Advanced Synthetic Route for Lotilaner: Enhancing Purity and Scalability for Veterinary APIs
Advanced Synthetic Route for Lotilaner: Enhancing Purity and Scalability for Veterinary APIs
The pharmaceutical and veterinary industries are constantly seeking more efficient pathways to produce complex active ingredients, and the recent patent CN115504971B presents a groundbreaking advancement in the synthesis of Lotilaner, a potent isoxazoline-class parasiticide. This novel methodology addresses critical bottlenecks found in earlier generations of synthetic routes, specifically targeting the high cost of raw materials and the difficulties associated with purification. By re-engineering the sequence of functional group introductions, this process allows for the construction of the core isoxazoline scaffold with unprecedented efficiency. For R&D directors and procurement managers alike, understanding this shift is vital, as it represents a move away from reliance on scarce, expensive precursors toward a more robust, commodity-chemical-based supply chain. The implications for cost reduction in veterinary drug manufacturing are substantial, offering a clearer path to commercial viability without compromising on the stringent quality standards required for animal health products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art, such as the routes disclosed in PCT patents WO2022020585A1 and WO2014090918A1, relied heavily on a strategy where halogen or carboxyl groups were introduced onto the thiophene ring before the formation of the isoxazoline ring. While chemically feasible on a small scale, this approach suffers from severe economic and practical drawbacks when viewed through the lens of industrial production. The primary issue lies in the availability and cost of these pre-functionalized thiophene starting materials, which are often niche chemicals with limited global supply chains, leading to volatile pricing and potential supply disruptions. Furthermore, the introduction of these groups early in the synthesis tends to reduce the overall polarity of the intermediate molecules. Low polarity compounds are notoriously difficult to purify using standard crystallization techniques, often necessitating expensive and time-consuming chromatographic separations. This not only increases the energy consumption of the manufacturing process but also introduces risks of residual solvent contamination, complicating the regulatory approval process for the final API.
The Novel Approach
In stark contrast, the method described in patent CN115504971B flips the synthetic logic by prioritizing the construction of the carbon skeleton before fine-tuning the functional groups. This route begins with a straightforward condensation reaction between readily available 2-acetyl-4-methylthiophene and a trifluorinated ketone. By delaying the introduction of complex side chains until after the core heterocyclic ring is established, the process maintains better control over molecular polarity throughout the synthesis. This strategic adjustment ensures that intermediates remain amenable to simple purification methods, such as aqueous washing and recrystallization, rather than relying on complex column chromatography. The result is a streamlined workflow that minimizes waste generation and maximizes throughput. For supply chain heads, this translates to a more predictable production schedule and a significant reduction in the lead time for high-purity veterinary drug intermediates, ensuring a steady flow of material for formulation teams.
Mechanistic Insights into Isoxazoline Ring Formation and Functionalization
The heart of this synthetic innovation lies in the precise construction of the isoxazoline ring, a critical pharmacophore responsible for Lotilaner's GABA-gated chloride channel antagonism. The process initiates with a base-catalyzed Claisen-Schmidt condensation to form a chalcone-like intermediate (Intermediate I), which serves as the dipolarophile for the subsequent cyclization. The key transformation occurs when this enone system reacts with hydroxylamine under controlled alkaline conditions. This reaction proceeds via a 1,3-dipolar cycloaddition mechanism, effectively closing the five-membered isoxazoline ring while preserving the stereochemical integrity required for biological activity. The careful control of temperature during this step, typically maintained between 10-20°C, is crucial to prevent the formation of regioisomers or over-oxidation byproducts. By optimizing the stoichiometry of hydroxylamine and the base, the process achieves a remarkable conversion rate, minimizing the presence of unreacted starting materials that could act as genotoxic impurities in the final drug substance.
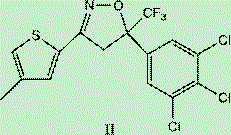
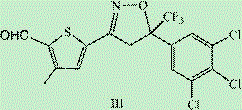
Following the successful formation of the isoxazoline core (Intermediate II), the synthesis proceeds to the functionalization of the thiophene moiety. This is achieved through a Vilsmeier-Haack formylation reaction, utilizing phosphorus oxychloride and DMF to introduce an aldehyde group at the 2-position of the thiophene ring. This step is particularly elegant because it activates the aromatic ring for the final coupling reaction without disturbing the sensitive isoxazoline structure. The resulting aldehyde (Intermediate III) is a versatile synthon that can be easily purified due to its distinct polarity profile compared to the reaction byproducts. The ability to isolate Intermediate III in high purity sets the stage for the final oxidative coupling, ensuring that the final API meets the rigorous specifications demanded by global regulatory bodies. This mechanistic clarity provides R&D teams with the confidence to scale the process, knowing exactly where potential impurities might arise and how to mitigate them.
How to Synthesize Lotilaner Efficiently
The transition from laboratory bench to commercial reactor requires a standardized protocol that balances reaction kinetics with safety and ease of operation. The patented method outlines a clear four-stage sequence that begins with the condensation of the thiophene and ketone precursors, followed by the critical cyclization step to build the heterocyclic core. Subsequent formylation activates the molecule for the final assembly, where the trifluoroethyl amide side chain is attached via an oxidative coupling mechanism. Each step has been optimized to use common solvents like methyl tert-butyl ether and ethanol, which are easy to recover and recycle, further enhancing the green chemistry profile of the synthesis. For process chemists looking to implement this route, the detailed parameters regarding temperature ramps and reagent addition rates provided in the patent serve as a robust foundation for technology transfer. The detailed standardized synthesis steps are outlined in the guide below.
- Condense 2-acetyl-4-methylthiophene with trifluoro ketone to form the chalcone intermediate.
- Perform cyclization with hydroxylamine to construct the isoxazoline core structure.
- Execute Vilsmeier-Haack formylation to introduce the aldehyde group on the thiophene ring.
- Complete the synthesis via oxidative coupling with the trifluoroethyl amide side chain.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers compelling advantages that extend far beyond simple yield improvements. The most significant impact is seen in the realm of raw material sourcing; by utilizing commodity chemicals like 2-acetyl-4-methylthiophene instead of exotic halogenated derivatives, manufacturers can insulate themselves from the volatility of the specialty chemical market. This shift fundamentally alters the cost structure of the API, allowing for more competitive pricing in the veterinary sector without sacrificing margins. Furthermore, the simplified purification protocols mean that manufacturing facilities do not need to invest in specialized chromatography equipment or consume vast quantities of silica gel and eluents. This reduction in operational complexity directly correlates to lower overhead costs and a smaller environmental footprint, aligning with the increasing demand for sustainable manufacturing practices in the pharmaceutical industry.
- Cost Reduction in Manufacturing: The elimination of expensive pre-functionalized starting materials creates a direct and substantial decrease in the Bill of Materials (BOM) cost. Additionally, the high efficiency of the cyclization and formylation steps reduces the amount of raw material wasted as byproducts, further driving down the cost per kilogram of the final active ingredient. The ability to use standard solvents and simple workup procedures like filtration and crystallization also lowers utility costs associated with solvent recovery and waste treatment. These cumulative savings allow for a more aggressive pricing strategy in the marketplace while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Relying on widely available building blocks mitigates the risk of supply chain disruptions that often plague projects dependent on single-source custom synthons. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without significant re-validation, providing flexibility in case of regional logistical issues. This reliability is crucial for meeting the consistent demand of the veterinary market, where treatment schedules for parasites like ticks and fleas are seasonal but predictable. A stable supply of high-quality intermediates ensures that formulation partners can maintain their production schedules without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction exotherms that are manageable and workup procedures that do not require exotic equipment. The high purity of the intermediates reduces the load on wastewater treatment plants, as fewer organic impurities need to be degraded before discharge. Moreover, the high atom economy of the condensation and cyclization steps aligns with green chemistry principles, reducing the overall E-factor of the process. This makes the technology attractive not just for its economic benefits, but also for its compliance with increasingly stringent environmental regulations governing chemical manufacturing.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common questions regarding the implementation and performance of this synthetic route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these details is key to assessing the feasibility of adopting this method for your own production lines or sourcing strategies. The following section addresses purity benchmarks, raw material availability, and scalability potential.
Q: What is the purity level achievable with this new Lotilaner synthesis method?
A: The patented process achieves an HPLC purity of up to 99.90% for the final Lotilaner product, significantly reducing the burden on downstream purification teams.
Q: How does this route improve upon previous synthetic methods regarding raw materials?
A: Unlike prior art requiring expensive pre-halogenated thiophenes, this method utilizes cheap and readily available 2-acetyl-4-methylthiophene, drastically lowering material costs.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process features high yields (up to 93.3% in the final step) and simple workup procedures like crystallization, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lotilaner Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires a partner with deep technical expertise and a commitment to quality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in the lab are faithfully reproduced at the plant scale. We understand the nuances of handling fluorinated intermediates and heterocyclic compounds, employing stringent purity specifications and rigorous QC labs to guarantee that every batch of Lotilaner meets the highest international standards. Our infrastructure is designed to support the complex chemistry involved in this synthesis, providing a secure and reliable source for your veterinary API needs.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our expertise in process optimization, we can help you navigate the complexities of scaling this novel route, ensuring a smooth transition from development to full-scale commercialization. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities can add value to your Lotilaner project.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
