Advanced Enzymatic Synthesis of Chiral Nilaparib Intermediates for Commercial Scale-Up
Advanced Enzymatic Synthesis of Chiral Nilaparib Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust, cost-effective, and scalable routes for the production of complex oncology therapeutics. A pivotal development in this domain is detailed in patent CN111072550A, which discloses a novel enzymatic synthesis method for a critical intermediate of Nilaparib (Niraparib), a potent PARP-1/2 inhibitor approved for treating ovarian cancer. This patent outlines a sophisticated seven-step sequence that transforms simple starting materials into a highly functionalized chiral piperidine precursor. By leveraging biocatalysis for the establishment of the chiral center, this methodology circumvents many of the economic and technical bottlenecks associated with traditional asymmetric synthesis. For R&D directors and supply chain managers alike, understanding this pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with consistent quality.
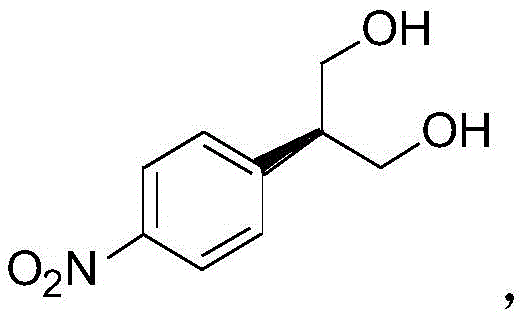
The significance of this technology lies in its ability to integrate green chemistry principles with industrial pragmatism. Unlike earlier synthetic strategies that relied heavily on stoichiometric chiral reagents or difficult-to-separate racemic mixtures, the approach described in CN111072550A utilizes an esterifying enzyme to achieve kinetic resolution. This not only enhances the optical purity of the intermediate but also streamlines the workflow by operating under mild reaction conditions. As we delve deeper into the technical specifics, it becomes evident that this route represents a substantial leap forward in cost reduction in API manufacturing, offering a viable alternative for companies aiming to optimize their supply chains for next-generation cancer therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of Nilaparib and its precursors has been fraught with challenges related to stereocontrol and operational complexity. Early methodologies, such as those reported by Jones et al. and later improved by Wallace et al., often involved multi-step sequences with modest overall yields. A significant drawback in these conventional approaches was the reliance on harsh chemical conditions to induce chirality or the necessity of resolving racemic mixtures late in the synthesis, which inherently limits the maximum theoretical yield to 50% unless dynamic kinetic resolution is employed. Furthermore, the use of expensive transition metal catalysts for asymmetric hydrogenation or coupling reactions often introduces issues related to heavy metal residue removal, necessitating additional purification steps that drive up both time and cost. These factors collectively contribute to a fragile supply chain where minor deviations in reaction parameters can lead to batch failures, posing a risk to commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data introduces a biocatalytic strategy that fundamentally alters the economic landscape of the synthesis. By employing an esterifying enzyme in conjunction with vinyl acetate, the process achieves high specificity in generating the desired (R)-chiral monoester intermediate directly from a prochiral diol. This enzymatic step operates at a moderate temperature of 45°C, eliminating the need for cryogenic conditions often required for organometallic asymmetric catalysis. The subsequent chemical transformations, including the conversion of the alcohol to a bromide and the subsequent Wittig-type olefination, are designed to be robust and high-yielding. This integration of biocatalysis with classical organic synthesis creates a hybrid pathway that maximizes efficiency while minimizing waste, thereby addressing the core pain points of reducing lead time for high-purity pharmaceutical intermediates and ensuring a more sustainable manufacturing footprint.
Mechanistic Insights into Enzymatic Kinetic Resolution and Chain Elongation
The cornerstone of this synthetic route is the enzymatic kinetic resolution step, which dictates the stereochemical outcome of the entire sequence. In this transformation, the prochiral diol intermediate, generated from the reduction of dimethyl 2-(4-nitrophenyl)malonate, serves as the substrate for a lipase or esterifying enzyme. The enzyme selectively acylates one of the enantiotopic hydroxyl groups using vinyl acetate as the acyl donor. This selectivity arises from the precise fit of the substrate within the enzyme's active site, where steric and electronic interactions favor the formation of the (R)-monoester over the (S)-isomer. The use of vinyl acetate is particularly advantageous as the byproduct, acetaldehyde, tautomerizes to volatile species, driving the equilibrium forward and preventing reverse hydrolysis. This mechanism ensures that the chiral information is installed early in the synthesis with high fidelity, setting the stage for the subsequent construction of the piperidine ring system without the need for further resolution steps.
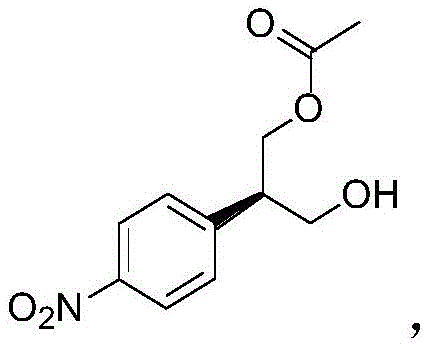
Following the establishment of chirality, the synthesis proceeds through a series of carbon-carbon bond-forming reactions designed to extend the side chain and introduce the necessary functionality for cyclization. The conversion of the chiral alcohol to the corresponding bromide using triphenylphosphine and bromine generates a reactive electrophile. This intermediate then undergoes a nucleophilic substitution or elimination-addition sequence with ethyl glyoxylate in the presence of sodium tert-butoxide. This step effectively installs the unsaturated ester moiety, which is subsequently reduced via catalytic hydrogenation over palladium on carbon. The mechanistic elegance here lies in the chemoselectivity of the reduction; the nitro group on the aromatic ring is simultaneously reduced to an amine, while the ester functionality remains intact. This dual reduction capability simplifies the synthetic tree, reducing the step count and improving the overall atom economy of the process, which is critical for maintaining stringent purity specifications in the final drug substance.
How to Synthesize Nilaparib Intermediate Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly during the enzymatic and hydrogenation stages. The process begins with the reduction of the starting diester to a diol, followed by the critical enzymatic resolution in acetonitrile. Subsequent steps involve the activation of the alcohol, chain extension via olefination, and global reduction to reveal the aniline and alcohol functionalities required for the final cyclization. The detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined below to ensure reproducibility and safety during scale-up operations.
- Reduce dimethyl 2-(4-nitrophenyl)malonate with sodium borohydride in methanol to form the diol intermediate.
- Perform enzymatic kinetic resolution using esterifying enzyme and vinyl acetate in acetonitrile to obtain the chiral monoester.
- Convert the chiral alcohol to a bromide using triphenylphosphine and bromine, followed by olefination with ethyl glyoxylate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic synthesis route offers compelling strategic advantages that extend beyond mere technical feasibility. The shift towards biocatalysis in the early stages of the synthesis significantly de-risks the supply chain by reducing dependence on scarce or volatile chiral reagents. Enzymes are renewable catalysts that can often be recycled or sourced from stable suppliers, providing a buffer against raw material shortages. Furthermore, the mild reaction conditions associated with enzymatic steps translate to lower energy consumption and reduced wear on reactor vessels, contributing to a lower total cost of ownership for the manufacturing process. This alignment with green chemistry principles not only enhances the corporate sustainability profile but also mitigates regulatory risks associated with solvent usage and waste generation.
- Cost Reduction in Manufacturing: The elimination of expensive chiral ligands and transition metal catalysts in the initial stereoselective step results in a direct reduction in material costs. Additionally, the high specificity of the enzyme minimizes the formation of diastereomeric impurities, which drastically simplifies the purification workflow. By avoiding complex chromatographic separations typically required for racemic mixtures, the process achieves substantial cost savings in both solvent usage and processing time, making it an economically superior choice for large-scale production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as dimethyl malonate derivatives and vinyl acetate ensures a stable and diversified supply base. Unlike proprietary chiral pools that may be subject to single-source bottlenecks, the reagents used in this pathway are widely available from multiple global vendors. This redundancy enhances supply security, ensuring that production schedules remain uninterrupted even in the face of market fluctuations. The robustness of the chemical steps following the enzymatic resolution further guarantees consistent output quality, fostering trust between the manufacturer and the end-user.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations that are easily transferable from laboratory to pilot and commercial scales. The use of common solvents like methanol, acetonitrile, and dichloromethane allows for established recovery and recycling protocols, minimizing environmental impact. Moreover, the avoidance of hazardous reagents like sodium amide (used in some older indazole syntheses) improves the safety profile of the plant, reducing insurance premiums and regulatory compliance burdens associated with handling dangerous substances.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Nilaparib intermediates. These insights are derived directly from the patent data and practical experience in process chemistry, aimed at clarifying the feasibility and benefits of this specific route for potential partners and stakeholders.
Q: What is the primary advantage of the enzymatic step in this Nilaparib intermediate synthesis?
A: The enzymatic kinetic resolution step offers superior stereoselectivity compared to traditional chemical resolution methods, significantly reducing the formation of unwanted enantiomers and simplifying downstream purification processes.
Q: How does this route address the high cost issues associated with conventional Nilaparib synthesis?
A: By utilizing readily available starting materials like dimethyl 2-(4-nitrophenyl)malonate and avoiding expensive chiral auxiliaries or harsh transition metal catalysts in the initial steps, the overall material cost is substantially lowered while maintaining high yield efficiency.
Q: Is this synthetic route scalable for industrial production?
A: Yes, the process utilizes standard unit operations such as reflux, filtration, and hydrogenation under moderate pressures (e.g., 5kg), making it highly amenable to scale-up from pilot plant to multi-ton commercial manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nilaparib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of life-saving oncology drugs. Our team of expert process chemists has extensively evaluated the enzymatic pathway described in CN111072550A and possesses the technical capability to implement this technology at an industrial level. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from clinical trials to market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring every step of the synthesis to meet stringent purity specifications, guaranteeing that the intermediates we supply are of the highest quality and fully compliant with global regulatory standards.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By leveraging our expertise in enzymatic catalysis and process intensification, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects. Let us be your partner in bringing innovative cancer therapies to patients faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
