Advanced Continuous Synthesis of High-Purity 1,3-Dialkyl-2-Imidazolidinone for Industrial Applications
The chemical industry is constantly seeking more efficient pathways to produce high-purity solvents that serve as critical media for pharmaceutical and agrochemical synthesis. Patent CN1176249A introduces a groundbreaking methodology for the preparation of 1,3-dialkyl-2-imidazolidinone, a compound renowned for its exceptional stability against acids and bases. This patent details a continuous reaction process where N,N'-dialkylethylenediamine and urea are simultaneously introduced into a heated aprotic polar solvent. Unlike traditional batch methods that struggle with byproduct accumulation, this novel approach ensures that the resulting solvent contains less than 0.1% by weight of the troublesome 1,3-dialkyl-2-imidazolinimine impurity. For R&D directors and procurement specialists, this represents a significant leap forward in obtaining reliable specialty solvent supplier materials that do not inhibit downstream polymerization or coupling reactions. The ability to produce such high-purity materials industrially opens new avenues for cost reduction in fine chemical manufacturing where solvent quality directly impacts final API purity.
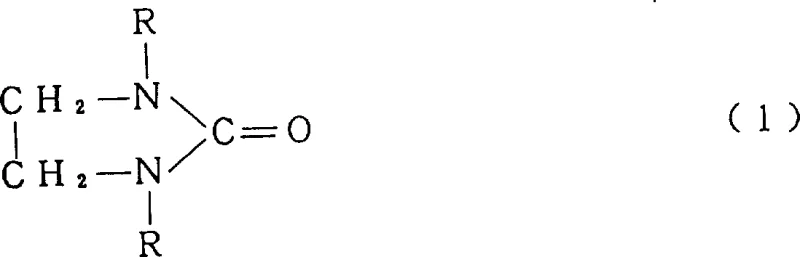
The structural integrity of the final product is paramount for its function as a polymerization solvent, particularly in the synthesis of aramids where impurities can act as chain terminators. The general formula depicted above illustrates the core 1,3-dialkyl-2-imidazolidinone structure where R represents an alkyl group, typically having 1 to 4 carbon atoms for optimal solvent properties. By strictly controlling the reaction environment through continuous feed mechanisms, manufacturers can avoid the formation of the double-bonded imine byproduct which possesses a boiling point dangerously close to the desired ketone. This proximity in physical properties traditionally necessitates complex distillation columns with very high theoretical plate counts, driving up both energy consumption and equipment costs. The innovation lies not just in the chemistry, but in the engineering control of reactant addition rates relative to the solvent volume, ensuring a consistent chemical environment that favors the desired cyclic urea formation over dehydration side reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3-dialkyl-2-imidazolidinone has been plagued by the persistent formation of 1,3-dialkyl-2-imidazolinimine, a byproduct that is chemically similar yet functionally detrimental. Prior art, such as USP 4,731,453, attempted to address yield issues by heating reactants to temperatures不低于 180°C, yet this often resulted in byproduct levels ranging from 0.5% to several percentage points. These impurities are notoriously difficult to remove via standard distillation because their volatility closely mirrors that of the target imidazolidinone, requiring elaborate fractionation setups that are energy-intensive and capital-heavy. Furthermore, when these contaminated solvents are utilized in sensitive applications like the preparation of aramids or electronic component cleaning, the residual imine can inhibit polymer growth or leave undesirable residues on precision surfaces. For supply chain heads, relying on processes that generate significant impurity loads means dealing with inconsistent batch quality and the potential for rejected shipments if purity specifications are not meticulously met, thereby increasing the overall cost of goods sold through waste and reprocessing.
The Novel Approach
The methodology disclosed in CN1176249A fundamentally alters the reaction kinetics by employing a continuous addition strategy into a pre-heated aprotic polar solvent maintained at elevated temperatures. Instead of mixing all reagents at once, which creates localized zones of high concentration conducive to byproduct formation, the N,N'-dialkylethylenediamine and urea are fed continuously into the reactor. This ensures that the reactants are immediately diluted into the hot solvent matrix, promoting rapid cyclization while minimizing the opportunity for the dehydration pathway that leads to the imine impurity. The process allows for the reaction to proceed at atmospheric pressure, eliminating the need for expensive high-pressure autoclaves often required in comparative batch examples. Additionally, the continuous discharge of the reaction product facilitates a steady-state operation that is inherently more stable and easier to control than batch processing. This shift from batch to continuous flow not only enhances the purity profile to exceed 99.9% but also streamlines the commercial scale-up of complex specialty chemicals by reducing the footprint and complexity of the required manufacturing infrastructure.
Mechanistic Insights into Continuous Cyclization Kinetics
The core chemical transformation involves the condensation of N,N'-dialkylethylenediamine with urea to form the cyclic urea structure, a reaction that is thermodynamically favorable but kinetically sensitive to reaction conditions. In the continuous process, the maintenance of a reaction temperature between 200°C and 260°C is critical; temperatures below 180°C risk leaving unreacted intermediates like 1,1'-dialkyl-1,1'-dimethylene diurea, while excessively high temperatures could promote thermal degradation. The choice of solvent plays a pivotal mechanistic role, with aprotic polar solvents like N-methyl-2-pyrrolidone or even the product itself acting as the reaction medium. These solvents stabilize the transition states without participating in proton transfer that might catalyze unwanted side reactions. The continuous flow ensures that the concentration of free amine and urea remains low at any given point in the reactor, effectively suppressing the bimolecular collisions that lead to the formation of the imine byproduct. This kinetic control is the key differentiator that allows the process to achieve yields exceeding 97% while maintaining impurity levels below the chromatographic detection limit of 0.1%.
From an impurity control perspective, the mechanism relies heavily on the residence time within the reactor, which is optimized to be not less than 5 hours. This extended duration ensures that the cyclization reaction goes to completion, allowing any transient intermediates to fully convert into the stable imidazolidinone ring. The absence of water in the system is another crucial mechanistic factor; the patent emphasizes using substantially anhydrous raw materials and solvents to prevent the hydrolysis of urea or the steam distillation of the volatile diamine reactant. By eliminating water, the equilibrium is driven firmly towards the formation of the cyclic product, and the risk of generating ammonia or other volatile byproducts is minimized. This rigorous control over the reaction environment results in a product stream that requires minimal downstream purification, often needing only a simple distillation through a column with as few as 5 theoretical plates to achieve ultra-high purity suitable for electronic or pharmaceutical grade applications.
How to Synthesize 1,3-Dialkyl-2-Imidazolidinone Efficiently
Implementing this synthesis route requires precise engineering controls to manage the continuous feed rates and thermal profiles effectively. The process begins by charging a reactor with an initial volume of aprotic polar solvent, which is then heated to the target operating temperature of approximately 220°C. Once thermal equilibrium is established, the mixture of N,N'-dialkylethylenediamine and urea is introduced via a pump system at a controlled rate that ensures the total volume of the reactor is displaced over a period of 5 to 10 hours. This specific residence time is vital for maximizing conversion and minimizing the carryover of intermediates. The detailed standardized synthesis steps, including specific molar ratios and equipment configurations for scaling this continuous process, are outlined in the technical guide below.
- Prepare an aprotic polar solvent system, such as N-methyl-2-pyrrolidone or the product itself, and maintain the reaction temperature at not lower than 180°C, preferably between 200°C and 260°C.
- Continuously add N,N'-dialkylethylenediamine and urea simultaneously into the heated solvent system, ensuring a molar ratio between 1.0: 1.0 and 1.0:1.2 to minimize side reactions.
- Maintain a residence time of not less than 5 hours within the reactor to ensure complete conversion and suppress the formation of 1,3-dialkyl-2-imidazolinimine byproducts before continuous discharge.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this continuous synthesis technology offers profound strategic benefits beyond mere chemical purity. The elimination of high-pressure requirements significantly reduces the capital expenditure associated with reactor fabrication and maintenance, as standard atmospheric vessels can be utilized instead of specialized autoclaves rated for 15 kg/cm²G or higher. This simplification of equipment directly translates to cost reduction in specialty solvent manufacturing, allowing for more competitive pricing structures without sacrificing margin. Furthermore, the continuous nature of the process ensures a consistent output quality that mitigates the risk of batch-to-batch variability, a common pain point in fine chemical sourcing that often leads to production delays for downstream customers. By securing a supply of high-purity imidazolidinones produced via this robust method, companies can stabilize their own manufacturing schedules and reduce the inventory buffers typically required to manage quality fluctuations.
- Cost Reduction in Manufacturing: The process utilizes readily available raw materials such as urea and dialkylethylenediamines, avoiding the need for expensive transition metal catalysts or exotic reagents that drive up input costs. The ability to operate at atmospheric pressure further lowers energy consumption and equipment depreciation costs compared to high-pressure batch alternatives. Additionally, the high selectivity of the reaction minimizes waste generation, reducing the expenses associated with waste treatment and disposal. The simplified purification train, requiring fewer distillation stages due to the low impurity load, also contributes to substantial operational savings by lowering utility usage and increasing throughput capacity.
- Enhanced Supply Chain Reliability: Continuous processing inherently offers greater stability and predictability than batch operations, which are susceptible to start-up and shut-down losses. This reliability ensures a steady flow of high-purity 1,3-dialkyl-2-imidazolidinone, reducing lead time for high-purity solvents and enabling just-in-time delivery models. The robustness of the method against minor fluctuations in feed composition means that supply interruptions due to off-spec material are significantly reduced. For global supply chains, this consistency is invaluable, as it reduces the need for extensive incoming quality control testing and allows for longer-term supply agreements with guaranteed specification adherence.
- Scalability and Environmental Compliance: The modular nature of continuous flow reactors allows for straightforward scale-up from pilot plants to full commercial production without the geometric complexities often encountered when enlarging batch vessels. This scalability supports the growing demand for green solvents in the pharmaceutical and electronics sectors. Environmentally, the process generates minimal byproduct waste, and the absence of heavy metal catalysts simplifies the regulatory compliance landscape regarding residue limits in final products. The high atom economy of the reaction ensures that the majority of raw materials are incorporated into the final product, aligning with modern sustainability goals and reducing the overall environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,3-dialkyl-2-imidazolidinone based on the patented continuous synthesis method. Understanding these details is crucial for R&D teams evaluating this solvent for sensitive reactions and for procurement officers assessing supplier capabilities. The answers provided are derived directly from the technical specifications and experimental data presented in the patent literature, ensuring accuracy and relevance for industrial decision-making.
Q: How does the continuous addition method reduce impurities compared to batch processing?
A: Conventional batch methods often result in 0.5% to several percent of 1,3-dialkyl-2-imidazolinimine byproduct due to localized concentration spikes. The continuous addition method described in patent CN1176249A maintains a steady state in the hot solvent, keeping the byproduct level below the detection limit of 0.1%.
Q: What are the critical temperature parameters for this cyclization reaction?
A: The reaction must be conducted at temperatures not lower than 180°C to achieve high reaction rates and avoid intermediate residues. The optimal range is identified as 200°C to 260°C, which balances reaction speed with the suppression of thermal degradation or excessive byproduct formation.
Q: Can this process be scaled for commercial solvent production without high-pressure equipment?
A: Yes, unlike some comparative examples requiring pressurized autoclaves up to 15 kg/cm²G, this invention can be effectively carried out under atmospheric pressure. This significantly reduces capital expenditure on pressure-rated vessels and enhances operational safety for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Dialkyl-2-Imidazolidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of your solvent dictates the success of your synthesis. Our technical team has extensively analyzed the continuous synthesis pathways described in CN1176249A and possesses the engineering expertise to implement these high-efficiency protocols at scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every shipment meets stringent purity specifications required for pharmaceutical and electronic applications. We are committed to delivering high-purity 1,3-dialkyl-2-imidazolidinone that empowers your research and production teams to achieve superior results without the interference of inhibitory byproducts.
We invite you to collaborate with us to optimize your solvent supply chain and leverage the cost benefits of this advanced manufacturing technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact us to request specific COA data and route feasibility assessments for your next project. By partnering with us, you gain access to a supply network that prioritizes consistency, purity, and technical support, ensuring that your critical chemical processes run smoothly and efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
