Advancing Pharmaceutical Intermediate Synthesis via Visible Light Photoredox Catalysis
Introduction to Patent CN111333526B Technology
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for synthesizing complex organic molecules. Patent CN111333526B introduces a groundbreaking preparation method for N-aryl glycine ester derivatives, utilizing visible light-induced photoredox catalysis. This technology represents a significant departure from conventional thermochemical processes, offering a green, mild, and highly selective route to these valuable intermediates. By leveraging a dual catalytic system involving an iridium photocatalyst and an iron additive, the method achieves high yields under ambient conditions, effectively addressing the long-standing challenges of energy consumption and environmental pollution associated with traditional synthesis. For R&D directors and procurement managers alike, this innovation signals a shift towards more cost-effective and environmentally compliant manufacturing strategies for critical pharmaceutical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-aryl glycine ester derivatives has relied heavily on thermochemical reactions that demand harsh operating conditions. Traditional protocols often necessitate elevated temperatures and the use of ethyl haloacetate as an alkylating agent. This approach not only consumes substantial energy due to heating requirements but also generates stoichiometric amounts of halide waste, posing significant environmental disposal challenges. Furthermore, complex molecules containing multiple active functional groups frequently require tedious protection and deprotection steps to ensure chemoselectivity, thereby increasing the number of synthetic steps and reducing overall atom economy. These inefficiencies result in higher production costs and longer lead times, making conventional methods less attractive for modern, large-scale industrial applications where sustainability and efficiency are paramount.
The Novel Approach
In stark contrast, the method disclosed in patent CN111333526B employs a visible light-induced strategy that operates efficiently at room temperature. By utilizing ethyl diazoacetate in conjunction with a specialized photocatalyst system, this novel approach eliminates the need for external heating and avoids the generation of halide byproducts. The reaction proceeds under mild conditions with excellent functional group tolerance, allowing for the direct coupling of various aryl amines without the need for protective groups. This streamlined one-step process significantly simplifies the workflow, reduces energy consumption, and aligns perfectly with the principles of green chemistry. The ability to synthesize these derivatives under such benign conditions offers a compelling alternative for manufacturers seeking to optimize their production lines for both economic and ecological performance.
Mechanistic Insights into Fe/Ir Dual Catalytic System
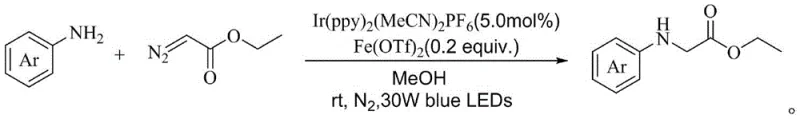
The core of this innovative synthesis lies in the synergistic interaction between the iridium photocatalyst, specifically [Ir(ppy)2(MeCN)2]PF6, and the iron additive, ferrous trifluoromethanesulfonate. Upon irradiation with blue LEDs, the iridium complex absorbs photons to reach an excited state, initiating a single-electron transfer process that activates the ethyl diazoacetate. This activation generates a reactive carbene or radical intermediate capable of inserting into the N-H bond of the aryl amine. The presence of the iron salt plays a crucial role as a Lewis acid or co-catalyst, potentially stabilizing transition states or facilitating the decomposition of the diazo compound in a controlled manner. This dual catalytic cycle ensures high reactivity and selectivity, minimizing side reactions such as dimerization of the diazo compound or over-alkylation, which are common pitfalls in non-catalyzed thermal reactions.
From an impurity control perspective, the mild nature of the photoredox mechanism inherently suppresses the formation of thermal degradation products. Since the reaction does not require high temperatures, heat-sensitive functional groups on the aromatic ring remain intact, preserving the integrity of the final product. The high selectivity of the catalyst system means that fewer byproducts are generated, simplifying the downstream purification process. For quality control teams, this translates to a cleaner crude reaction mixture, which facilitates easier isolation of the target N-aryl glycine ester with high purity, often exceeding 99% as demonstrated in the patent examples. This level of purity is critical for pharmaceutical intermediates where strict impurity profiles must be maintained to meet regulatory standards.
How to Synthesize N-Aryl Glycine Ester Efficiently
The practical implementation of this synthesis is straightforward and designed for ease of operation in a standard laboratory or pilot plant setting. The protocol involves dissolving the aryl primary amine, ethyl diazoacetate, the iron reaction aid, and the iridium photocatalyst in an organic solvent such as anhydrous methanol. The molar ratio of reactants is carefully optimized, typically employing a slight excess of the diazo compound to drive the reaction to completion. Once the mixture is homogenized, it is subjected to a freeze-pump-thaw cycle under nitrogen protection to remove dissolved oxygen, which can quench the excited state of the photocatalyst. The reaction vessel is then placed under a 30W blue LED lamp and stirred at room temperature for a period ranging from 12 to 40 hours, depending on the specific substrate reactivity.
- Dissolve aryl primary amine, ethyl diazoacetate, ferrous trifluoromethanesulfonate, and [Ir(ppy)2(MeCN)2]PF6 photocatalyst in anhydrous methanol.
- Seal the reaction vessel, perform freeze-pump-thaw cycles under nitrogen protection to remove oxygen, and place under 30W blue LED irradiation.
- Stir at room temperature for 12-40 hours, monitor by TLC, then concentrate and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photoredox technology offers tangible strategic benefits beyond mere technical novelty. The elimination of high-temperature heating requirements directly translates to significant reductions in utility costs and energy consumption during the manufacturing process. Furthermore, the avoidance of ethyl haloacetate removes the need for handling corrosive halide reagents and managing hazardous halide waste streams, thereby lowering compliance costs and environmental liabilities. The simplified one-step nature of the reaction reduces the overall processing time and labor intensity, allowing for faster throughput and improved asset utilization in production facilities. These factors collectively contribute to a more robust and cost-efficient supply chain for N-aryl glycine ester derivatives.
- Cost Reduction in Manufacturing: The transition from thermal to photochemical synthesis fundamentally alters the cost structure of production. By removing the need for energy-intensive heating and complex protection-deprotection sequences, the overall operational expenditure is drastically simplified. The use of commercially available and relatively stable catalysts, combined with the high atom economy of the diazo insertion reaction, ensures that raw material costs are optimized. Additionally, the reduced formation of byproducts minimizes the loss of valuable starting materials, further enhancing the economic viability of the process for large-scale commercial production.
- Enhanced Supply Chain Reliability: The mild reaction conditions inherent to this method significantly improve operational safety and reliability. Operating at room temperature and ambient pressure reduces the risk of thermal runaway incidents, making the process safer for operators and equipment. The broad substrate scope allows for the use of diverse and readily available aryl amines, reducing dependency on specialized or hard-to-source precursors. This flexibility ensures a more resilient supply chain capable of adapting to fluctuations in raw material availability without compromising on production schedules or product quality.
- Scalability and Environmental Compliance: Scalability is a critical consideration for any new synthetic method, and this photoredox approach is well-suited for scale-up. The simplicity of the setup, requiring only LED irradiation and stirring, facilitates the transition from benchtop to pilot and commercial scales. Moreover, the green chemistry credentials of the process, characterized by reduced waste generation and lower energy usage, align with increasingly stringent global environmental regulations. This compliance not only mitigates regulatory risks but also enhances the corporate sustainability profile, appealing to eco-conscious partners and stakeholders in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of N-aryl glycine ester derivatives using the patented visible light method. These insights are derived directly from the experimental data and technical specifications outlined in patent CN111333526B, providing clarity on reaction scope, conditions, and potential applications for industry professionals evaluating this technology for their supply chains.
Q: What are the advantages of this photoredox method over traditional thermal synthesis?
A: Unlike traditional methods requiring high temperatures and ethyl haloacetate which generate halide waste, this visible-light protocol operates at room temperature with high atom economy and reduced environmental impact.
Q: What is the substrate scope for the aryl amine component?
A: The method tolerates a wide range of substituents including esters, halogens, alkoxy groups, hydroxyls, and alkyls on the benzene ring, as well as heterocycles like naphthalene, pyridine, and quinoline.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the mild reaction conditions (room temperature, ambient pressure) and simple workup procedures significantly enhance scalability and operational safety compared to high-energy thermal processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Aryl Glycine Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN111333526B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of N-aryl glycine ester meets the exacting standards required for drug substance manufacturing.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green methodology can optimize your budget. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a reliable, high-quality supply of these critical intermediates for your global operations.
