Advanced Industrial Synthesis of Cefozopran Hydrochloride via GCLE Intermediates
Advanced Industrial Synthesis of Cefozopran Hydrochloride via GCLE Intermediates
The pharmaceutical industry continuously seeks robust manufacturing pathways for fourth-generation cephalosporins, and patent CN102040616A presents a significant breakthrough in the preparation of Cefozopran Hydrochloride. This technical disclosure outlines a novel synthetic strategy that diverges from traditional, cost-prohibitive methods by utilizing 7-phenylacetamide-3-chloromethyl-3-cephem-4-carboxylic acid p-methoxybenzyl ester (GCLE) as the primary starting material. By shifting the synthetic focus to this accessible precursor, the methodology effectively circumvents the reliance on expensive silylating agents like Iodotrimethylsilane, which have historically burdened the cost structure of similar beta-lactam antibiotics. The process integrates a sequence of nucleophilic substitutions, enzymatic deprotections, and condensation reactions that are not only chemically elegant but also engineered for industrial feasibility. For global procurement teams and R&D directors, this represents a pivotal opportunity to optimize the supply chain for critical anti-infective agents, ensuring both economic efficiency and consistent high-quality output in the production of this potent microbiotic.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefozopran Hydrochloride has been constrained by two primary technological bottlenecks that hindered efficient commercial scale-up. The first conventional approach utilizes 7-amino-cephalosporanic acid (7-ACA) as the foundational raw material, a pathway documented in earlier literature such as CN200510064421.1. While chemically viable, this route necessitates the use of Iodotrimethylsilane, a reagent that is not only prohibitively expensive but also introduces significant handling complexities and safety concerns in large-volume reactors. The second established method relies on 7-beta-amino-3-(3-oxo butyryl acyloxy methyl)-3-cephem-4-carboxylic acid (7-AACA), which suffers from severe supply chain vulnerabilities due to the difficulty in sourcing high-purity raw materials. Furthermore, the 7-AACA route is plagued by low output capacity and intricate post-processing requirements, making it unsuitable for the rigorous demands of modern pharmaceutical manufacturing where throughput and cost-efficiency are paramount. These legacy methods create substantial barriers to entry for generic manufacturers and increase the overall cost of goods sold for the final API.
The Novel Approach
In stark contrast to these legacy limitations, the innovative process detailed in CN102040616A leverages GCLE to establish a streamlined and economically superior synthetic trajectory. This novel approach capitalizes on the ready availability and lower cost of GCLE, effectively decoupling the production process from the volatility of silane reagent markets. The methodology introduces a sophisticated iodide-mediated transformation followed by a specific enzymatic hydrolysis step, which collectively enhance the overall yield and purity profile of the intermediate compounds. By replacing harsh chemical deprotection steps with biocatalytic processes, the new route significantly reduces the generation of hazardous waste and simplifies the purification workflow. This strategic shift not only lowers the direct material costs but also mitigates the environmental impact associated with traditional cephalosporin synthesis. For supply chain stakeholders, this translates into a more resilient production model that is less susceptible to raw material shortages and regulatory pressures regarding solvent and reagent usage.
Mechanistic Insights into GCLE-Based Cephalosporin Functionalization
The core of this synthetic advancement lies in the precise manipulation of the cephem nucleus through a controlled nucleophilic substitution mechanism. In the initial stage, the chloromethyl group at the C-3 position of the GCLE molecule undergoes a halogen exchange reaction with an alkali metal iodide, such as sodium iodide or potassium iodide, within a ketone solvent system. This in situ generation of the reactive iodide species facilitates a subsequent nucleophilic attack by imidazo[1,2-b]pyridazine, leading to the formation of the quaternary ammonium salt intermediate, designated as Formula (II). This transformation is critical as it installs the essential cationic side chain required for the antibacterial activity of the final product. The reaction conditions are meticulously optimized to proceed at temperatures ranging from -20°C to 55°C, ensuring high conversion rates while minimizing thermal degradation of the sensitive beta-lactam ring. The use of ketone solvents like acetone or butanone provides an ideal medium for solubilizing the reactants and stabilizing the transition states involved in this complex heterocyclic substitution.
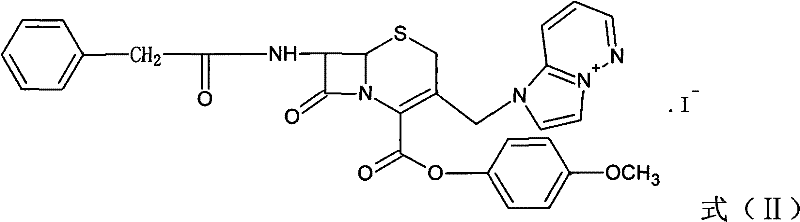
Following the construction of the quaternary ammonium scaffold, the process advances to a highly selective deprotection and hydrolysis sequence to reveal the free amino group at the C-7 position. The p-methoxybenzyl ester protecting group is removed via a phenol-mediated reaction under mild thermal conditions, typically between 40°C and 80°C. This step is immediately followed by an enzymatic hydrolysis using a specific acylase, such as IPA-750, which cleaves the phenylacetamide side chain with exceptional regioselectivity. This biocatalytic step is performed in an aqueous environment at a controlled pH of 7.2 to 8.2, preserving the integrity of the newly formed quaternary nitrogen center while efficiently generating the 7-aminocephalosporanic acid derivative (Formula III). The precision of this enzymatic cleavage is vital for preventing racemization or ring-opening side reactions that could compromise the stereochemical purity of the API. The resulting 7-ACP intermediate serves as the pivotal building block for the final coupling reaction.
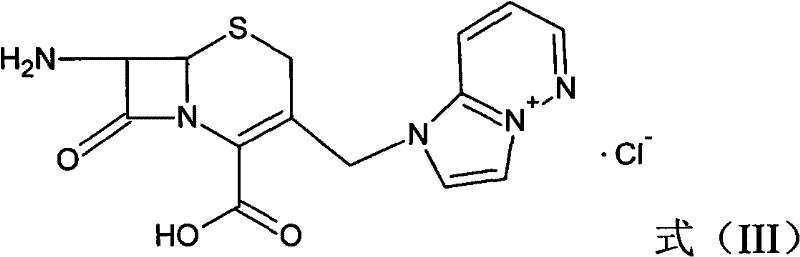
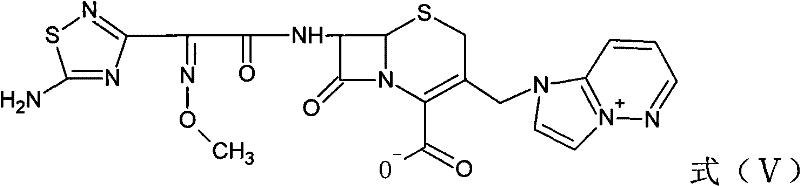
The final assembly of the Cefozopran molecule involves the condensation of the 7-ACP intermediate with a pre-activated side chain acid derivative, specifically the active ester represented by Formula (IV). This coupling reaction is executed in a mixed solvent system comprising methylene dichloride and methyl alcohol, utilizing an organic base such as triethylamine or pyridine to scavenge the generated acid. The reaction proceeds under controlled low-temperature conditions, typically between 0°C and 35°C, to manage the exothermic nature of the amide bond formation and prevent epimerization at the chiral centers. Upon completion, the resulting protected cephalosporin (Formula V) undergoes a salt-forming reaction with hydrochloric acid to yield the final Cefozopran Hydrochloride product. This final crystallization step is crucial for establishing the physical properties of the drug substance, including particle size distribution and polymorphic form, which are essential for downstream formulation and bioavailability.
How to Synthesize Cefozopran Hydrochloride Efficiently
The implementation of this synthesis route requires strict adherence to the reaction parameters defined in the patent to ensure optimal yield and purity. The process begins with the careful preparation of the iodide intermediate, followed by the enzymatic deprotection which demands precise pH control to maintain enzyme activity. The final condensation step necessitates anhydrous conditions and stoichiometric balance to minimize impurities. Detailed standardized operating procedures for each unit operation, including solvent recovery and crystallization protocols, are essential for transferring this technology from the laboratory to the pilot plant. For a comprehensive breakdown of the specific reaction times, temperatures, and workup procedures, please refer to the technical guide below.
- React GCLE with alkali metal iodide in ketone solvent followed by imidazo[1,2-b]pyridazine to form the quaternary ammonium intermediate.
- Perform phenol-mediated deprotection and subsequent enzymatic hydrolysis using acylase to obtain the 7-ACP core structure.
- Condense the 7-ACP intermediate with the specific active ester side chain in a mixed solvent system, followed by hydrochloric acid salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this GCLE-based synthesis route offers transformative advantages for procurement managers and supply chain directors seeking to optimize their antibiotic portfolios. The most immediate benefit is the substantial reduction in raw material costs achieved by eliminating the dependency on Iodotrimethylsilane, a high-value reagent that significantly inflates the bill of materials in conventional processes. By substituting this with inexpensive alkali metal iodides and leveraging the economies of scale associated with GCLE production, manufacturers can achieve a much more competitive cost structure. Furthermore, the use of enzymatic hydrolysis reduces the consumption of aggressive chemical reagents, thereby lowering waste disposal costs and simplifying environmental compliance. This economic efficiency allows for greater pricing flexibility in the market while maintaining healthy margins, a critical factor in the highly competitive generic pharmaceutical sector.
- Cost Reduction in Manufacturing: The elimination of expensive silylating agents and the use of cost-effective solvents like acetone and methylene dichloride directly lower the variable costs of production. The process avoids the complex purification steps associated with the 7-AACA route, reducing labor and utility consumption per kilogram of API produced. Additionally, the high selectivity of the enzymatic step minimizes the loss of valuable intermediates, improving the overall mass balance and yield of the process. These cumulative efficiencies result in a significantly reduced cost of goods sold, enabling manufacturers to offer more attractive pricing to downstream partners without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing GCLE is inherently more stable than procuring specialized precursors like 7-AACA, as GCLE is a widely produced intermediate in the cephalosporin industry. This availability ensures a continuous supply of starting materials, reducing the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent ratios, further enhances process reliability and reduces the likelihood of batch failures. For supply chain heads, this means improved forecast accuracy and the ability to maintain consistent inventory levels to meet market demand fluctuations.
- Scalability and Environmental Compliance: The synthetic pathway is designed with industrial scalability in mind, utilizing unit operations that are easily transferable to multi-purpose reactors. The mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufacturing site. Moreover, the reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations, facilitating smoother regulatory approvals and audits. This sustainability profile not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible producer of essential medicines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of the GCLE route. Understanding these details is crucial for technical teams evaluating the potential for technology transfer and for procurement professionals assessing the long-term viability of the supply source.
Q: Why is the GCLE route preferred over the traditional 7-ACA route for Cefozopran?
A: The traditional 7-ACA route relies on expensive Iodotrimethylsilane for silylation, significantly increasing raw material costs. The GCLE route utilizes readily available alkali metal iodides and avoids costly silane reagents, offering a more economically viable pathway for large-scale manufacturing.
Q: How does the enzymatic step improve product quality?
A: The use of specific acylase enzymes (such as IPA-750) allows for highly selective hydrolysis under mild pH and temperature conditions. This specificity minimizes the formation of unwanted by-products and degradation of the sensitive beta-lactam ring, resulting in higher purity intermediates compared to harsh chemical hydrolysis methods.
Q: Is this synthesis method suitable for commercial scale-up?
A: Yes, the process is designed for industrial suitability. It employs common solvents like acetone and methylene dichloride, operates at moderate temperatures (-20°C to 55°C), and uses stable starting materials. The elimination of complex post-processing steps associated with 7-AACA routes further enhances its scalability for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefozopran Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN102040616A to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity Cefozopran Hydrochloride and its key intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to process optimization allows us to consistently provide products that adhere to the highest international quality standards, safeguarding the efficacy and safety of the final medicinal products.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage these technological advancements for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can drive value for your organization. Let us help you secure a reliable, cost-effective, and high-quality supply of this essential fourth-generation cephalosporin.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
