Advanced Continuous Synthesis of High Purity 1,3-Dimethyl-2-Imidazolidinone for Industrial Applications
Introduction to Patent CN1067058C: A Breakthrough in Imidazolidinone Purity
The pharmaceutical and electronic chemical industries demand solvents and intermediates of exceptional purity to ensure the reliability of downstream reactions and final product performance. Patent CN1067058C, titled 'High Purity 1,3-dialkyl-2-imidazolidinone and preparation,' represents a significant technological leap in the manufacturing of 1,3-dialkyl-2-imidazolidinones, particularly 1,3-dimethyl-2-imidazolidinone (DMI). This intellectual property discloses a novel continuous addition process that reacts N,N'-dialkylethylenediamine with urea in an aprotic polar solvent. Unlike traditional batch methodologies which struggle with persistent impurity profiles, this invention achieves a manufacturing breakthrough by continuously feeding reactants into a heated solvent environment. The result is a high-yield production capability that efficiently generates very high purity 1,3-dialkyl-2-imidazolidinone containing less than 0.1% by weight of the problematic by-product, 1,3-dialkyl-2-imidazolidinimine. For R&D directors and procurement specialists seeking a reliable fine chemical intermediates supplier, understanding the mechanistic advantages of this continuous thermal cyclization is crucial for optimizing supply chain stability and product quality.
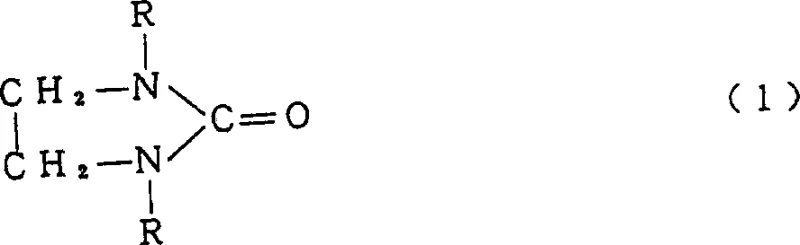
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3-dialkyl-2-imidazolidinone has been plagued by the formation of stubborn by-products that compromise the utility of the solvent in sensitive applications. Conventional methods typically involve reacting N,N'-dialkylethylenediamine with urea, often requiring a two-stage heating process where an intermediate, 1,1'-dimethyl-1,1'-dimethylene diurea, is formed at lower temperatures before cyclization at higher temperatures. A critical flaw in this approach is the inevitable generation of 1,3-dialkyl-2-imidazolidinimine, a dehydration by-product that can constitute 0.5% to several percentage points of the final mixture. The removal of this impurity is notoriously difficult because its boiling point is extremely close to that of the desired 1,3-dimethyl-2-imidazolidinone. Consequently, manufacturers are forced to utilize distillation towers with a very high number of theoretical plates or employ complex additional processing steps to achieve acceptable purity levels. Furthermore, even trace amounts of this imine by-product can act as a reaction inhibitor in downstream applications, such as suppressing polymerization reactions or interfering with the synthesis of aromatic amides, thereby limiting the commercial viability of the solvent in high-performance sectors.
The Novel Approach
The methodology disclosed in CN1067058C fundamentally alters the reaction kinetics to bypass these limitations through a sophisticated continuous addition strategy. Instead of a static batch charge, the process involves continuously adding both N,N'-dialkylethylenediamine and urea simultaneously into an aprotic polar solvent that is already maintained at a reaction temperature of not less than 180°C. This dynamic approach ensures that the reactants are immediately subjected to cyclization conditions, preventing the accumulation of the unstable intermediates that lead to imine formation. By maintaining a residence time of no less than 5 hours within this high-temperature environment, the reaction drives towards the complete formation of the imidazolidinone ring while thermodynamically suppressing the dehydration pathway. The outcome is a process capable of producing 1,3-dialkyl-2-imidazolidinone with a by-product content of less than 0.1% by weight, effectively rendering the impurity undetectable by standard gas chromatography. This structural purity is vital for applications requiring high-purity OLED material or pharmaceutical intermediates, where trace contaminants can ruin entire batches of final product.
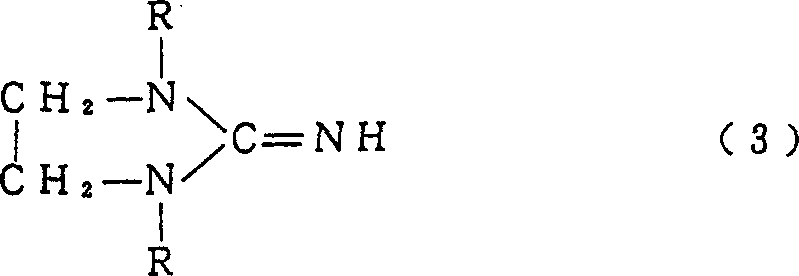
Mechanistic Insights into Continuous Thermal Cyclization
The success of this novel synthesis route lies in the precise control of reactant concentration and thermal energy distribution within the reaction vessel. In a conventional batch setup, the initial high concentration of diamine and urea at lower temperatures favors the formation of the linear diurea intermediate. As the temperature is subsequently raised to induce ring closure, the localized presence of this intermediate and the evolving water of condensation creates an environment conducive to dehydration, yielding the imine by-product. In contrast, the continuous addition method described in the patent maintains a steady-state concentration of reactants that is optimized for direct cyclization. By introducing the reagents into a solvent bath already heated to between 200°C and 260°C, the activation energy barrier for ring closure is overcome almost instantaneously upon contact. This kinetic control minimizes the lifetime of any potential dehydration-prone intermediates. Furthermore, the use of the product itself (1,3-dialkyl-2-imidazolidinone) as the reaction solvent creates an autogenic environment that stabilizes the transition state for cyclization, further enhancing selectivity towards the desired carbonyl-containing ring structure over the imine variant.
From an impurity control perspective, the mechanism effectively eliminates the 'batch effect' where temperature gradients and concentration spikes occur. The patent specifies that the reaction should be carried out under atmospheric pressure with continuous discharge of the product, which serves to remove the formed water and drive the equilibrium forward without allowing the reverse reaction or side reactions to gain a foothold. The strict requirement for an aprotic polar solvent, such as N-methyl-2-pyrrolidone or the product DMI itself, ensures that no proton exchange interferes with the nucleophilic attack of the amine on the urea carbonyl. This rigorous control over the reaction micro-environment results in a crude product purity that is so high (>98% before distillation) that the subsequent purification steps are drastically simplified. For a research director evaluating cost reduction in electronic chemical manufacturing, this mechanistic efficiency translates directly into lower energy consumption and higher throughput, as the distillation columns do not need to work as hard to separate components with nearly identical volatilities.
How to Synthesize 1,3-Dimethyl-2-Imidazolidinone Efficiently
The implementation of this continuous synthesis route requires careful attention to feed rates and thermal management to replicate the high yields reported in the patent data. The process is designed to be scalable, moving seamlessly from laboratory verification to commercial quantity production. To achieve the reported purity levels where the imine by-product is less than 0.1%, operators must ensure that the addition speed of the diamine and urea mixture allows for a residence time of at least 5 hours within the heated reactor zone. Detailed standardized synthetic steps for replicating this high-efficiency pathway are provided in the technical guide below, outlining the specific molar ratios and temperature profiles required for optimal performance.
- Prepare a reactor with an aprotic polar solvent (such as DMI itself) and heat to a reaction temperature of not less than 180°C, preferably between 200°C and 260°C.
- Continuously and simultaneously add N,N'-dialkylethylenediamine and urea into the heated solvent, maintaining a residence time of no less than 5 hours.
- Continuously discharge the reaction product and purify via distillation to obtain 1,3-dialkyl-2-imidazolidinone with less than 0.1% imine by-product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1067058C offers substantial strategic advantages beyond mere chemical purity. The shift from a batch process prone by-product accumulation to a continuous, high-selectivity process fundamentally alters the cost structure and reliability of the supply chain. By eliminating the formation of the difficult-to-separate imine by-product at the source, manufacturers can significantly reduce the complexity of the downstream purification train. This reduction in processing complexity translates to lower operational expenditures, as there is less need for extensive fractional distillation columns with high theoretical plate counts, which are capital-intensive and energy-hungry. Furthermore, the ability to run the reaction continuously at atmospheric pressure enhances plant safety and operational stability, reducing the risks associated with high-pressure batch autoclaves and enabling more predictable production schedules.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the drastic simplification of the purification process. In conventional methods, removing the imine by-product requires significant energy input and large distillation infrastructure due to the close boiling points of the product and impurity. By suppressing the by-product to levels below 0.1% during the reaction phase, the new method minimizes the load on purification units. This leads to substantial cost savings in terms of steam consumption, cooling water usage, and equipment maintenance. Additionally, the high yield (reported up to 97-98% in examples) ensures that raw material utilization is maximized, reducing the cost per kilogram of the final active ingredient or solvent.
- Enhanced Supply Chain Reliability: Continuous processing inherently offers greater supply chain resilience compared to batch operations. The ability to run the reactor for extended periods with continuous feed and discharge allows for a steady output of high-purity 1,3-dialkyl-2-imidazolidinone, smoothing out inventory fluctuations. This stability is critical for customers in the pharmaceutical and agrochemical sectors who require consistent quality for their own regulatory filings. The robustness of the process, which tolerates standard industrial heating means and operates at atmospheric pressure, further reduces the likelihood of unplanned shutdowns due to equipment failure or safety interlocks, ensuring a reliable flow of materials to downstream users.
- Scalability and Environmental Compliance: The process is designed with commercial scale-up in mind, utilizing standard industrial unit operations such as pumps, heated reactors, and distillation columns. The elimination of heavy metal catalysts or exotic reagents simplifies waste stream management, aligning with increasingly stringent environmental regulations. The high selectivity of the reaction means less chemical waste is generated in the form of off-spec by-products, contributing to a greener manufacturing footprint. This environmental efficiency not only reduces disposal costs but also enhances the marketability of the product to eco-conscious multinational corporations seeking sustainable supply chain partners for their complex polymer additives or specialty chemical needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of high-purity imidazolidinones based on the patented continuous addition methodology. These insights are derived directly from the experimental data and claims within CN1067058C, providing clarity on how this process overcomes historical manufacturing bottlenecks. Understanding these details is essential for technical teams evaluating the feasibility of integrating this material into sensitive electronic or pharmaceutical formulations.
Q: Why is the continuous addition method superior to batch processing for DMI synthesis?
A: Conventional batch methods often result in 0.5% to several percent of 1,3-dialkyl-2-imidazolidinimine by-product due to intermediate accumulation. The continuous addition method described in CN1067058C maintains optimal reactant concentrations and high temperatures (>180°C) throughout, suppressing this dehydration side reaction to below 0.1%.
Q: What are the critical temperature parameters for minimizing impurities?
A: The reaction must be conducted at temperatures not less than 180°C, with an optimal range of 200°C to 260°C. Temperatures below this threshold risk the accumulation of the 1,1'-dialkyl-1,1'-dimethylene diurea intermediate, which lowers yield and complicates purification.
Q: How does this process impact downstream purification costs?
A: Since the boiling point of the imine by-product is very close to the target DMI product, conventional methods require distillation columns with many theoretical plates for separation. By reducing the by-product to undetectable levels (<0.1%) at the source, this process significantly simplifies the distillation burden and energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Dimethyl-2-Imidazolidinone Supplier
The technological advancements detailed in patent CN1067058C highlight the critical importance of process control in achieving ultra-high purity standards for polar aprotic solvents. At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovative synthetic routes to life. Our engineering team specializes in adapting continuous thermal cyclization processes to ensure that every batch meets stringent purity specifications, specifically targeting the elimination of trace imine impurities that can compromise downstream performance. With our rigorous QC labs and state-of-the-art distillation capabilities, we guarantee that our 1,3-dimethyl-2-imidazolidinone delivers the consistency and reliability required by top-tier global manufacturers.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for high-purity 1,3-dialkyl-2-imidazolidinones. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates how switching to our continuously produced grades can reduce your overall manufacturing costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique application requirements, ensuring that your production processes remain uninterrupted and compliant with the highest industry standards.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
