Advanced In Situ Synthesis of Quaternary Ammonium Carbonates for Industrial Scale Production
Advanced In Situ Synthesis of Quaternary Ammonium Carbonates for Industrial Scale Production
The chemical industry is constantly seeking more efficient and safer pathways for producing high-value biocidal agents, and patent CN1464873A presents a transformative approach to synthesizing quaternary ammonium carbonates. This intellectual property details a robust in situ method that converts tertiary amines directly into quaternary ammonium methylcarbonates and alkylcarbonates using methanol and cyclic carbonates or esters. Unlike conventional techniques that rely on hazardous intermediate steps, this innovation enables a one-pot reaction sequence that significantly streamlines the manufacturing workflow. For R&D directors and procurement specialists alike, understanding this technology is crucial for optimizing the production of wood preservatives and industrial antimicrobials. The ability to bypass the formation of corrosive quaternary ammonium hydroxides represents a major leap forward in process safety and operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of quaternary ammonium carbonates has been plagued by significant logistical and safety challenges inherent to the traditional ion-exchange pathways. Conventional processes typically involve reacting a quaternary ammonium chloride with a metal hydroxide to generate a highly corrosive quaternary ammonium hydroxide intermediate. This intermediate is unstable and dangerous to handle, requiring specialized equipment and rigorous safety protocols to prevent equipment degradation and personnel injury. Furthermore, this exchange reaction inevitably produces stoichiometric amounts of metal chloride salts as by-products, which must be laboriously filtered out from the reaction mixture. This filtration step not only adds substantial operational time and cost but also introduces potential points of failure where product loss or contamination can occur, thereby reducing the overall yield and economic viability of the process.
The Novel Approach
In stark contrast, the methodology disclosed in CN1464873A circumvents these issues by employing a direct alkylation strategy using cyclic carbonates or esters in the presence of methanol. This novel approach allows for the in situ generation of the carbonate anion without ever forming the hazardous hydroxide species. By reacting a tertiary amine directly with a cyclic carbonate like propylene carbonate, the system produces the desired quaternary ammonium methylcarbonate along with a valuable diol by-product. This eliminates the need for metal hydroxides entirely and removes the requirement for filtration steps to separate inorganic salts. The result is a cleaner reaction profile that simplifies downstream processing and enhances the overall safety of the chemical plant environment.
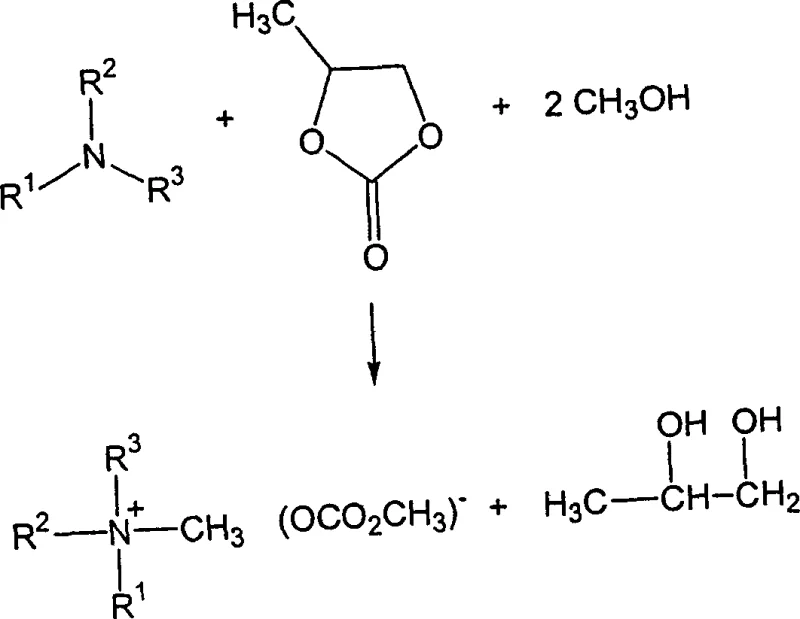
The reaction pathway illustrated above demonstrates the elegance of this transformation, where the cyclic carbonate ring opens to facilitate the quaternization of the nitrogen center. This direct route ensures that the counter-ion is introduced simultaneously with the alkyl group, maintaining charge balance without external salt formation. For a reliable wood preservative intermediate supplier, adopting this technology means delivering a product with a superior impurity profile compared to legacy methods. The absence of metal ions is particularly beneficial for applications where ionic contamination could interfere with formulation stability or end-use performance.
Mechanistic Insights into In Situ Transesterification and Alkylation
To fully appreciate the technical depth of this patent, one must examine the proposed mechanistic hypothesis regarding the transesterification events that drive the reaction kinetics. The inventors postulate that the cyclic carbonate first undergoes a transesterification reaction with methanol, catalyzed by the tertiary amine itself, to form dimethyl carbonate and a diol. This generated dimethyl carbonate then acts as the active methylating agent that reacts with the remaining tertiary amine to yield the final quaternary ammonium methylcarbonate. This two-step cascade occurring within a single vessel highlights the sophisticated interplay between nucleophilic attack and leaving group ability in this system.
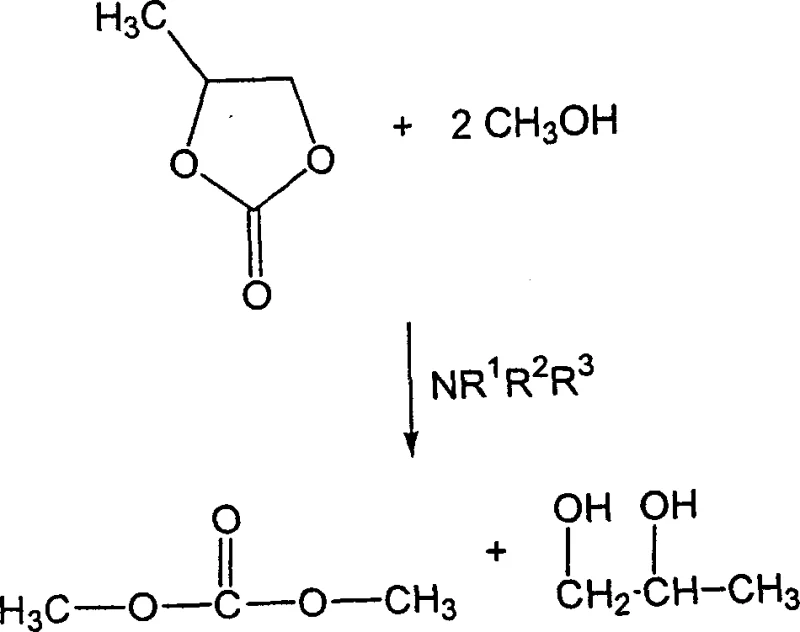
Understanding this mechanism is vital for process optimization, as it suggests that the concentration of methanol and the choice of cyclic carbonate can be tuned to control the rate of dimethyl carbonate formation. If the transesterification is too slow, it may become the rate-limiting step, whereas an excess of methanol can drive the equilibrium towards the reactive methyl carbonate species. Additionally, the co-production of diols such as propylene glycol is not merely a waste stream but a functional component that can enhance the flash point of the final formulation and act as an antifreeze agent. This dual functionality adds value to the process output, turning a potential by-product into a performance-enhancing additive within the final commercial product.
How to Synthesize Quaternary Ammonium Methyl Carbonates Efficiently
Implementing this synthesis route requires precise control over reaction parameters to ensure high conversion rates and product purity. The process generally involves charging a reactor with the tertiary amine, methanol, and the carbonate source, followed by heating to temperatures between 120°C and 160°C. Maintaining these conditions for a duration of 3 to 40 hours allows the equilibrium to shift sufficiently towards the quaternary salt. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Charge a reactor with tertiary amine, methanol, and a cyclic carbonate source such as propylene carbonate in a molar ratio favoring excess carbonate.
- Heat the mixture to between 120°C and 160°C under pressure for 3 to 40 hours to facilitate transesterification and quaternization.
- Distill off excess methanol and dimethyl carbonate, then hydrolyze the intermediate methyl carbonate with water to yield the final bicarbonate or carbonate salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this in situ synthesis method offers profound advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies. The elimination of metal hydroxides and the associated filtration steps translates directly into reduced operational expenditures and lower capital requirements for waste treatment infrastructure. By simplifying the unit operations involved, manufacturers can achieve faster batch turnover times and reduce the overall energy consumption per kilogram of product produced. This efficiency gain is critical for maintaining competitiveness in the global market for specialty chemicals and biocides.
- Cost Reduction in Manufacturing: The removal of expensive metal hydroxides and the avoidance of filtration equipment significantly lowers the variable costs associated with production. Furthermore, the ability to recover and recycle excess methanol and dimethyl carbonate through simple distillation enhances the atom economy of the process. This closed-loop capability minimizes raw material waste and reduces the dependency on volatile commodity pricing for disposable reagents. Consequently, the overall cost structure for producing high-purity quaternary ammonium carbonates becomes more predictable and resilient against market fluctuations.
- Enhanced Supply Chain Reliability: The raw materials required for this process, such as propylene carbonate and methanol, are commodity chemicals with robust and established global supply chains. Unlike specialized reagents that may suffer from availability bottlenecks, these feedstocks ensure a consistent input flow for continuous manufacturing operations. This reliability is essential for meeting the demanding delivery schedules of large-scale agrochemical and industrial clients who depend on uninterrupted supply lines. Diversifying the supply base for these common inputs further mitigates the risk of production stoppages due to raw material shortages.
- Scalability and Environmental Compliance: The one-pot nature of this reaction makes it inherently scalable from pilot plants to multi-ton commercial reactors without complex engineering modifications. The reduction in hazardous waste generation, specifically the absence of metal salt sludge, simplifies environmental compliance and lowers disposal costs. This aligns perfectly with modern green chemistry principles, allowing companies to market their products as sustainably manufactured. The streamlined process also reduces the physical footprint required for production, enabling higher output volumes within existing facility constraints.
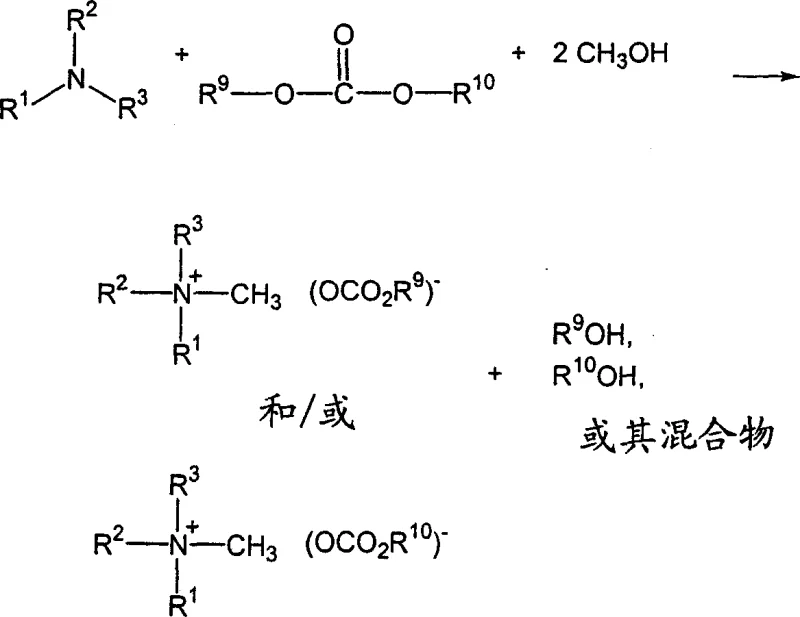
The versatility of this chemistry is further demonstrated by the ability to use various esters as carbonate sources, as shown in the reaction scheme above. This flexibility allows manufacturers to tailor the alkyl chain of the carbonate anion by selecting specific esters, thereby customizing the physicochemical properties of the final salt. Such adaptability is invaluable for developing specialized formulations that require specific solubility or stability characteristics. It empowers R&D teams to innovate rapidly without being constrained by rigid synthetic limitations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance. They serve as a quick reference for stakeholders evaluating the feasibility of integrating this process into their current manufacturing portfolios.
Q: Why is the in situ method superior to traditional hydroxide exchange?
A: Traditional methods require handling highly corrosive quaternary ammonium hydroxides and generate metal salt by-products that necessitate expensive filtration steps. The in situ method described in CN1464873A avoids these hazards entirely by generating the carbonate anion directly within the reaction matrix.
Q: What are the primary raw materials required for this synthesis?
A: The process utilizes readily available tertiary amines, methanol, and cyclic carbonates like propylene carbonate or ethylene carbonate. These feedstocks are commercially abundant, ensuring a stable supply chain for large-scale manufacturing.
Q: How does this process impact the purity of the final biocide?
A: By eliminating metal chloride contaminants and avoiding the degradation pathways associated with strong hydroxides, this route yields high-purity quaternary ammonium carbonates. The co-produced diols can remain in the solution to enhance flash points without compromising antimicrobial efficacy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quaternary Ammonium Carbonates Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this in situ synthesis technology to redefine the standards of quality and efficiency in the biocide industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab scale to full manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the highest international standards for industrial applications.
We invite you to collaborate with our technical procurement team to explore how this innovative route can benefit your specific product lines. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic advantages of switching to this metal-free process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique requirements, ensuring a partnership built on transparency and technical excellence.
