Advanced Manufacturing of Pyrimidinetrione Derivatives via Novel Carbamoylation Pathways
The pharmaceutical industry continuously seeks robust synthetic routes for active pharmaceutical ingredients (APIs) and their key intermediates that balance efficiency with purity. A pivotal advancement in this domain is documented in Chinese Patent CN1034806C, which details a sophisticated process for preparing pyrimidinetrione derivatives, specifically focusing on compounds like febarbamate. This patent addresses longstanding challenges in heterocyclic chemistry by introducing a novel pathway that bypasses the formation of difficult-to-separate byproducts common in earlier methodologies. The core innovation lies in the strategic use of isocyanate intermediates to install the carbamate functionality, rather than relying solely on direct alkylation which often suffers from poor regioselectivity. For R&D directors and process chemists, understanding this mechanism offers a blueprint for optimizing the synthesis of similar sedative-hypnotic agents. By shifting the synthetic logic from simple nucleophilic substitution to a controlled addition-hydrolysis sequence, manufacturers can achieve superior impurity profiles. This report analyzes the technical nuances of this patent to demonstrate how it serves as a reliable foundation for a reliable pharmaceutical intermediate supplier aiming to deliver high-quality materials to the global market.
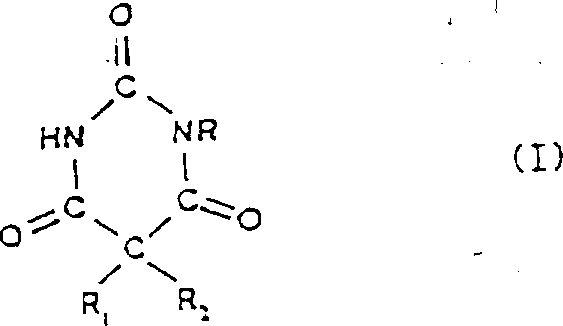
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5,5-disubstituted pyrimidinetriones involved the alkylation of sodium salts of malonylurea derivatives. As highlighted in the background of the patent data, this traditional approach is fraught with significant chemical engineering hurdles. The reaction typically proceeds in a heterogeneous, highly viscous mass that is difficult to control thermally and mechanically. A major drawback is the inevitable formation of a complex mixture containing unconverted starting materials, the desired N'-monosubstituted product, and the undesired N,N'-disubstituted derivative. Separating these structurally similar components requires rigorous purification steps, often involving multiple distillations under reduced pressure, which are energy-intensive and result in substantial material loss. Furthermore, the alkylating agents used, such as 1-halo-2-carbamoyloxy-3-alkoxypropanes, are not only difficult to prepare but also prone to containing isomeric impurities that further complicate the reaction profile. These factors collectively drive up the cost of goods sold and extend the lead time for high-purity pharmaceutical intermediates, making the conventional route less attractive for modern, lean manufacturing environments.
The Novel Approach
In contrast, the methodology disclosed in the patent introduces a transformative strategy that fundamentally alters the bond construction sequence. Instead of attempting to attach the entire side chain in one difficult step, the process first establishes the hydroxy-propyl linkage on the pyrimidine ring and subsequently installs the carbamate group via an isocyanate reaction. This two-stage functionalization allows for much greater control over the reaction environment. The initial alkylation can be optimized using glycidyl ethers or halo-alcohols in the presence of organic bases, ensuring a cleaner formation of the hydroxy-intermediate. The subsequent reaction with dihalophosphinyl isocyanate or halosulfonyl isocyanate proceeds under mild conditions, typically between -10°C and 50°C, avoiding the harsh thermal stress of older methods. This approach effectively decouples the formation of the carbon-nitrogen bond from the carbon-oxygen bond formation, thereby minimizing side reactions. The result is a process that yields products basically free of the troublesome N,N'-disubstituted impurities, significantly simplifying downstream processing and enhancing the overall economic viability of producing these complex heterocycles.
Mechanistic Insights into Isocyanate-Mediated Carbamoylation
The heart of this synthetic innovation is the mechanistic pathway involving the reaction of the hydroxy-substituted pyrimidinetrione with reactive isocyanate species. When the compound of Formula (II), which contains a free hydroxyl group on the side chain, reacts with dihalophosphinyl isocyanate, it undergoes an electrophilic addition. The oxygen of the hydroxyl group attacks the electrophilic carbon of the isocyanate group, leading to the formation of a transient N-dihalophosphinyl intermediate. This step is critical because it protects the nitrogen atom of the newly formed carbamate linkage with a phosphinyl group, preventing unwanted further reactions or cyclizations. The patent specifies that this reaction is best conducted in anhydrous organic solvents such as toluene or dichloromethane to prevent premature hydrolysis. The use of substantially equimolar amounts of reactants ensures that the reagent is consumed efficiently without leaving excessive residues that could complicate purification. This precise stoichiometric control is a hallmark of fine chemical manufacturing, ensuring that the process remains atom-economical and waste-minimized while maintaining high conversion rates.
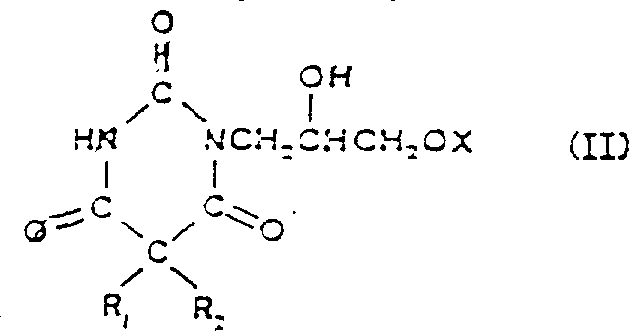
Following the formation of the protected intermediate, the process moves to a hydrolysis step that reveals the final carbamate functionality. Interestingly, the patent reveals that the nature of the solvent plays a crucial role in the stability of the intermediate bonds. In chlorinated solvents like dichloromethane, the cleavage of the N-P bond is hindered, allowing for the isolation of a novel intermediate of Formula (IV). This intermediate, an N-dihydroxyphosphoryl derivative, is itself a valuable entity, potentially serving as a water-soluble prodrug. However, for the synthesis of the target pyrimidinetrione, controlled hydrolysis is performed. By adjusting the pH to a range of 4 to 6 and heating the mixture to approximately 70°C, the phosphinyl or sulfonyl protecting group is cleanly removed. This hydrolysis is highly selective, breaking the P-N or S-N bond while leaving the sensitive carbamate ester linkage intact. This selectivity is paramount for maintaining the integrity of the final API structure and ensures that the impurity profile remains within stringent regulatory limits required for pharmaceutical applications.
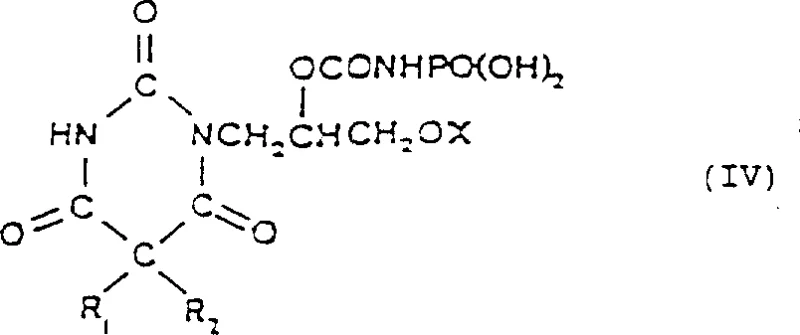
How to Synthesize Febarbamate Efficiently
The practical implementation of this chemistry involves a sequence of well-defined unit operations that are amenable to scale-up. The process begins with the preparation of the hydroxy-propyl precursor, followed by the carbamoylation and final hydrolysis. Each step has been optimized in the patent examples to demonstrate reproducibility and high yield. For instance, the use of triethylamine as a base in the initial alkylation step facilitates the reaction in homogeneous media, improving heat transfer and mixing efficiency. The subsequent isocyanate reaction is exothermic and requires careful temperature control, typically keeping the mixture below 30°C during addition to prevent runaway reactions. Finally, the hydrolysis is managed in a biphasic system, allowing for easy separation of the organic product from inorganic byproducts. Detailed standardized synthesis steps see the guide below.
- React 5,5-disubstituted barbituric acid with an alkylating agent (glycidyl ether or halo-alcohol) in the presence of an organic base like triethylamine at 70°C to form the hydroxy-propyl intermediate.
- Treat the hydroxy-propyl intermediate with dihalophosphinyl isocyanate or chlorosulfonyl isocyanate in an anhydrous solvent at temperatures below 30°C to generate the N-substituted intermediate.
- Hydrolyze the resulting intermediate in a biphasic system of water and organic solvent at pH 4-6 and 70°C to yield the final carbamate product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling advantages that go beyond mere chemical elegance. The primary benefit lies in the drastic simplification of the purification workflow. By eliminating the formation of N,N'-disubstituted byproducts, the need for complex and costly separation techniques such as preparative HPLC or multiple fractional distillations is removed. This directly translates to cost reduction in pharmaceutical intermediate manufacturing, as fewer processing steps mean lower energy consumption, reduced solvent usage, and shorter cycle times. Furthermore, the reliance on classical operations like extraction and crystallization means that the process can be executed in standard stainless steel reactors without the need for specialized equipment capable of handling high-viscosity slurries or extreme vacuum conditions. This compatibility with existing infrastructure reduces capital expenditure risks for contract manufacturing organizations (CMOs) looking to adopt the technology.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the improved selectivity and yield. By using an excess of the barbituric acid starting material relative to the alkylating agent, the formation of double-substituted waste is suppressed, maximizing the utilization of raw materials. This stoichiometric optimization ensures that expensive reagents are not wasted on generating unusable byproducts. Additionally, the ability to isolate intermediates in good yield without extensive chromatography lowers the operational costs significantly. The process avoids the use of hard-to-source alkylating agents that require rigorous purification, instead utilizing more readily available glycidyl ethers or halo-alcohols. These factors combine to create a leaner cost structure, allowing suppliers to offer competitive pricing while maintaining healthy margins in a volatile market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthetic routes. This patented method enhances reliability by shortening the critical path of production. The reaction times are reasonable, with the isocyanate step completing in about 1 hour at room temperature and the hydrolysis taking approximately 6 hours. This predictability allows for better production planning and inventory management. Moreover, the reagents involved, such as toluene, dichloromethane, and triethylamine, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages. The robustness of the reaction conditions, which tolerate slight variations in temperature and pH without catastrophic failure, further ensures consistent batch-to-batch quality. This stability is crucial for long-term supply agreements with multinational pharmaceutical companies that demand unwavering consistency.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process is designed for scalability. The avoidance of highly viscous reaction masses improves mixing and heat dissipation, which are critical safety parameters when scaling from kilograms to tons. The use of aqueous workups and the ability to recycle solvents like toluene align with green chemistry principles, reducing the environmental footprint of the manufacturing site. The hydrolysis step generates inorganic salts that are easily separated and disposed of according to standard waste treatment protocols. By minimizing the generation of hazardous organic waste and avoiding the use of heavy metal catalysts, the process simplifies regulatory compliance. This makes it an attractive option for facilities operating under strict environmental regulations, ensuring that the commercial scale-up of complex pharmaceutical intermediates can proceed without regulatory bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and claims found within the patent documentation to provide clarity on feasibility and performance. Understanding these details is essential for stakeholders evaluating the potential for technology transfer or procurement partnerships.
Q: How does the new process improve purity compared to traditional alkylation methods?
A: Traditional methods often result in mixtures of N-monosubstituted and N,N'-disubstituted byproducts due to non-selective alkylation. The patented process utilizes a specific carbamoylation followed by controlled hydrolysis, which significantly minimizes these impurities and allows for easier purification via crystallization.
Q: What are the critical reaction conditions for the hydrolysis step?
A: The hydrolysis of the N-dihalophosphinyl derivative is optimally conducted in a mixture of organic hydrocarbon solvent and water at a pH range of 4 to 6, preferably around 5.5. The temperature is maintained between 40°C and 110°C, typically at 70°C, to ensure complete conversion without degrading the sensitive carbamate linkage.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process relies on classical unit operations such as extraction, filtration, and crystallization rather than complex chromatography or high-vacuum distillation. The use of common solvents like toluene and dichloromethane, along with stable intermediates, makes it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Febarbamate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team has extensively analyzed the pathways described in CN1034806C and possesses the expertise to implement this high-efficiency route for the production of febarbamate and related pyrimidinetrione derivatives. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facility is equipped with state-of-the-art rigorous QC labs and analytical instruments capable of verifying stringent purity specifications, ensuring that every batch meets the highest international standards. We understand that consistency is key in API manufacturing, and our process controls are designed to minimize variability and maximize yield.
We invite potential partners to engage with us to explore how this optimized synthesis can benefit your supply chain. By leveraging our technical capabilities, you can secure a stable source of high-quality intermediates that support your drug development timelines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our commitment to quality and transparency. Let us collaborate to bring efficient, cost-effective, and high-purity chemical solutions to your pipeline, driving value through scientific excellence and operational reliability.
