Scalable Reformatsky Synthesis of 4-Halo-2-Methyl-2-Butenoic Acid Esters for Industrial Applications
The chemical landscape for producing high-value carotenoid intermediates is undergoing a significant transformation driven by the need for safer and more economically viable synthetic routes. Patent CN116041181A introduces a groundbreaking method for preparing 4-halo-2-methyl-2-butenoic acid alkyl esters, which serve as critical building blocks in the synthesis of complex natural pigments and pharmaceutical agents. This innovation leverages a modified Reformatsky reaction mechanism that utilizes haloacetaldehyde and 2-zinc bromide propionate as primary raw materials, effectively bypassing the severe safety hazards and environmental burdens associated with traditional halogenation techniques. By operating under mild conditions with a high safety coefficient, this process addresses the longstanding challenges of toxicity and pollution that have historically plagued the manufacturing of these fine chemical intermediates. The strategic implementation of this technology allows for the generation of high-purity products with minimal organic byproducts, primarily yielding recoverable inorganic zinc salts as solid waste. For industry leaders seeking a reliable fine chemical intermediates supplier, this patent represents a pivotal shift towards sustainable and scalable production methodologies that align with modern regulatory and economic demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
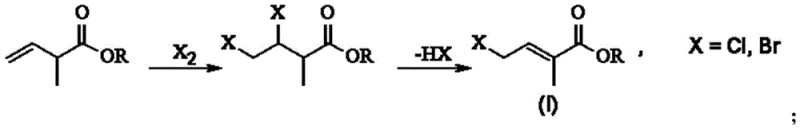 Historical synthetic routes for 4-chloro or bromo-2-methyl-2-butenoic acid esters have been fraught with significant operational and environmental drawbacks that hinder efficient commercial scale-up of complex polymer additives and pharmaceutical precursors. Traditional methods, such as the addition-elimination pathway depicted above, rely heavily on the direct use of elemental halogens as reaction reagents, which introduces extreme safety risks due to their high toxicity and corrosive nature. Furthermore, the atom economy of these conventional processes is inherently poor, as the theoretical utilization rate of the halogen is often limited to merely fifty percent, resulting in substantial waste generation. Alternative approaches like the Wittig reaction, while effective in forming the carbon-carbon double bond, necessitate the use of organophosphorus reagents that generate large quantities of oxidized phosphorus byproducts which are notoriously difficult to recover and regenerate. The disposal of these phosphorus-containing wastes imposes a heavy financial burden on manufacturers and creates significant environmental compliance challenges, making these legacy methods increasingly untenable in a modern green chemistry context. Consequently, the industry has long sought a cost reduction in fine chemical intermediates manufacturing that eliminates these hazardous reagents without compromising yield or product quality.
Historical synthetic routes for 4-chloro or bromo-2-methyl-2-butenoic acid esters have been fraught with significant operational and environmental drawbacks that hinder efficient commercial scale-up of complex polymer additives and pharmaceutical precursors. Traditional methods, such as the addition-elimination pathway depicted above, rely heavily on the direct use of elemental halogens as reaction reagents, which introduces extreme safety risks due to their high toxicity and corrosive nature. Furthermore, the atom economy of these conventional processes is inherently poor, as the theoretical utilization rate of the halogen is often limited to merely fifty percent, resulting in substantial waste generation. Alternative approaches like the Wittig reaction, while effective in forming the carbon-carbon double bond, necessitate the use of organophosphorus reagents that generate large quantities of oxidized phosphorus byproducts which are notoriously difficult to recover and regenerate. The disposal of these phosphorus-containing wastes imposes a heavy financial burden on manufacturers and creates significant environmental compliance challenges, making these legacy methods increasingly untenable in a modern green chemistry context. Consequently, the industry has long sought a cost reduction in fine chemical intermediates manufacturing that eliminates these hazardous reagents without compromising yield or product quality.
The Novel Approach
The innovative methodology disclosed in the patent data offers a robust solution by replacing hazardous halogenation and phosphorus-based olefination with a streamlined Reformatsky condensation followed by dehydration. This novel approach utilizes readily available and inexpensive raw materials, specifically metal zinc powder and 2-bromopropionate, to generate the reactive zinc enolate species in situ, thereby avoiding the need for isolating unstable intermediates. The reaction conditions are remarkably mild, typically proceeding at temperatures between 0°C and 100°C, which significantly reduces energy consumption and enhances operational safety compared to the high-temperature or cryogenic conditions required by older methods. By eliminating the use of elemental halogens and organophosphorus compounds, this process drastically simplifies the downstream purification workflow and reduces the generation of toxic organic waste streams. The primary byproducts are inorganic zinc halides, which exist as solid waste that can be conveniently recovered and managed, while the reaction solvents can be recycled with minimal pollution. This transition to a zinc-mediated pathway not only improves the overall economic feasibility of the synthesis but also aligns perfectly with the growing global emphasis on environmentally friendly and sustainable chemical manufacturing practices.
Mechanistic Insights into Reformatsky-Catalyzed Cyclization
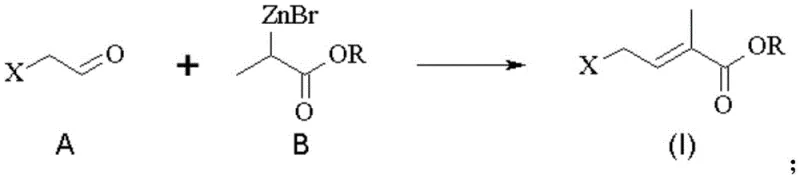 The core of this technological advancement lies in the precise mechanistic execution of the Reformatsky reaction between the anhydrous haloacetaldehyde and the zinc enolate derived from 2-bromopropionate. As illustrated in the reaction scheme, the process begins with the formation of the organozinc intermediate, which then nucleophilically attacks the carbonyl group of the haloacetaldehyde to form a beta-hydroxy ester intermediate. This step is critical as it establishes the carbon skeleton required for the final unsaturated ester, and the use of zinc ensures a high degree of chemoselectivity that minimizes the formation of unwanted side products. Following the initial addition, the reaction mixture undergoes an in-situ dehydration step, often facilitated by the reaction conditions or mild acid workup, to yield the conjugated 4-halo-2-methyl-2-butenoic acid alkyl ester. The ability to perform this transformation in a one-pot or telescoped manner significantly reduces the number of unit operations required, thereby lowering capital expenditure and increasing throughput efficiency for commercial production facilities. Understanding this mechanism is vital for R&D directors focused on purity and impurity profiles, as it highlights the controlled nature of the bond formation which leads to a cleaner crude product.
The core of this technological advancement lies in the precise mechanistic execution of the Reformatsky reaction between the anhydrous haloacetaldehyde and the zinc enolate derived from 2-bromopropionate. As illustrated in the reaction scheme, the process begins with the formation of the organozinc intermediate, which then nucleophilically attacks the carbonyl group of the haloacetaldehyde to form a beta-hydroxy ester intermediate. This step is critical as it establishes the carbon skeleton required for the final unsaturated ester, and the use of zinc ensures a high degree of chemoselectivity that minimizes the formation of unwanted side products. Following the initial addition, the reaction mixture undergoes an in-situ dehydration step, often facilitated by the reaction conditions or mild acid workup, to yield the conjugated 4-halo-2-methyl-2-butenoic acid alkyl ester. The ability to perform this transformation in a one-pot or telescoped manner significantly reduces the number of unit operations required, thereby lowering capital expenditure and increasing throughput efficiency for commercial production facilities. Understanding this mechanism is vital for R&D directors focused on purity and impurity profiles, as it highlights the controlled nature of the bond formation which leads to a cleaner crude product.
Impurity control is another paramount aspect of this synthesis, particularly given the sensitivity of carotenoid precursors to structural isomers and halogenated byproducts. The patent specifies that the use of anhydrous haloacetaldehyde solutions, prepared through rigorous drying with calcium chloride and molecular sieves, is essential to prevent hydrolysis and side reactions that could compromise the integrity of the zinc enolate. By maintaining strict control over the water content and reaction temperature, the process effectively suppresses the formation of isomeric impurities that are commonly observed in radical substitution or addition-elimination methods. The subsequent workup involving hydrolysis with sulfuric acid allows for the separation of the organic product from the inorganic zinc salts, which precipitate as a muddy solid that can be easily filtered or decanted. This physical separation mechanism ensures that the final distillate contains minimal metal contamination, reducing the need for extensive chromatographic purification. For manufacturers aiming to produce high-purity OLED material or pharmaceutical intermediates, this inherent ability to manage impurity profiles through reaction engineering rather than extensive downstream processing offers a distinct competitive advantage in terms of both cost and quality assurance.
How to Synthesize 4-Halo-2-Methyl-2-Butenoic Acid Alkyl Ester Efficiently
Implementing this synthesis route requires careful attention to the preparation of reagents and the control of reaction parameters to ensure optimal yield and safety. The process begins with the preparation of a dry haloacetaldehyde solution, where aqueous haloacetaldehyde is treated with anhydrous calcium chloride and extracted into a solvent such as 2-methyltetrahydrofuran to remove water completely. This step is crucial as the presence of water can deactivate the zinc reagent and lead to poor conversion rates. Following this, metal zinc powder is dispersed in the solvent and reacted with 2-bromopropionate at elevated temperatures to generate the active zinc species in situ. The detailed standardized synthesis steps see the guide below.
- Prepare anhydrous haloacetaldehyde solution by drying aqueous haloacetaldehyde with calcium chloride and molecular sieves in a solvent like 2-methyltetrahydrofuran.
- Generate the zinc enolate in situ by reacting metal zinc powder with 2-bromopropionate in the solvent at 70°C under nitrogen protection.
- Add the anhydrous haloacetaldehyde solution to the enolate mixture, react, then hydrolyze with sulfuric acid and distill to isolate the final ester product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, the adoption of this Reformatsky-based synthesis route offers substantial benefits that extend far beyond simple chemical transformation. The elimination of expensive and hazardous reagents such as elemental halogens and organophosphorus compounds directly translates into a more stable and predictable cost structure for raw material acquisition. By relying on commodity chemicals like zinc powder and bromopropionates, manufacturers can mitigate the risks associated with price volatility in the specialty chemical market, ensuring a more reliable fine chemical intermediates supplier relationship. Furthermore, the simplified waste profile, characterized mainly by recoverable inorganic salts, significantly reduces the operational costs associated with hazardous waste disposal and environmental compliance monitoring. This reduction in regulatory burden allows supply chain heads to focus on logistics and throughput rather than managing complex environmental permits, thereby enhancing the overall agility of the production network. The robustness of the reaction conditions also implies a lower risk of batch failures due to sensitive parameters, contributing to a more consistent supply of critical intermediates for downstream customers.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of high-cost reagents with inexpensive, commodity-grade raw materials that are readily available in the global market. By avoiding the use of organophosphorus reagents, the process eliminates the need for complex and costly waste treatment systems required to handle phosphorus-containing effluents, leading to significant operational savings. Additionally, the ability to recycle reaction solvents with minimal pollution further reduces the consumption of expensive organic solvents, contributing to a lower overall cost of goods sold. The high atom economy of the Reformatsky reaction compared to addition-elimination methods ensures that a greater proportion of the raw material mass is converted into the desired product, minimizing waste and maximizing resource efficiency. These factors combined create a compelling economic case for adopting this technology in large-scale manufacturing environments where margin optimization is critical.
- Enhanced Supply Chain Reliability: The reliance on stable and widely available starting materials such as metal zinc and haloacetaldehyde derivatives ensures a resilient supply chain that is less susceptible to disruptions caused by the scarcity of specialty reagents. Unlike methods that depend on specific catalysts or unstable intermediates, this process utilizes robust chemistry that can be sustained over long production runs without significant degradation in performance. The mild reaction conditions also reduce the wear and tear on production equipment, lowering maintenance costs and minimizing unplanned downtime that could impact delivery schedules. For supply chain managers, this translates into a more predictable lead time for high-purity fine chemical intermediates, allowing for better inventory planning and stronger service levels for end customers. The scalability of the process from laboratory to industrial scale further ensures that supply can be ramped up quickly to meet surging market demand without compromising quality.
- Scalability and Environmental Compliance: The environmental profile of this synthesis route is exceptionally favorable, making it highly suitable for production in regions with stringent environmental regulations. The primary waste streams are inorganic zinc salts which are non-toxic and easy to handle, contrasting sharply with the hazardous organic waste generated by conventional methods. This simplifies the permitting process for new production facilities and reduces the risk of regulatory fines or shutdowns due to compliance issues. The process is designed for commercial scale-up of complex fine chemical intermediates, with reaction parameters that are easily controlled in large reactors, ensuring consistent quality across different batch sizes. By adopting this green chemistry approach, companies can enhance their corporate sustainability credentials while simultaneously improving their operational efficiency and reducing their environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patent-protected synthesis method. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The information provided here serves as a foundational guide for further discussions on process optimization and customization.
Q: What are the primary advantages of the Reformatsky method over Wittig olefination for this intermediate?
A: The Reformatsky method avoids the use of expensive and difficult-to-remove organophosphorus reagents associated with Wittig reactions, significantly reducing waste treatment costs and environmental impact while simplifying the purification process.
Q: How does this process handle the toxicity concerns of haloacetaldehyde?
A: The patent specifies a rigorous drying and solvent extraction protocol using 2-methyltetrahydrofuran and calcium chloride to create a stable anhydrous solution, minimizing direct handling hazards and ensuring reaction stability.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (0-100°C), the reagents are cheap and readily available, and the byproducts are primarily inorganic zinc salts which are easily separated, making it highly viable for industrial mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Halo-2-Methyl-2-Butenoic Acid Alkyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like the one described in CN116041181A to maintain competitiveness in the global fine chemicals market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of Reformatsky chemistry, including the safe handling of zinc reagents and the efficient recovery of solvents. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify the identity and quality of every batch. By partnering with us, clients gain access to a supply chain that is not only robust and reliable but also deeply knowledgeable about the nuances of complex intermediate synthesis.
We invite you to engage with our technical procurement team to explore how this innovative synthesis route can be tailored to your specific production needs. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to this greener and more efficient method. Our team is ready to provide specific COA data and route feasibility assessments to support your decision-making process. Whether you are looking to optimize an existing supply chain or develop a new product line, NINGBO INNO PHARMCHEM is dedicated to providing the technical expertise and manufacturing capacity required to succeed.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
