Revolutionizing Cytosine Precursor Production: A Low-Pressure, High-Safety Synthetic Route for Commercial Scale-Up
The global demand for antiviral therapeutics, particularly nucleoside analogs like lamivudine and emtricitabine, has placed immense pressure on the supply chains of their core heterocyclic building blocks. Cytosine serves as the indispensable scaffold for these life-saving medications, yet its traditional manufacturing pathways have long been plagued by severe safety hazards and operational complexities. Patent CN1594287A introduces a transformative methodology for the preparation of 3-hydroxyacrylonitrile metal salts, a critical precursor that bypasses the need for high-pressure carbon monoxide carbonylation. By shifting the synthetic paradigm to a mild condensation reaction between acetonitrile and formic acid esters under 1-5 bar of pressure, this technology offers a robust alternative for industrial production. This report analyzes the technical merits of this low-pressure route, evaluating its potential to redefine the standards for a reliable pharmaceutical intermediates supplier seeking to optimize both safety profiles and production economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of cytosine and its derivatives has relied heavily on routes involving 3-alkoxyacrylonitrile or 3,3-dialkoxypropionitrile intermediates. These precursors were typically generated through the carbonylation of acetonitrile using carbon monoxide gas under extreme conditions, often requiring pressures around 50 atmospheres and the use of sodium alkoxide catalysts. Such high-pressure operations necessitate specialized, expensive reactor vessels equipped with rigorous safety monitoring systems to handle toxic CO gas, creating a substantial barrier to entry and increasing capital expenditure. Furthermore, the subsequent conversion of these intermediates often results in complex mixtures of 3-alkoxyacrylonitrile and 3,3-dialkoxypropionitrile, which are difficult to separate and purify efficiently.
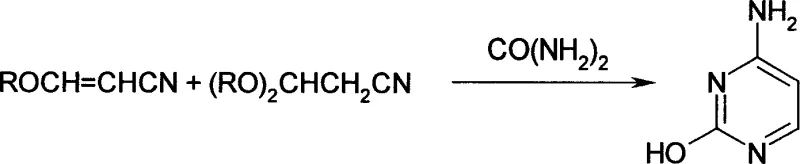
As illustrated in the reaction schemes above, the conventional pathway involves multiple equilibrium steps that generate inseparable mixtures, complicating the downstream cyclization with urea. The reliance on gaseous carbon monoxide not only introduces significant occupational health risks but also creates supply chain vulnerabilities related to the sourcing and storage of hazardous gases. Additionally, the energy consumption required to maintain high-pressure environments and the associated maintenance costs for pressure-rated equipment significantly inflate the operational expenses, making cost reduction in API manufacturing a persistent challenge for producers adhering to these legacy technologies.
The Novel Approach
In stark contrast, the methodology disclosed in CN1594287A utilizes a direct condensation strategy that eliminates the need for carbon monoxide entirely. By reacting acetonitrile with formic acid esters (such as methyl formate or ethyl formate) in the presence of metal alkoxides, the process generates the 3-hydroxyacrylonitrile metal salt directly under mild conditions of 20-60°C and 1-5 bar. This liquid-phase reaction avoids the handling of toxic gases and operates at near-atmospheric pressures, allowing for the use of standard glass-lined or stainless steel reactors rather than specialized high-pressure autoclaves. The simplicity of the reagent system—comprising commodity chemicals like acetonitrile and formates—ensures a stable and predictable supply chain, crucial for maintaining continuity in the production of high-purity pharmaceutical intermediates.
Mechanistic Insights into Metal Alkoxide-Catalyzed Condensation
The core chemical transformation driving this innovation is a base-catalyzed Claisen-type condensation between the alpha-protons of acetonitrile and the carbonyl group of the formic ester. In the presence of a strong base such as sodium methoxide or potassium tert-butoxide, acetonitrile is deprotonated to form a resonance-stabilized carbanion (nitrile anion). This nucleophilic species attacks the electrophilic carbonyl carbon of the formate ester, leading to the formation of a tetrahedral intermediate which subsequently collapses to eliminate an alkoxide leaving group. The result is the formation of the beta-keto nitrile structure, specifically stabilized as its metal salt form (3-hydroxyacrylonitrile metal salt). This mechanism is highly favorable thermodynamically under the specified conditions, driving the reaction to completion without the need for harsh forcing conditions.
From an impurity control perspective, this mechanism offers distinct advantages over the CO insertion route. The absence of carbon monoxide prevents the formation of carbonylation byproducts and reduces the risk of over-carbonylation side reactions that often plague high-pressure processes. Furthermore, because the reaction proceeds through a well-defined anionic pathway in a homogeneous or semi-homogeneous solution, the generation of polymeric tars or complex oligomeric impurities is minimized. The patent data indicates that the resulting salt solution is sufficiently pure to proceed directly to the next step—acidification and cyclization with urea—without intermediate isolation. This 'telescoped' capability suggests that the impurity profile is manageable and does not interfere with the subsequent ring-closing step to form the pyrimidine nucleus of cytosine, ensuring final product purity levels exceeding 99.0%.
How to Synthesize 3-Hydroxyacrylonitrile Metal Salt Efficiently
The operational protocol for this synthesis is designed for seamless integration into existing fine chemical infrastructure. The process begins by charging acetonitrile and a suitable organic solvent, such as toluene or xylene, into a reactor followed by the addition of the metal alkoxide catalyst. The formic ester is then introduced carefully to manage the exotherm, maintaining the temperature within the optimal 20-30°C window to prevent decomposition while ensuring rapid kinetics. Detailed standardized synthetic steps see the guide below.
- Charge acetonitrile, an organic solvent (such as toluene), and a metal alkoxide (e.g., sodium methoxide) into a reactor under inert atmosphere.
- Add formic acid ester (methyl or ethyl formate) gradually while maintaining temperature between 20-30°C and pressure at 2-3 bar.
- Stir the reaction mixture for 4-6 hours to complete condensation, yielding the 3-hydroxyacrylonitrile metal salt solution ready for direct cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from high-pressure carbonylation to low-pressure condensation represents a strategic opportunity to de-risk operations and optimize total cost of ownership. The elimination of carbon monoxide removes a major hazardous material from the facility, potentially lowering insurance premiums and reducing the regulatory burden associated with toxic gas storage. Moreover, the ability to operate at near-atmospheric pressure means that existing general-purpose reactors can be utilized, avoiding the need for costly capital investments in high-pressure autoclaves. This flexibility allows for faster deployment of production capacity and greater agility in responding to market demand fluctuations for antiviral intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven primarily by the simplification of the equipment train and the reduction in energy consumption. By operating at 1-5 bar instead of 50 bar, the process significantly lowers the energy input required for compression and heating. Additionally, the use of liquid formates instead of gaseous CO simplifies dosing and metering systems, reducing maintenance costs and downtime. The telescoping of the synthesis, where the intermediate salt is not isolated but used directly in the next step, further reduces solvent usage, filtration time, and drying costs, leading to substantial overall process savings without compromising yield.
- Enhanced Supply Chain Reliability: The raw materials for this process—acetonitrile, formic esters, and alkali metal alkoxides—are bulk commodity chemicals with mature, global supply networks. Unlike specialized catalysts or hazardous gases that may face logistical bottlenecks, these reagents are readily available from multiple vendors, ensuring a resilient supply chain. This diversification of sourcing options mitigates the risk of production stoppages due to raw material shortages. Furthermore, the mild reaction conditions reduce the wear and tear on reactor vessels, extending equipment lifespan and ensuring consistent batch-to-batch reliability for long-term contracts.
- Scalability and Environmental Compliance: Scaling this chemistry from pilot to commercial production is straightforward due to the absence of mass transfer limitations often associated with gas-liquid reactions (like CO absorption). The liquid-phase condensation scales linearly, facilitating the commercial scale-up of complex heterocycles with predictable heat transfer profiles. Environmentally, the process generates fewer hazardous wastes; the primary byproducts are alcohols which can often be recovered and recycled. The avoidance of heavy metal catalysts (often used in carbonylation) simplifies waste treatment and ensures the final product meets stringent heavy metal specifications required for pharmaceutical applications, aligning with modern green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this low-pressure synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on yield expectations, safety protocols, and downstream processing capabilities for potential partners.
Q: What are the safety advantages of this new synthesis route compared to traditional CO carbonylation?
A: The traditional method requires carbon monoxide at approximately 50 atmospheres of pressure, posing significant toxicity and explosion risks. The patented process operates at merely 1-5 bar using liquid esters, drastically reducing operational hazards and equipment costs.
Q: Can the intermediate salt solution be used directly without purification?
A: Yes, a key feature of this invention is that the crude 3-hydroxyacrylonitrile metal salt solution does not require isolation or purification. It can be directly reacted with alcoholic hydrochloric acid and urea to form cytosine, streamlining the workflow.
Q: What is the expected purity and yield for the final cytosine product?
A: Experimental data in the patent indicates consistent yields ranging from 53% to 56% based on acetonitrile, with final cytosine purity exceeding 99.0% as determined by HPLC area normalization methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxyacrylonitrile Metal Salt Supplier
The technological advancements detailed in CN1594287A underscore the importance of adopting safer, more efficient synthetic routes for critical pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial realities. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-hydroxyacrylonitrile metal salt or downstream cytosine meets the exacting standards required for antiviral API synthesis.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this low-pressure technology, we can help you secure a stable supply of high-quality intermediates while optimizing your manufacturing budget. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our engineering expertise can support your long-term supply chain goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
