Electrochemical Production of 7-Keto Lithocholic Acid: A Green Manufacturing Breakthrough
The pharmaceutical industry is constantly seeking greener, more efficient pathways for synthesizing critical bile acid intermediates, and patent CN1912192B presents a transformative approach to producing 7-keto lithocholic acid. This compound serves as a pivotal precursor in the synthesis of Ursodeoxycholic Acid (UDCA), a vital therapeutic agent for treating primary biliary cirrhosis and dissolving cholesterol gallstones. Traditional synthetic routes have long been plagued by the use of hazardous heavy metal oxidants and multi-step protection strategies that inflate costs and environmental burdens. The disclosed invention leverages indirect electrochemical oxidation, utilizing inexpensive potassium bromide as a redox mediator to achieve selective oxidation of the C-7 hydroxyl group on Chenodeoxycholic Acid (CDCA). 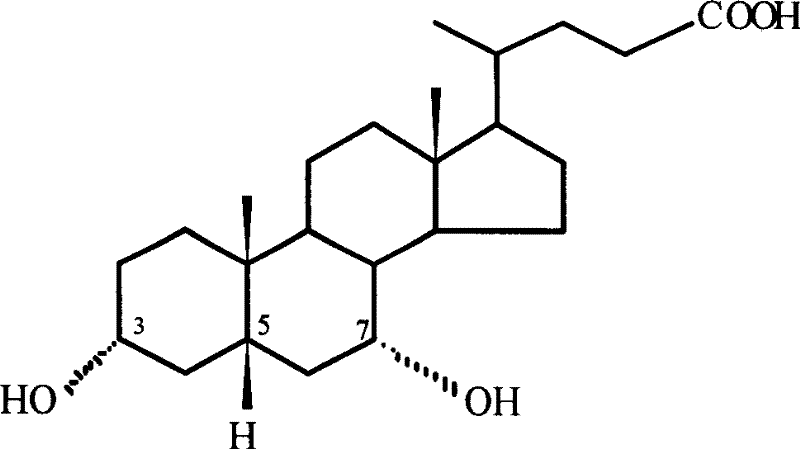 This electrochemical strategy not only simplifies the operational workflow but also aligns perfectly with modern green chemistry principles, offering a sustainable alternative for the reliable pharmaceutical intermediate supplier market.
This electrochemical strategy not only simplifies the operational workflow but also aligns perfectly with modern green chemistry principles, offering a sustainable alternative for the reliable pharmaceutical intermediate supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 7-keto lithocholic acid and its subsequent conversion to UDCA has relied heavily on classical chemical oxidation methods that pose significant safety and environmental challenges. Conventional protocols often involve the use of stoichiometric amounts of toxic oxidizing agents such as chromium trioxide or ceric ammonium nitrate, which generate substantial quantities of hazardous heavy metal waste requiring complex and costly disposal procedures. Furthermore, older synthetic pathways frequently necessitate intricate protection and deprotection sequences, such as esterifying the 3-hydroxyl group prior to oxidation to prevent over-oxidation or side reactions, thereby increasing the number of unit operations and reducing overall atom economy. These multi-step processes inherently suffer from cumulative yield losses, extended production cycles, and higher energy consumption due to the need for rigorous temperature control and extensive solvent exchanges. For procurement managers, these factors translate into volatile pricing and supply chain fragility, as the availability of specialized reagents and the capacity for hazardous waste treatment can become bottlenecks in large-scale manufacturing.
The Novel Approach
In stark contrast, the novel electrochemical method detailed in the patent data introduces a streamlined, single-step oxidation process that bypasses the need for toxic stoichiometric oxidants entirely. By employing constant-current electrolysis in the presence of a water-soluble bromide salt, the system generates the active oxidizing species in situ at the anode surface, effectively turning electricity into a clean reagent. This approach allows for the direct conversion of Chenodeoxycholic Acid to 7-keto lithocholic acid under mild conditions, typically using common organic solvents like acetonitrile or ethanol mixed with aqueous electrolytes. The elimination of protection groups significantly shortens the synthetic route, reducing both the time-to-market and the physical footprint required for production. From a commercial perspective, this simplification drastically lowers the barrier to entry for cost reduction in pharmaceutical intermediates manufacturing, as it minimizes raw material complexity and reduces the dependency on fluctuating markets for exotic chemical oxidants.
Mechanistic Insights into Indirect Electrochemical Oxidation
The core of this innovative technology lies in the mechanism of indirect electro-oxidation mediated by bromide ions, which offers superior selectivity and control compared to direct electrolysis. In this system, bromide ions (Br-) present in the electrolyte are oxidized at the anode surface to generate elemental bromine (Br2) or hypobromous acid species, depending on the specific pH and solvent composition of the reaction medium. 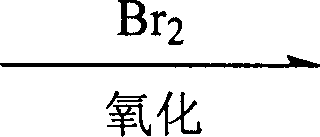 These electro-generated bromine species then act as powerful yet selective chemical oxidants that diffuse into the bulk solution to react with the secondary alcohol at the C-7 position of the steroid nucleus. This indirect mechanism is crucial because it separates the electron transfer event at the electrode from the chemical transformation of the organic substrate, preventing the formation of radical species that could lead to polymerization or degradation of the sensitive steroid backbone. The current density, optimized between 47.6 A/m² and 190.4 A/m², ensures a steady supply of the oxidant without causing excessive over-potential that might degrade the solvent or the product.
These electro-generated bromine species then act as powerful yet selective chemical oxidants that diffuse into the bulk solution to react with the secondary alcohol at the C-7 position of the steroid nucleus. This indirect mechanism is crucial because it separates the electron transfer event at the electrode from the chemical transformation of the organic substrate, preventing the formation of radical species that could lead to polymerization or degradation of the sensitive steroid backbone. The current density, optimized between 47.6 A/m² and 190.4 A/m², ensures a steady supply of the oxidant without causing excessive over-potential that might degrade the solvent or the product.
Furthermore, this mechanistic pathway provides inherent advantages in terms of impurity profile control, which is a primary concern for R&D directors overseeing quality assurance. Because the oxidizing power is governed by the applied current and the concentration of the bromide mediator, the reaction can be precisely tuned to stop at the ketone stage without progressing to over-oxidized byproducts like carboxylic acids or cleaved ring structures. The use of a diaphragm cell configuration, as described in several embodiments, further enhances purity by preventing the reduced species at the cathode from interfering with the anodic oxidation process or reducing the newly formed ketone back to an alcohol. This level of control results in a crude product with high HPLC content, often exceeding 86%, which significantly reduces the burden on downstream purification units and ensures a consistent quality profile suitable for stringent regulatory requirements in API manufacturing.
How to Synthesize 7-Keto Lithocholic Acid Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing the simplicity of mixing readily available reagents and applying controlled electrical current. The process begins by dissolving the starting material, Chenodeoxycholic Acid, in a suitable organic solvent such as acetonitrile, methanol, or ethanol, which serves to solubilize the hydrophobic steroid skeleton. This organic phase is then combined with an aqueous solution containing potassium bromide, creating a biphasic or homogeneous electrolytic system depending on the solvent ratios used. The mixture is subjected to constant-current electrolysis using durable electrode materials like lead dioxide-coated titanium mesh or graphite plates, which offer excellent corrosion resistance in the oxidative environment. Detailed standardized synthesis steps see the guide below.
- Dissolve Chenodeoxycholic acid (CDCA) in an organic solvent such as acetonitrile or ethanol, and mix with an aqueous solution of Potassium Bromide (KBr) to form the electrolyte.
- Perform constant-current electrolysis in a diaphragm or non-diaphragm cell using a PbO2/Ti or graphite anode, maintaining a current density between 47.6 A/m² and 190.4 A/m².
- Monitor reaction progress via TLC, and upon completion, pour the anolyte into deionized water to precipitate the crude 7-keto lithocholic acid, followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the shift to this electrochemical methodology offers profound strategic benefits that extend beyond mere technical novelty. By replacing expensive and hazardous chemical oxidants with electricity and common salt, the process fundamentally alters the cost structure of manufacturing this high-value intermediate. The reliance on commodity chemicals like potassium bromide and standard industrial solvents ensures that raw material sourcing is stable and immune to the supply shocks often associated with specialized fine chemical reagents. Moreover, the simplified workflow reduces the number of processing stages, which directly correlates to lower labor costs, reduced equipment occupancy time, and decreased utility consumption per kilogram of product. These factors combine to create a manufacturing model that is not only economically superior but also resilient against regulatory tightening regarding environmental emissions and worker safety.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the complete elimination of stoichiometric heavy metal oxidants, which are not only costly to purchase but also expensive to dispose of in compliance with environmental regulations. By generating the oxidant in situ via electrolysis, the process removes the need for purchasing chromium or cerium reagents, leading to substantial savings in raw material expenditures. Additionally, the reduction in synthetic steps means fewer solvent swaps and purification operations, which drastically cuts down on solvent recovery costs and energy usage for heating and cooling. This lean manufacturing approach allows for a more competitive pricing structure, making it an attractive option for cost reduction in pharmaceutical intermediates manufacturing without compromising on product quality.
- Enhanced Supply Chain Reliability: Supply chain heads will appreciate the robustness of the raw material portfolio required for this process, as potassium bromide and common alcohols are produced globally in massive quantities with stable pricing trends. Unlike complex chiral catalysts or sensitive organometallic reagents that may have single-source suppliers and long lead times, the inputs for this electrochemical method are widely available from multiple vendors, mitigating the risk of production stoppages due to material shortages. The scalability of electrolytic cells is also well-understood in the chemical industry, allowing for straightforward capacity expansion by adding more cell units rather than redesigning the entire chemical process. This modularity supports reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API production schedules are met consistently.
- Scalability and Environmental Compliance: As global regulations on industrial effluents become increasingly stringent, the environmental profile of a manufacturing process becomes a critical license to operate. This electrochemical method generates minimal hazardous waste, primarily consisting of spent electrolyte which can often be regenerated or treated easily compared to heavy metal sludge. The absence of toxic byproducts simplifies the wastewater treatment process and reduces the carbon footprint associated with waste disposal logistics. Furthermore, the process operates under mild conditions without the need for extreme temperatures or pressures, enhancing the inherent safety of the plant and lowering insurance and compliance costs. This alignment with green chemistry principles positions the manufacturer favorably for audits and partnerships with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route, derived directly from the experimental data and claims within the patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of adopting this technology for their own production lines or for procurement specialists negotiating supply contracts. The answers reflect the specific conditions and outcomes reported in the patent examples, providing a realistic expectation of performance and operational requirements.
Q: What are the advantages of the electrochemical method over traditional chemical oxidation for 7-keto lithocholic acid?
A: The electrochemical method described in patent CN1912192B eliminates the need for toxic stoichiometric oxidants like chromium reagents or complex protection-deprotection sequences. It utilizes electricity and inexpensive potassium bromide, resulting in milder operating conditions, reduced environmental impact, and a simplified downstream purification process.
Q: What is the typical purity and yield achievable with this electrolytic process?
A: According to the patent embodiments, the crude product typically achieves an HPLC content ranging from approximately 86% to 90%. After standard purification steps such as column chromatography and recrystallization, the product meets high purity specifications with a melting point consistent with literature values (201-203°C).
Q: Can this process be scaled for industrial production of ursodeoxycholic acid intermediates?
A: Yes, the process is highly scalable. It operates under constant current conditions using standard electrode materials like PbO2/Ti mesh or graphite plates. The use of common solvents like acetonitrile and simple salts like KBr ensures that raw material supply chains are robust and suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Keto Lithocholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain, and we are uniquely positioned to leverage advanced technologies like the one described in CN1912192B. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of 7-keto lithocholic acid meets the exacting standards required for API synthesis. Our commitment to continuous improvement drives us to adopt greener, more efficient manufacturing methods that benefit our partners through better pricing and more reliable delivery schedules.
We invite you to collaborate with us to explore how this electrochemical technology can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for sustainable and cost-effective pharmaceutical production.
