Advanced Manufacturing of Triazolo Pyrimidine Derivatives for Pharmaceutical Applications
Advanced Manufacturing of Triazolo Pyrimidine Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for bioactive heterocyclic compounds, particularly those serving as critical intermediates for respiratory therapies. Patent CN1217947C discloses a highly optimized manufacture method for triazolo pyrimidine derivatives characterized by the general chemical formula (I). These compounds are renowned for their efficacy in preventing bronchospasm and treating diseases associated with bronchial muscle contraction, such as asthma and bronchitis. The disclosed innovation represents a significant leap forward in process chemistry, addressing the longstanding inefficiencies of prior art by introducing a rapid preparation protocol for diamino-1,2,4-triazole and a highly selective condensation strategy. By effectively managing amine protection and deprotection cycles, this methodology not only streamlines the synthetic pathway but also drastically improves the overall economic viability of producing these valuable pharmacological agents.
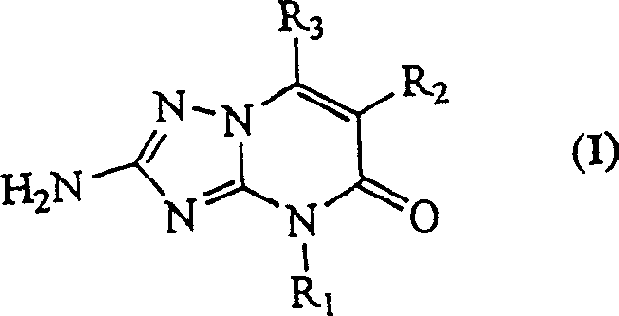
For procurement specialists and supply chain managers, understanding the structural versatility of Formula (I) is paramount. The substituents R1, R2, and R3 offer a wide range of chemical diversity, allowing for the tuning of physicochemical properties to meet specific drug development requirements. R1 can vary from hydrogen to various alkyl, cycloalkyl, or alkenyl groups, while R2 and R3 accommodate halogens, hydroxyalkyls, and alkyl chains. This flexibility ensures that the manufacturing process described is not limited to a single molecule but serves as a versatile platform technology. As a reliable triazolo pyrimidine derivative supplier, leveraging such a flexible and robust synthetic platform allows for the rapid adaptation to changing market demands and the efficient production of diverse analogues required for clinical trials and commercial launches.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of triazolo pyrimidine derivatives was plagued by significant operational bottlenecks that hindered commercial scalability. As documented in United States Patent 3,689,488, the conventional route involved a multi-step process that was both time-consuming and chemically inefficient. The preparation of the key intermediate, diamino-1,2,4-triazole, typically required several days to complete, creating a major bottleneck in the production schedule. Furthermore, the subsequent condensation reaction with alpha,beta-unsaturated acid derivatives was notoriously non-selective, often yielding complex mixtures of isomers that necessitated extensive and costly purification procedures. Perhaps most critically, the deprotection of the benzyl group via hydrogenolysis was an arduous process, reportedly taking up to ten days to reach completion. These factors combined to create a manufacturing process with low throughput, high operational costs, and significant supply chain risks.
The Novel Approach
The methodology presented in CN1217947C fundamentally re-engineers the synthetic pathway to overcome these historical limitations. The core innovation lies in the rapid generation of the triazole core using dialkylcyanodisulfide imine as a starting material. This approach bypasses the sluggish kinetics of traditional methods, enabling the formation of the diamino-1,2,4-triazole scaffold in a fraction of the time. Additionally, the introduction of an imine-based protection strategy allows for precise control over the reactivity of the amine groups during the critical cyclization step. By selectively condensing the protected imine with the unsaturated acid derivative, the process minimizes the formation of unwanted isomers, thereby simplifying downstream purification. This strategic shift from non-selective to selective chemistry is a game-changer for cost reduction in pharmaceutical intermediates manufacturing, as it directly translates to higher yields and reduced waste generation.
Mechanistic Insights into Selective Condensation and Cyclization
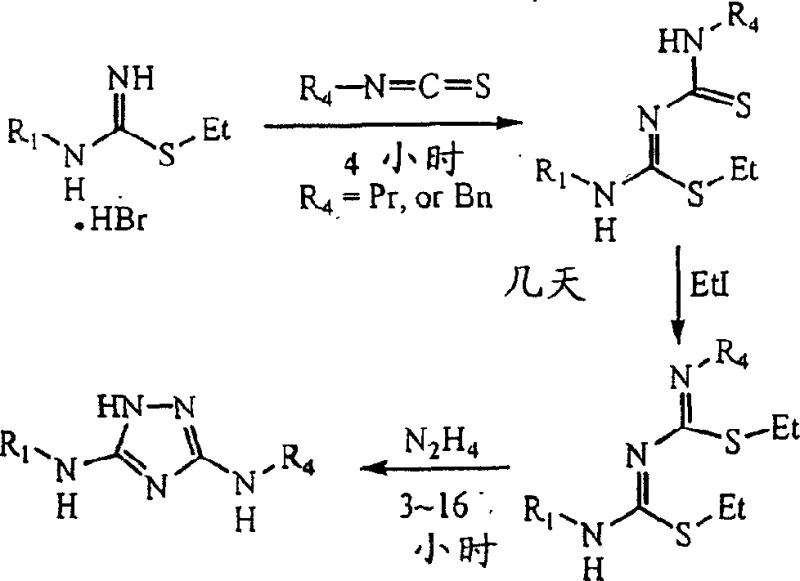
The mechanistic elegance of this process is best understood by examining the stepwise transformation of the intermediates. The synthesis initiates with the reaction of dialkylcyanodisulfide imine (Formula V) with an alkylamine (Formula IV) under reflux conditions. This nucleophilic attack facilitates the rapid assembly of the 1,2,4-triazole ring, yielding the diamino compound (Formula III). The efficiency of this step is attributed to the high electrophilicity of the cyano group in the presence of the disulfide moiety, which drives the reaction to completion rapidly at room temperature followed by reflux. Following this, the amine group is protected by reacting with an aldehyde to form the imine (Formula VI). This protection is crucial as it masks the nucleophilicity of one amine, directing the subsequent cyclization to occur at the desired position, thus ensuring regioselectivity.
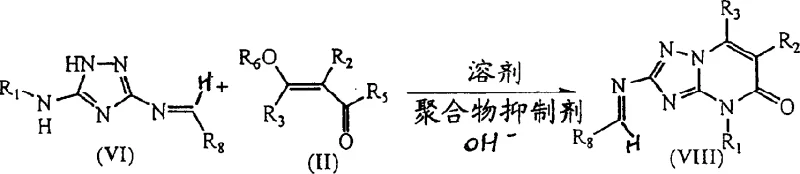
The pivotal step in the entire sequence is the condensation of the protected imine (Formula VI) with the alpha,beta-unsaturated acid derivative (Formula II). This reaction is conducted in the presence of a base, such as a metal carbonate, and critically, a polymer inhibitor like mequinol. The base facilitates the Michael addition of the triazole nitrogen to the unsaturated system, initiating the ring closure to form the pyrimidine moiety. The polymer inhibitor plays a vital role in scavenging free radicals that could otherwise lead to the polymerization of the unsaturated acid derivative, a common side reaction that plagues such condensations. The result is the formation of the protected triazolo pyrimidine intermediate (Formula VIII) with high fidelity. Finally, mild acid hydrolysis removes the imine protecting group, revealing the free amine and delivering the final target molecule (Formula I) in high purity.
How to Synthesize Triazolo Pyrimidine Derivative Efficiently
The synthesis of these complex heterocycles requires precise control over reaction parameters to maximize yield and purity. The patented process outlines a clear, four-step protocol that balances reaction kinetics with operational simplicity. The initial formation of the triazole core sets the stage for the entire synthesis, requiring careful stoichiometry of the dialkylcyanodisulfide imine and the amine source. Subsequent protection and condensation steps demand strict temperature control and the use of specific additives to prevent side reactions. For R&D teams looking to implement this chemistry, adherence to the specified solvent systems and catalyst loads is essential to replicate the high efficiency reported in the patent examples. The detailed standardized synthesis steps for this process are outlined below.
- Rapidly prepare diamino-1,2,4-triazole from dialkylcyanodisulfide imine and alkylamine under reflux.
- React the diamino-triazole with an aldehyde to form an imine, effectively protecting the amine group.
- Perform selective condensation with an alpha,beta-unsaturated acid derivative in the presence of a base and polymer inhibitor.
- Hydrolyze the resulting imine intermediate under acidic conditions to yield the final triazolo pyrimidine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel manufacturing route offers substantial advantages for procurement and supply chain stakeholders. The primary benefit stems from the drastic reduction in cycle times compared to legacy methods. By eliminating the ten-day hydrogenolysis step and accelerating the triazole formation from days to hours, the overall throughput of the manufacturing facility is significantly enhanced. This acceleration allows for more frequent production batches, thereby improving inventory turnover and reducing the capital tied up in work-in-progress materials. Furthermore, the use of selective condensation minimizes the generation of difficult-to-remove isomers, which reduces the burden on purification resources and lowers the consumption of chromatography media or recrystallization solvents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and time-intensive steps. Traditional methods relying on prolonged hydrogenolysis require specialized high-pressure equipment and precious metal catalysts, both of which represent significant capital and operational expenditures. By replacing this with a simple acid hydrolysis step, the new method removes the need for costly catalyst recovery and specialized reactor infrastructure. Additionally, the high selectivity of the condensation step reduces raw material waste, as less starting material is lost to side products. This streamlined approach leads to substantial cost savings in pharmaceutical intermediates manufacturing, making the final API more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the predictability and speed of the manufacturing process. The conventional route, with its multi-day reaction times, was highly susceptible to disruptions and equipment bottlenecks. The new method, with its rapid reaction kinetics and robust operating conditions, ensures a consistent and reliable output. The use of common, commercially available solvents like acetonitrile, toluene, and ethanol further mitigates supply risk, as these materials are readily sourced from multiple vendors. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug manufacturers receive their materials on schedule to meet clinical and commercial deadlines.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden challenges, but this methodology is inherently scalable. The reactions operate under standard reflux conditions without the need for extreme temperatures or pressures, simplifying the engineering requirements for large-scale reactors. Moreover, the reduction in reaction time and the avoidance of heavy metal catalysts contribute to a greener manufacturing profile. The simplified workup procedures reduce the volume of organic waste generated per kilogram of product, aligning with modern environmental compliance standards. This ease of commercial scale-up of complex pharmaceutical intermediates ensures that the supply can grow seamlessly with market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of triazolo pyrimidine derivatives. These answers are derived directly from the technical specifications and comparative data provided in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of this route for their specific projects. The insights provided here clarify the operational benefits and chemical advantages that distinguish this method from traditional approaches.
Q: How does this novel method improve upon conventional synthesis routes?
A: Conventional methods, such as those disclosed in US Patent 3,689,488, suffer from extremely long reaction times, often requiring several days for triazole preparation and up to ten days for deprotection. Furthermore, they often produce non-selective mixtures requiring complex purification. The novel method described in CN1217947C utilizes dialkylcyanodisulfide imine for rapid triazole formation and employs a selective condensation strategy that significantly enhances production efficiency and reduces overall processing time.
Q: What are the critical reagents for ensuring high selectivity in the condensation step?
A: The selectivity of the condensation reaction between the protected imine and the alpha,beta-unsaturated acid derivative is crucial. The process relies on the presence of specific bases, such as metal carbonates or bicarbonates, and critically, the addition of polymer inhibitors like quinhydrones or monomethyl ether quinhydrones. These additives prevent unwanted polymerization side reactions, ensuring the formation of the desired triazolo pyrimidine skeleton with high purity.
Q: Is this manufacturing process suitable for large-scale commercial production?
A: Yes, the process is designed with scalability in mind. It utilizes common organic solvents such as acetonitrile, toluene, and alcohols, and operates under standard reflux conditions. The elimination of prolonged hydrogenolysis steps and the use of robust acid hydrolysis for deprotection make the workflow highly amenable to industrial scale-up, offering a reliable supply chain solution for high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazolo Pyrimidine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the development of life-saving respiratory medications. The technology described in CN1217947C exemplifies the kind of process innovation we champion to deliver value to our partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of triazolo pyrimidine derivative meets the highest international standards for pharmaceutical intermediates.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains. By leveraging our expertise in this advanced synthesis method, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can accelerate the development of your respiratory therapy pipeline while achieving significant operational efficiencies.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
