Advanced Manufacturing of 5-p-tolyl-1-2-4-triazine-3-2H-ketone for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic intermediates that serve as critical building blocks for novel therapeutic agents. Patent CN114478417A introduces a significant advancement in the preparation of 5-p-tolyl-1, 2, 4-triazine-3(2H)-ketone, a valuable scaffold in medicinal chemistry. This disclosure outlines a streamlined three-step methodology that transforms inexpensive 4-methylacetophenone into the target triazine derivative with remarkable efficiency. By leveraging stepwise oxidation and cyclization strategies, the process addresses common bottlenecks associated with heterocycle synthesis, such as low yields and complex purification requirements. For R&D directors and procurement specialists, this technology represents a viable pathway to secure high-quality intermediates while optimizing manufacturing economics. The method's reliance on standard chemical transformations ensures that it can be rapidly integrated into existing production facilities without necessitating specialized equipment or hazardous high-pressure conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to substituted 1, 2, 4-triazines often suffer from excessive step counts and the utilization of unstable or difficult-to-handle precursors. Many legacy processes require the pre-formation of hydrazonoyl halides or the use of toxic nitrile oxides, which pose significant safety risks and environmental compliance challenges during scale-up. Furthermore, conventional methods frequently employ harsh acidic or basic conditions that can lead to the degradation of sensitive functional groups, resulting in complex impurity profiles that are difficult to separate. The reliance on precious metal catalysts in some cross-coupling approaches further inflates the cost of goods sold, making the final intermediate less competitive in a price-sensitive market. Additionally, the lack of atom economy in older routes generates substantial chemical waste, complicating waste stream management and increasing the overall environmental footprint of the manufacturing process.
The Novel Approach
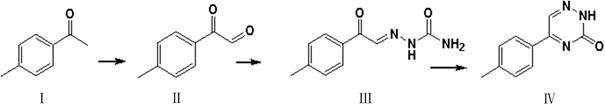
In stark contrast, the methodology described in patent CN114478417A utilizes a direct and convergent strategy starting from the commercially abundant 4-methylacetophenone. This novel approach capitalizes on the inherent reactivity of the methyl ketone moiety, transforming it directly into an alpha-keto aldehyde functionality through selective oxidation. This intermediate then undergoes a straightforward condensation with semicarbazide hydrochloride, a stable and inexpensive reagent, to form the requisite hydrazone precursor. The final cyclization step is achieved under mild alkaline conditions, driving the formation of the triazine ring with high selectivity. This streamlined sequence not only reduces the number of isolation steps but also minimizes the accumulation of by-products, thereby enhancing the overall purity of the crude product before final crystallization. The simplicity of the operational protocol allows for greater flexibility in batch sizing and facilitates easier technology transfer between different manufacturing sites.
Mechanistic Insights into Selenium Dioxide Oxidation and Cyclization
The cornerstone of this synthetic route is the initial oxidation step, typically employing selenium dioxide (SeO2) as the oxidant to convert the methyl group of 4-methylacetophenone into an aldehyde function, yielding the alpha-keto aldehyde intermediate (Compound II). This transformation, known as the Riley oxidation, proceeds via an ene reaction mechanism followed by a [2,3]-sigmatropic rearrangement, which is highly specific for activating allylic or benzylic positions adjacent to carbonyl groups. The use of SeO2 in solvents like dichloromethane or dioxane ensures high conversion rates while maintaining the integrity of the aromatic ring. Alternatively, the patent discloses the use of nitric and sulfuric acid mixtures, offering a cost-effective variation for facilities equipped to handle strong mineral acids. The precise control of reaction temperature between 20°C and 60°C is critical to preventing over-oxidation to the corresponding carboxylic acid, ensuring the preservation of the aldehyde functionality required for the subsequent condensation step.
Following the formation of the alpha-keto aldehyde, the mechanism shifts to nucleophilic addition-elimination as semicarbazide hydrochloride reacts with the aldehyde carbonyl to form the semicarbazone derivative (Compound III). This step is reversible and is driven to completion by the removal of water or the precipitation of the product in alcoholic media. The final transformation involves an intramolecular cyclization where the terminal amine of the semicarbazone attacks the adjacent ketone carbonyl, facilitated by the presence of a base such as sodium carbonate. This cyclodehydration process closes the six-membered triazine ring, accompanied by the loss of a water molecule to establish the aromatic character of the heterocycle. The use of mild bases prevents the hydrolysis of the newly formed amide bond within the ring, ensuring high stability of the final 5-p-tolyl-1, 2, 4-triazine-3(2H)-ketone product.
How to Synthesize 5-p-tolyl-1-2-4-triazine-3-2H-ketone Efficiently
The synthesis of this high-value intermediate is designed for operational simplicity, allowing chemists to achieve high yields with minimal technical difficulty. The process begins with the careful selection of the oxidant, where selenium dioxide provides superior selectivity, though mineral acid variants offer economic benefits for large-scale runs. Following the oxidation, the crude alpha-keto aldehyde can often be used directly or after a simple recrystallization, reducing solvent consumption. The condensation with semicarbazide is exothermic and requires controlled addition to manage heat release, ensuring the formation of large, filterable crystals of the hydrazone intermediate. Finally, the cyclization in aqueous base is a robust reaction that tolerates minor variations in pH and temperature, making it ideal for industrial reactors. Detailed standardized synthesis steps see the guide below.
- Oxidize 4-methylacetophenone using selenium dioxide or nitric acid/sulfuric acid mixtures in organic solvents like dichloromethane to form the alpha-keto aldehyde intermediate.
- Condense the resulting alpha-keto aldehyde with semicarbazide hydrochloride in alcoholic solvents such as methanol to generate the hydrazone derivative.
- Perform base-catalyzed cyclization of the hydrazone intermediate using alkali carbonates or hydroxides in aqueous media at elevated temperatures to yield the final triazine ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits in terms of cost stability and supply security. The primary driver of cost reduction lies in the utilization of 4-methylacetophenone, a commodity chemical produced in massive volumes for the fragrance and polymer industries, ensuring a stable and low-cost entry point for the synthesis. By eliminating the need for exotic catalysts or specialized ligands, the process significantly lowers the raw material expenditure per kilogram of the final product. Furthermore, the simplified three-step sequence reduces the total processing time and labor hours required, directly translating to lower manufacturing overheads. The ability to perform reactions in common solvents like methanol and water also simplifies solvent recovery and recycling protocols, contributing to a more sustainable and cost-efficient operation.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavenging steps and rigorous heavy metal testing, which are often cost-prohibitive in pharmaceutical manufacturing. By relying on main group elements like selenium or nitrogen-based oxidants, the process avoids the volatility associated with precious metal pricing. Additionally, the high yields reported in the patent examples indicate a very efficient use of starting materials, minimizing waste disposal costs and maximizing the output from each batch. The overall reduction in unit operations means less energy consumption for heating, cooling, and agitation, further driving down the utility costs associated with production.
- Enhanced Supply Chain Reliability: Since all key reagents, including semicarbazide hydrochloride and sodium carbonate, are widely available from multiple global suppliers, the risk of supply disruption is drastically minimized. This multi-sourcing capability ensures that production schedules can be maintained even if one vendor faces logistical issues. The robustness of the chemistry also means that the process is less sensitive to minor fluctuations in reagent quality, reducing the rate of batch failures and ensuring consistent delivery timelines to downstream customers. This reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery windows required by major pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable as it avoids the use of pyrophoric reagents or high-pressure hydrogenation steps that require specialized containment. The aqueous workups and simple filtration steps are easily adapted from laboratory glassware to multi-ton reactor systems without significant engineering changes. From an environmental perspective, the generation of waste is primarily inorganic salts and aqueous streams, which are easier to treat than complex organic solvent mixtures containing heavy metals. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the long-term liability associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 5-p-tolyl-1, 2, 4-triazine-3(2H)-ketone. These answers are derived directly from the technical specifications and advantageous effects outlined in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their broader supply networks. The responses cover aspects of purity, scalability, and raw material sourcing to provide a comprehensive overview of the technology's capabilities.
Q: What are the key advantages of this synthesis route over conventional methods?
A: This patented method utilizes readily available 4-methylacetophenone as a starting material and achieves the target molecule in only three efficient steps. It avoids the use of expensive transition metal catalysts and employs mild reaction conditions, significantly simplifying the purification process and reducing overall production costs compared to traditional multi-step pathways.
Q: How does the process ensure high purity for pharmaceutical applications?
A: The process incorporates specific recrystallization steps, such as hot water recrystallization for the intermediate and controlled precipitation for the final product. The use of selective oxidants like selenium dioxide minimizes side reactions, while the final aqueous workup effectively removes inorganic salts, ensuring the final API intermediate meets stringent purity specifications required for downstream drug synthesis.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method is highly scalable as it relies on standard unit operations like reflux, filtration, and rotary evaporation which are easily adapted to large-scale reactors. The reagents used, including semicarbazide hydrochloride and sodium carbonate, are commodity chemicals available in bulk quantities, ensuring a robust and continuous supply chain for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-p-tolyl-1-2-4-triazine-3-2H-ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercialization. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-p-tolyl-1, 2, 4-triazine-3(2H)-ketone meets the exacting standards required for API synthesis. Our commitment to technical excellence allows us to optimize the patented route further, delivering a product that balances cost-efficiency with uncompromising quality.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and logistical constraints. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your project timelines and budgetary goals efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
