Advanced Synthesis of 2-Hydroxyl-1-{4-(2-hydroxyethyl) Phenyl}-2-methyl-1-acetone for Industrial UV Curing Applications
The global demand for high-performance ultraviolet (UV) curing systems continues to drive innovation in the synthesis of specialized photoinitiators. Patent CN101811951A introduces a robust and economically viable preparation method for 2-hydroxyl-1-{4-(2-hydroxyethyl) phenyl}-2-methyl-1-acetone, a critical component in food-contact-safe UV applications. This technical disclosure outlines a streamlined three-step synthetic pathway that addresses significant bottlenecks found in traditional manufacturing protocols. By leveraging Lewis acid-catalyzed Friedel-Crafts acylation followed by a novel bromination strategy and phase-transfer hydrolysis, the process achieves superior yields while minimizing environmental impact. For R&D directors and procurement specialists, understanding the mechanistic nuances of this patent is essential for evaluating supply chain resilience and cost-efficiency in the production of next-generation coating additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of alpha-hydroxy ketone photoinitiators has been plagued by inefficient solvent systems and harsh reaction conditions that compromise both equipment longevity and operational throughput. Traditional protocols typically rely on glacial acetic acid as the primary solvent for the bromination step, a practice that necessitates extended reaction times often exceeding 10 hours to reach completion. This prolonged exposure to corrosive acidic environments accelerates the degradation of reactor linings and increases maintenance downtime, creating hidden costs that erode profit margins. Furthermore, the subsequent hydrolysis step in conventional methods requires substantial quantities of ethanol as a co-solvent to facilitate the reaction between the organic brominated intermediate and aqueous base. This reliance on ethanol not only inflates raw material costs but also complicates the downstream purification process, requiring energy-intensive distillation steps to recover solvents and isolate the final crystalline product.
The Novel Approach
The methodology described in CN101811951A represents a paradigm shift by replacing corrosive glacial acetic acid with chlorinated solvents such as dichloromethane or dichloroethane, coupled with catalytic amounts of N,N-dimethylformamide (DMF) or iodine. This strategic substitution drastically reduces the bromination reaction window to merely 1 to 2 hours, thereby enhancing reactor turnover rates and overall plant capacity. Additionally, the introduction of phase-transfer catalysts in the hydrolysis stage enables the direct interaction of the brominated intermediate with aqueous sodium hydroxide, effectively eliminating the need for bulk ethanol solvents. This modification simplifies the workup procedure, reduces waste generation, and lowers the thermal load required for solvent recovery, presenting a compelling value proposition for manufacturers seeking to optimize their production economics without sacrificing product quality.
Mechanistic Insights into the Three-Step Synthetic Route
The core of this synthesis lies in the precise control of electrophilic aromatic substitution and subsequent functional group transformations. The first stage involves the Friedel-Crafts acylation of ethylene glycol phenyl ether acetate with isobutyryl chloride, mediated by a Lewis acid catalyst such as aluminum chloride or ferric chloride. Maintaining the reaction temperature between -5°C and 5°C is critical to suppress polyacylation and ensure regioselectivity on the aromatic ring. This step generates the key ketone intermediate, which serves as the substrate for the subsequent alpha-functionalization. The choice of Lewis acid and the strict temperature control are pivotal in minimizing the formation of tar-like byproducts that can complicate purification later in the sequence.
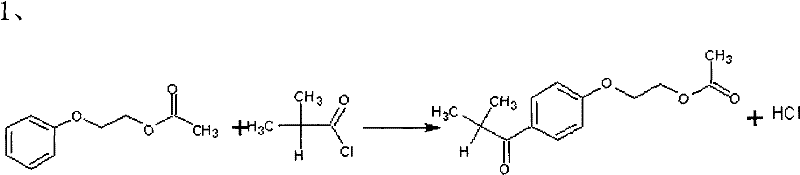
Following acylation, the process moves to the alpha-bromination of the ketone intermediate. Unlike traditional radical bromination which can be uncontrollable, this patent utilizes a catalytic amount of DMF or iodine to generate an reactive brominating species in situ. This catalytic cycle facilitates the rapid and selective substitution of the alpha-hydrogen with bromine under mild conditions (20-25°C). The use of dichloromethane or dichloroethane as the solvent ensures excellent solubility of both the organic substrate and molecular bromine, promoting homogeneous reaction kinetics. This step is crucial as it installs the leaving group necessary for the final hydrolysis, and the high selectivity achieved here directly correlates to the high purity of the final photoinitiator.
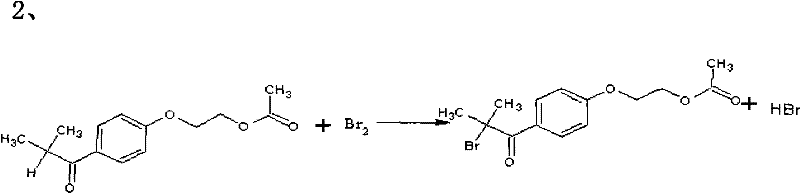
The final transformation involves the hydrolysis of the alpha-bromo ketone to the corresponding alpha-hydroxy ketone. In this novel approach, a phase-transfer catalyst, such as tetrabutylammonium bromide, is employed to shuttle hydroxide ions from the aqueous phase into the organic phase where the substrate resides. This interfacial catalysis allows the nucleophilic substitution to proceed rapidly at room temperature without the need for miscible organic co-solvents like ethanol. The mechanism avoids the harsh conditions that might lead to elimination side reactions, ensuring that the hydroxyl group is installed cleanly. The resulting crude product is then purified through extraction and recrystallization, yielding the target molecule with exceptional clarity and low odor, suitable for sensitive applications like food packaging coatings.
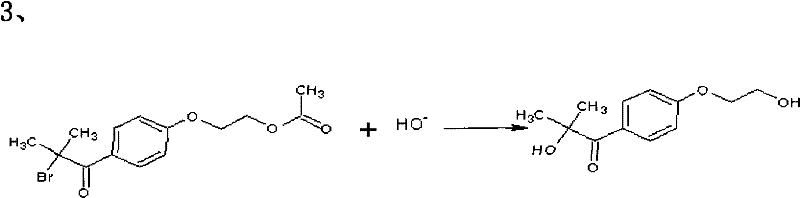
How to Synthesize 2-Hydroxyl-1-{4-(2-hydroxyethyl) Phenyl}-2-methyl-1-acetone Efficiently
Implementing this synthesis requires careful attention to stoichiometry and thermal management, particularly during the exothermic acylation and bromination steps. The standardized protocol begins with the dissolution of the Lewis acid in the chlorinated solvent, followed by the controlled addition of the ester substrate and acyl chloride. Once the intermediate is isolated, it undergoes bromination with precise monitoring of temperature to prevent dibromination. Finally, the hydrolysis is conducted in a biphasic system where the phase-transfer catalyst concentration is optimized to maximize mass transfer rates. Detailed standard operating procedures regarding reagent grades, addition rates, and quenching protocols are essential for reproducibility.
- Perform Friedel-Crafts acylation of ethylene glycol phenyl ether acetate with isobutyryl chloride using Lewis acid catalyst at -5 to 5°C.
- Conduct alpha-bromination of the intermediate ketone using bromine with DMF or Iodine catalysis in dichloromethane or dichloroethane.
- Execute hydrolysis of the brominated intermediate using aqueous sodium hydroxide and a phase-transfer catalyst at room temperature.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented process offers tangible benefits that extend beyond simple yield improvements. The elimination of glacial acetic acid and bulk ethanol from the process flow significantly reduces the inventory of hazardous and flammable solvents that must be stored and managed on-site. This reduction in solvent complexity translates to lower compliance costs regarding environmental health and safety regulations, as well as decreased expenditure on solvent recovery infrastructure. Furthermore, the shortened reaction times mean that existing reactor assets can be utilized more intensively, effectively increasing production capacity without the need for capital investment in new hardware.
- Cost Reduction in Manufacturing: The economic model of this synthesis is strengthened by the removal of expensive and voluminous ethanol solvents from the hydrolysis step. By utilizing water as the primary medium for hydrolysis aided by catalytic amounts of phase-transfer agents, the process drastically cuts down on raw material procurement costs. Additionally, the switch from glacial acetic acid to chlorinated solvents in the bromination step reduces equipment corrosion, leading to extended asset life and reduced maintenance frequency. These factors combine to lower the overall cost of goods sold (COGS), providing a competitive pricing advantage in the global photoinitiator market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as dichloromethane, sodium hydroxide, and isobutyryl chloride ensures a stable supply chain that is less susceptible to the volatility of specialty solvent markets. The robustness of the reaction conditions, particularly the ability to run the hydrolysis at ambient temperatures, reduces the risk of batch failures due to thermal runaway or cooling system limitations. This operational stability ensures consistent delivery schedules, allowing downstream formulators of UV coatings and adhesives to maintain lean inventory levels with confidence in the continuity of supply.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is facilitated by the homogeneous nature of the initial steps and the efficient biphasic workup in the final step. The reduction in solvent waste volume, specifically the avoidance of ethanol-contaminated aqueous streams, simplifies wastewater treatment protocols. This aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions and effluent discharge. Manufacturers adopting this technology can market their products as sustainably produced, appealing to end-users in the food and packaging industries who prioritize green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. Understanding these details is vital for process engineers evaluating the feasibility of technology transfer. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance to industrial application.
Q: How does the new bromination method improve upon conventional glacial acetic acid processes?
A: The novel method utilizes dichloromethane or dichloroethane as solvents with DMF or Iodine catalysis, reducing reaction time from over 10 hours to just 1-2 hours and eliminating equipment corrosion associated with glacial acetic acid.
Q: What are the benefits of using phase-transfer catalysis in the hydrolysis step?
A: Phase-transfer catalysis allows the brominated intermediate to react directly with alkali in water, eliminating the need for large volumes of ethanol solvent, which simplifies post-treatment and significantly lowers production costs.
Q: What purity levels can be achieved with this synthesis route?
A: Through optimized purification and crystallization steps involving ethyl acetate extraction and dichloroethane recrystallization, the process consistently achieves product content exceeding 98%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxyl-1-{4-(2-hydroxyethyl) Phenyl}-2-methyl-1-acetone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced UV curing materials depends on the seamless integration of innovative chemistry with robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is managed with precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of photoinitiator meets the exacting standards required for food-contact and medical applications. Our commitment to quality assurance ensures that the low odor and low yellowing characteristics of this molecule are preserved throughout the supply chain.
We invite industry partners to collaborate with us on optimizing their UV formulation portfolios using this high-performance initiator. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to reach out for specific COA data and route feasibility assessments to determine how this patented synthesis can enhance your product line. Let us help you engineer a more efficient and cost-effective supply chain for your next-generation coating solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
