Advanced Manufacturing of 2-Chloro-5-Picoline via Novel Adduct Intermediates
Introduction to Advanced Pyridine Functionalization
The global demand for high-purity heterocyclic building blocks continues to surge, driven by the relentless innovation in crop protection and pharmaceutical sectors. Patent CN1075476A introduces a transformative methodology for the synthesis of 2-chloro-5-picoline, a critical scaffold in modern organic synthesis. This document details a robust two-step protocol that overcomes the historical limitations of direct chlorination, specifically addressing the persistent challenge of isomeric impurity formation. By leveraging a unique activation strategy involving N-oxide precursors, this technology offers a pathway to superior product quality that aligns with the rigorous standards expected by a reliable agrochemical intermediate supplier. The process begins with the strategic functionalization of 3-methylpyridine-1-oxide, setting the stage for a highly selective transformation that defines the next generation of fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-chloro-5-methylpyridine has been plagued by selectivity issues that compromise both yield and economic efficiency. Traditional routes, such as those described in prior art like EP-A-439745, typically involve the direct reaction of 3-picoline-1-oxide with phosgene in the presence of triethylamine. While chemically feasible, these legacy processes suffer from a critical flaw: the concurrent formation of substantial quantities of the 2-chloro-3-picoline isomer. This structural analog is notoriously difficult to separate due to similar physical properties, necessitating energy-intensive purification steps such as repeated crystallization or complex chromatography. For large-scale operations, this lack of selectivity translates directly into increased operational expenditures and reduced throughput, creating a bottleneck for cost reduction in agrochemical intermediate manufacturing.
The Novel Approach
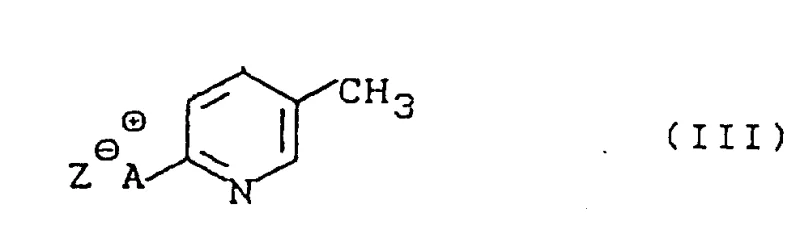
In stark contrast, the methodology disclosed in CN1075476A employs a sophisticated intermediate isolation strategy that fundamentally alters the reaction trajectory. Instead of a direct one-pot chlorination, the process first generates a stable quaternary ammonium adduct (Formula III) through the reaction of the N-oxide with an organic nitrogen base and an electrophile. This intermediate acts as a masked reactive species that, upon subsequent thermal treatment with a chlorinating agent, undergoes a highly regioselective transformation. This decoupling of the activation and chlorination events allows for precise control over the reaction environment, effectively suppressing the formation of the undesired 3-isomer. The result is a streamlined workflow that delivers high-purity 2-chloro-5-picoline with significantly improved isomer ratios, representing a valuable improvement to prior art that enhances both technical feasibility and commercial viability.
Mechanistic Insights into N-Oxide Activation and Chlorination
The core innovation of this synthesis lies in the formation and utilization of the Formula III adduct. The reaction initiates with 3-methylpyridine-1-oxide, where the electron-rich oxygen atom and the pyridine ring system are primed for nucleophilic and electrophilic interactions respectively.
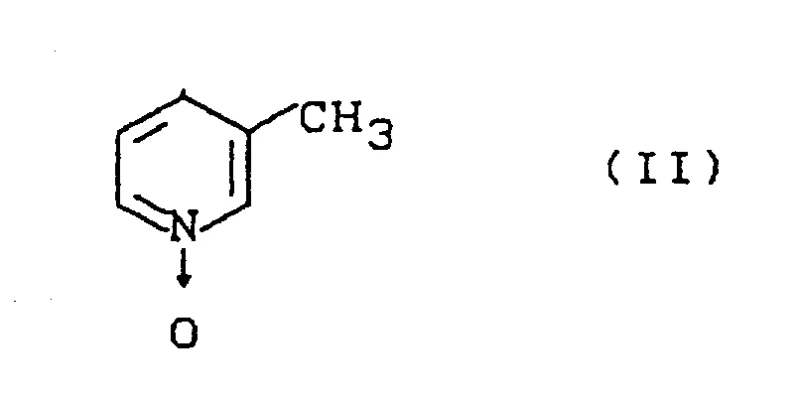
Upon introduction of an organic nitrogenous base, such as trimethylamine or triethylamine, alongside an electrophilic compound like phosgene or thionyl chloride, a complex addition reaction occurs. This results in the formation of a quaternary ammonium salt intermediate, depicted generally as Formula III. In this structure, the nitrogen base is covalently attached to the pyridine ring, creating a highly activated cationic center that stabilizes the molecule against premature side reactions. The anion Z, derived from the electrophile, balances the charge and can be tailored to influence solubility and reactivity. This intermediate is not merely a transient species but a isolable entity that serves as the precursor for the final chlorination event.
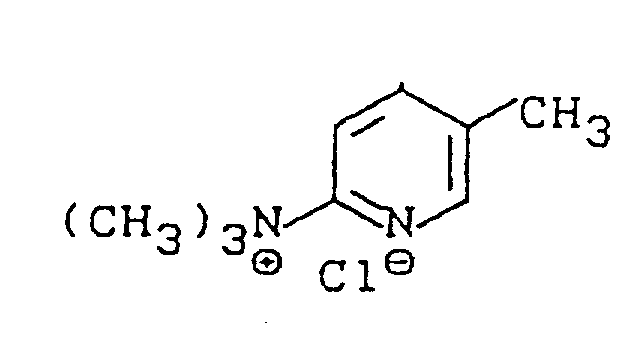
The second mechanistic phase involves the thermal decomposition of this adduct in the presence of a chlorinating source. Heating the Formula III compound to temperatures ranging from 50°C to 150°C triggers the elimination of the amine moiety and the simultaneous introduction of the chlorine atom at the 2-position. The specific geometry of the intermediate ensures that the chlorination occurs exclusively at the desired site, thereby maintaining high isomer purity. Furthermore, the mechanism allows for the recovery of unreacted intermediate, as the adduct remains stable enough to be reclaimed from the reaction mixture if conversion is incomplete. This recyclability is a crucial feature for industrial applications, ensuring that raw material costs are minimized and waste generation is kept to a theoretical minimum.
How to Synthesize 2-Chloro-5-Picoline Efficiently
Implementing this synthesis requires careful attention to temperature control and reagent stoichiometry to maximize the yield of the key intermediate before proceeding to chlorination. The process is designed to be flexible, accommodating various solvents and bases, but optimal results are achieved through a standardized protocol that balances reaction kinetics with safety considerations. The following overview outlines the critical operational parameters derived from the patent examples, providing a roadmap for laboratory validation and pilot plant scaling.
- React 3-methylpyridine-1-oxide with an organic nitrogen base (e.g., trimethylamine) and an electrophilic compound (e.g., phosgene) at low temperatures (-50°C to 0°C) to form the quaternary ammonium adduct intermediate.
- Isolate the intermediate adduct or proceed directly in the same vessel, heating the mixture to elevated temperatures (50°C to 150°C) in the presence of a chlorinating agent.
- Perform standard workup including aqueous washing, drying, and solvent removal to isolate high-purity 2-chloro-5-picoline with minimized isomeric impurities.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling strategic advantages that extend beyond simple chemical yield. The ability to produce high-purity 2-chloro-5-picoline with minimal isomeric contamination directly impacts the bottom line by reducing the need for expensive downstream purification infrastructure. This process optimization translates into tangible operational efficiencies that strengthen the overall supply chain resilience for critical fine chemical intermediates.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic simplification of the purification process. By significantly improving the isomer ratio during the reaction phase, manufacturers can avoid the capital-intensive and time-consuming separation steps required by conventional methods. The elimination of complex chromatographic purification or multiple recrystallizations reduces solvent consumption, energy usage, and labor hours. Additionally, the capability to recycle unreacted intermediates ensures that raw material utilization is maximized, further driving down the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: The robustness of this synthetic pathway contributes to a more stable and predictable supply chain. The reagents required, such as 3-methylpyridine-1-oxide, trimethylamine, and common chlorinating agents like phosgene or thionyl chloride, are widely available commodity chemicals with established global supply networks. This reliance on mature feedstock markets mitigates the risk of raw material shortages that often plague specialty syntheses dependent on exotic catalysts. Furthermore, the flexibility to operate across a broad temperature range and with various solvents allows production facilities to adapt quickly to local resource availability without compromising product quality.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this process is exceptionally well-suited for commercial expansion. The reaction conditions, while requiring thermal control, do not necessitate extreme pressures or cryogenic temperatures that would limit reactor size. The use of standard organic solvents like methylene chloride or acetonitrile facilitates straightforward solvent recovery and recycling systems, aligning with modern green chemistry principles. The reduction in waste streams, particularly the minimization of isomeric byproducts, simplifies effluent treatment and helps facilities maintain strict compliance with environmental regulations, ensuring uninterrupted production schedules.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical teams evaluating this route for potential integration into their manufacturing portfolios.
Q: How does this novel method improve isomer purity compared to conventional routes?
A: Conventional methods often yield significant amounts of the unwanted 2-chloro-3-picoline isomer. This patented process utilizes a specific quaternary ammonium adduct intermediate (Formula III) which directs the chlorination selectively to the 2-position, drastically reducing isomeric contamination and simplifying downstream purification.
Q: Can the intermediate adduct be recycled if the second step is incomplete?
A: Yes, a key advantage of this process is the recoverability of the unreacted Formula III intermediate. If the conversion in the second chlorination step is not complete, the intermediate can be reclaimed via water evaporation and reused, minimizing material loss and improving overall atom economy.
Q: What are the preferred chlorinating agents for industrial scale-up?
A: While various agents like thionyl chloride and sulfuryl chloride are effective, the patent highlights phosgene and hydrogen chloride as particularly preferred for the second step due to their reactivity profiles and the ability to drive the reaction to completion at moderate temperatures (30-110°C).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-Picoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and state-of-the-art infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of this novel synthesis are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-chloro-5-picoline meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to quality assurance means that clients can rely on us for consistent supply without the variability often associated with complex heterocyclic synthesis.
We invite forward-thinking organizations to collaborate with us to optimize their supply chains through advanced chemical manufacturing. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our mastery of this patented process can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
