully maintained between 50°C and 60°C, a range that provides sufficient kinetic energy for the transformation without triggering the decomposition of the sensitive phenolic groups or the excessive hydrolysis of the trichloromethyl group to benzoic acid before acylation can occur.
Impurity control is another critical aspect where this mechanism excels, particularly regarding the management of benzoic acid, a competing byproduct formed via the complete hydrolysis of trichlorotoluene. The patent details a sophisticated workup procedure where the reaction mixture is centrifuged to isolate the crude product, leaving benzoic acid and hydrogen chloride in the mother liquor. By adjusting the pH of the filtrate with sodium hydroxide, the benzoic acid is converted into water-soluble sodium benzoate, allowing for effective separation from organic impurities via activated carbon decolorization. Subsequent acidification with hydrochloric acid precipitates high-purity benzoic acid, which can be recycled or sold. This rigorous separation strategy ensures that the final 2,4-dihydroxybenzophenone product achieves a GC purity of over 99.2%, meeting the stringent specifications required for pharmaceutical and high-end polymer applications without the need for energy-intensive recrystallization steps typically associated with removing Lewis acid complexes.
How to Synthesize 2,4-Dihydroxybenzophenone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and minimize byproduct formation. The process begins with the preparation of a reaction mixture containing resorcinol, water, and ethanol in a specific mass ratio, typically favoring water to facilitate the hydrolysis mechanism while maintaining solubility. Trichlorotoluene is added dropwise over a period of 2 to 4 hours to manage the exotherm and control the concentration of the reactive intermediate. Following the addition, the temperature is raised slightly to drive the reaction to completion. The downstream processing is equally critical, involving a sequence of concentration, filtration, alkalization, and acidification to recover solvents and byproducts. For a detailed, step-by-step standard operating procedure including exact masses and equipment specifications, please refer to the technical guide below.
- Mix resorcinol, water, and ethanol in a reactor, heat to 30-40°C, and dropwise add trichlorotoluene over 2-4 hours.
- Heat the mixture to 50-60°C for 1-4 hours, then cool and centrifuge to isolate the crude 2,4-dihydroxybenzophenone solid.
- Concentrate the mother liquor to recover ethanol, then alkalize and acidify the filtrate to recover benzoic acid byproducts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this clean production technology represents a strategic opportunity to optimize costs and mitigate supply risks. Traditional manufacturing routes for UV-0 are increasingly vulnerable to regulatory pressures regarding hazardous waste disposal and the volatility of Lewis acid prices. By transitioning to the trichlorotoluene-based aqueous method, manufacturers can eliminate the procurement of expensive and corrosive catalysts like aluminum chloride and phosphotungstic acid, leading to a direct reduction in raw material expenditure. Furthermore, the ability to recover and recycle ethanol significantly lowers solvent consumption costs, while the recovery of benzoic acid creates an additional revenue stream that offsets production expenses. This multi-faceted cost reduction strategy enhances the overall margin profile of the product, making it more resilient against market fluctuations.
- Cost Reduction in Manufacturing: The elimination of stoichiometric Lewis acids removes the substantial cost associated with purchasing, handling, and disposing of these hazardous materials. Additionally, the simplified workup procedure reduces labor and utility costs related to waste treatment and complex purification steps. The recovery of benzoic acid transforms a waste liability into a saleable asset, further improving the economic efficiency of the plant.
- Enhanced Supply Chain Reliability: The raw materials for this process, trichlorotoluene and resorcinol, are commodity chemicals with stable and abundant global supplies, reducing the risk of feedstock shortages that often plague specialized catalyst-dependent processes. The robustness of the aqueous-ethanol system also allows for more flexible manufacturing schedules, as the reaction is less sensitive to moisture and atmospheric conditions compared to anhydrous Friedel-Crafts reactions.
- Scalability and Environmental Compliance: The "zero discharge" claim of this process aligns perfectly with increasingly strict environmental regulations worldwide, future-proofing the supply chain against potential shutdowns due to non-compliance. The absence of heavy metal catalysts and acidic sludge simplifies the permitting process for capacity expansion, enabling faster scale-up from pilot to commercial production volumes without the need for expensive waste treatment infrastructure upgrades.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis route. These answers are derived directly from the experimental data and claims presented in patent CN112142576B, providing a reliable basis for feasibility assessments. Understanding these details is crucial for technical teams evaluating the transition from legacy processes to this modern, sustainable methodology.
Q: What are the primary advantages of the trichlorotoluene route over traditional Friedel-Crafts acylation?
A: The trichlorotoluene route eliminates the need for stoichiometric amounts of corrosive Lewis acids like aluminum chloride, thereby removing the generation of hazardous acidic sludge and simplifying the downstream purification process significantly.
Q: How does this patent address environmental concerns regarding wastewater?
A: The process utilizes a closed-loop system where the mother liquor is concentrated to recover ethanol for reuse, and byproduct benzoic acid is recovered via alkalization and acidification, theoretically achieving zero wastewater discharge.
Q: What purity levels can be achieved with this green synthesis method?
A: According to the provided patent data, the method consistently yields product with a GC content exceeding 99.2%, demonstrating high selectivity and effective impurity control without complex chromatographic separation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Dihydroxybenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just an environmental imperative but a commercial necessity. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate the innovations described in patent CN112142576B into commercial reality. Our facilities are equipped to handle diverse synthetic pathways, ranging from small-scale optimization to full-scale manufacturing, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering 2,4-dihydroxybenzophenone that meets stringent purity specifications, supported by our rigorous QC labs that ensure every batch complies with the highest international standards for pharmaceutical and polymer additives.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Whether you are looking to secure a long-term supply of high-quality UV-0 or need assistance in optimizing your current manufacturing process, our team is ready to provide a Customized Cost-Saving Analysis. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your specific volume requirements and quality targets.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
