Revolutionizing Picoplatin Manufacturing: A Green Aqueous Route for High-Purity API Intermediates
The landscape of oncology drug manufacturing is constantly evolving, driven by the need for higher purity, lower costs, and greener processes. A pivotal advancement in this domain is detailed in patent CN101775040B, which outlines a novel preparation method for Picoplatin (CAS: 181630-15-9), also known as ZD0473 or JM473. As a next-generation platinum-based antitumor agent designed to overcome cisplatin resistance through steric hindrance, Picoplatin represents a high-value target for pharmaceutical supply chains. The patented technology introduces a groundbreaking aqueous synthetic route that replaces traditional organic solvents with water, utilizing a unique iodide activation strategy to enhance reaction kinetics and product yield. This shift not only addresses the stringent purity requirements for injectable anticancer drugs but also offers a scalable, environmentally benign alternative to legacy manufacturing protocols.
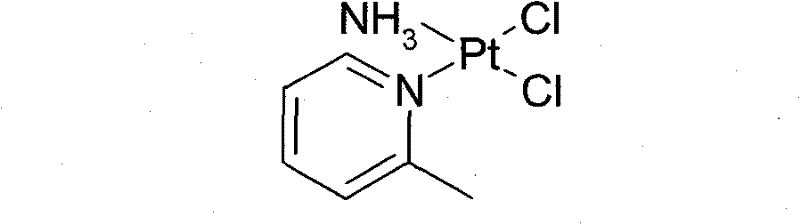
For R&D directors evaluating process feasibility, understanding the limitations of historical synthesis routes is crucial. Prior art methods, such as those disclosed in EP072430 and US5665771, often relied on expensive and difficult-to-source starting materials like potassium amminetrichloroplatinate, resulting in overall yields of merely 50%. Furthermore, alternative routes utilizing potassium tetrachloroplatinate in solvents like DMF (N,N-dimethylformamide) faced significant hurdles. As illustrated in earlier literature (Can. J. Chem., 1978), reactions in DMF at 65°C–80°C frequently generated unstable Pt-DMF complexes and insoluble black platinum impurities, complicating purification.
Subsequent improvements attempted to mitigate these issues by switching to solvents like NMP (N-Methyl-2-pyrrolidone) as seen in US6413953. While this reduced side reactions, it introduced new supply chain bottlenecks. The high boiling point of NMP (204°C) made solvent recovery energy-intensive and difficult under reduced pressure. To isolate the product, manufacturers were forced to use large volumes of methylene chloride and ether as anti-solvents, creating a complex, multi-solvent workflow that increased both operational expenditure and environmental liability. These conventional methods highlight a clear industry pain point: the trade-off between reaction efficiency and process complexity in platinum coordination chemistry.
Mechanistic Insights into Iodide-Activated Platinum Coordination
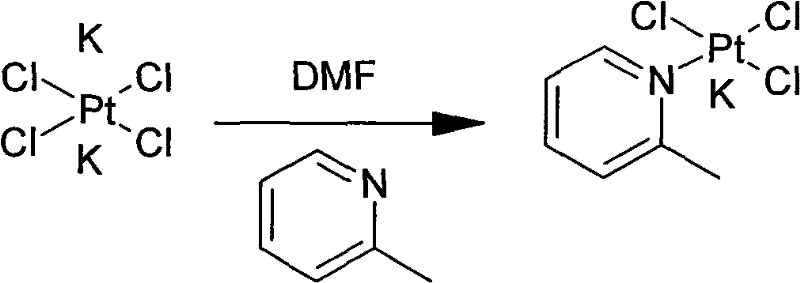
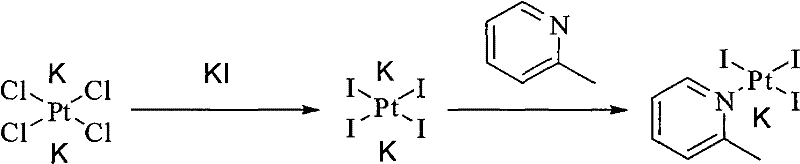
The core innovation of the patented process lies in its strategic manipulation of the platinum coordination sphere through halide exchange. Instead of forcing the weakly basic 2-picoline to displace chloride ligands directly under harsh conditions, the process first converts potassium tetrachloroplatinate into potassium tetraiodoplatinate using potassium iodide. Iodide is a softer ligand and a better leaving group than chloride in certain substitution contexts, which significantly activates the platinum center. This activation allows the subsequent coordination of 2-picoline to proceed smoothly in water at mild temperatures (15°C–30°C), forming the potassium triiodo(2-picoline)platinate(II) intermediate with high fidelity.
Following the formation of the triiodo intermediate, the process employs a controlled ligand substitution with ammonia. The reaction conditions are meticulously optimized to ensure the formation of the cis-diiodo-ammonia-(2-picoline)platinum(II) species. The final transformation involves a classic metathesis reaction where silver nitrate is introduced to precipitate iodide ions as silver iodide (AgI). Due to the extremely low solubility product constant (Ksp) of AgI, this step effectively scavenges iodide from the solution. The subsequent addition of potassium chloride facilitates the final ligand exchange, crystallizing the target Picoplatin. This mechanism ensures that the final product is free from heavy metal contaminants and residual silver, a critical quality attribute for parenteral formulations.
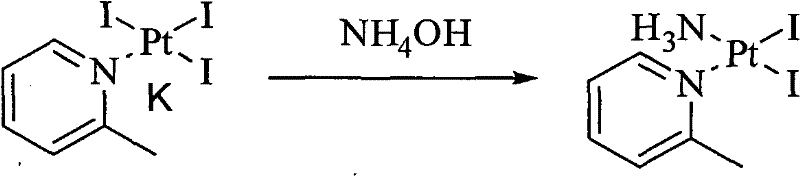
How to Synthesize Picoplatin Efficiently
The patented methodology offers a streamlined, three-step protocol that is highly amenable to industrial scale-up. By leveraging simple inorganic reagents and water as the primary medium, the process minimizes the need for specialized equipment required for handling toxic volatiles. The following guide outlines the standardized synthesis steps derived from the patent embodiments, ensuring reproducibility and high yield.
- React potassium tetrachloroplatinate with potassium iodide in water to form potassium tetraiodoplatinate, then add 2-picoline to generate potassium triiodo(2-picoline)platinate(II).
- Treat the triiodo intermediate with ammonium hydroxide in an aqueous medium to substitute one iodide ligand with ammonia, forming cis-diiodo-ammonia-(2-picoline)platinum(II).
- React the diiodo complex with silver nitrate to precipitate silver iodide, filter, and add potassium chloride to the filtrate to crystallize the final Picoplatin product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous-based synthesis represents a significant opportunity for cost optimization and risk mitigation. The elimination of high-boiling organic solvents like NMP and DMF removes the need for complex distillation and recovery units, thereby reducing capital expenditure and energy consumption. Furthermore, the reliance on commodity chemicals such as potassium iodide, silver nitrate, and potassium chloride ensures a robust and resilient supply chain, unaffected by the volatility of specialized organic solvent markets.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by replacing expensive organic solvents with deionized water. In traditional methods, the recovery of solvents like NMP is energy-intensive due to high boiling points, often requiring vacuum systems that increase utility costs. By contrast, the aqueous nature of this reaction allows for simpler work-up procedures. Additionally, the high yields reported in the patent embodiments (up to 96-100% in final steps) indicate a highly atom-economical process that maximizes the utilization of precious platinum raw materials, directly lowering the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Sourcing high-purity intermediates is often a bottleneck in API production. This method utilizes widely available starting materials like potassium tetrachloroplatinate and 2-picoline, avoiding the dependency on scarce precursors like potassium amminetrichloroplatinate mentioned in older patents. The simplified reaction conditions (15°C–30°C) also reduce the risk of batch failures due to thermal runaway or equipment malfunction, ensuring consistent delivery schedules for downstream pharmaceutical partners.
- Scalability and Environmental Compliance: Regulatory pressure to reduce VOC emissions is intensifying globally. This synthesis route is inherently greener, generating minimal organic waste. The primary byproduct, silver iodide, is an insoluble solid that can be easily filtered and potentially recycled for silver recovery, aligning with circular economy principles. The absence of toxic solvent residues simplifies the purification process, reducing the burden on quality control laboratories and accelerating the release of batches for clinical or commercial use.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this novel Picoplatin synthesis route. These insights are derived directly from the comparative data and experimental embodiments provided in the patent documentation, offering clarity on process robustness and quality control.
Q: Why is the iodide activation strategy superior to direct chlorination in Picoplatin synthesis?
A: The patent CN101775040B demonstrates that converting the platinum precursor to a tetraiodo species significantly increases substrate activity. This allows subsequent ligand substitutions with 2-picoline and ammonia to proceed efficiently at mild temperatures (15°C–30°C) in water, avoiding the harsh conditions and stable solvent-complex impurities associated with DMF or NMP-based chlorination routes.
Q: How does this process address silver ion contamination concerns?
A: The process utilizes the extremely low solubility product of silver iodide (AgI, Ksp=8.51×10^-17). By reacting the iodo-intermediate with silver nitrate, silver ions are quantitatively removed as an insoluble AgI precipitate. This ensures the final Picoplatin product is virtually free of residual silver ions, a critical quality attribute for injectable oncology drugs.
Q: What are the environmental advantages of this aqueous synthesis method?
A: Unlike prior art methods that rely on high-boiling polar aprotic solvents like NMP or DMF which require complex recovery systems or large volumes of anti-solvents for precipitation, this invention uses deionized water as the sole reaction medium. This drastically reduces volatile organic compound (VOC) emissions, simplifies waste treatment, and lowers the overall environmental footprint of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Picoplatin Supplier
The technological breakthroughs detailed in CN101775040B underscore the potential for more efficient and sustainable production of critical oncology intermediates. At NINGBO INNO PHARMCHEM, we specialize in translating such innovative laboratory protocols into robust commercial realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of aqueous chemistry are fully realized in large-scale reactors. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of Picoplatin meets the exacting standards required for global pharmaceutical registration.
We invite procurement leaders and technical directors to collaborate with us on optimizing their supply chains for platinum-based therapeutics. By leveraging our expertise in coordination chemistry and process engineering, we can help you achieve significant operational efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your project's unique volume and timeline requirements.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
