Advanced Synthesis of Azanol Ether Intermediates for Crop Protection and Pharma
Advanced Synthesis of Azanol Ether Intermediates for Crop Protection and Pharma
The development of efficient synthetic routes for functionalized hydroxylamine derivatives is a critical bottleneck in the manufacturing of modern crop protection agents and pharmaceutical active ingredients. Patent CN1095708A introduces a transformative methodology for the preparation of azanol ethers of Formula I and their corresponding salts, addressing long-standing challenges in yield, purity, and process safety. This technology enables the direct O-alkylation of oximido compounds using stable sulfonic acid esters, bypassing the complex and waste-generating protecting group strategies historically employed in the industry. By leveraging mild alkaline conditions and dipolar aprotic solvents, this approach offers a robust pathway for producing high-purity intermediates essential for herbicides and medicinal chemistry applications.
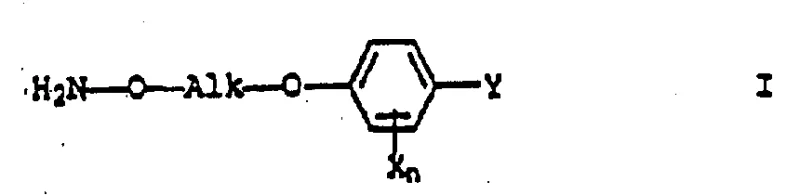
As a reliable agrochemical intermediate supplier, understanding the structural versatility of Formula I is paramount. The substituents X and Y can be independently selected from nitro, cyano, halogen, or alkyl groups, allowing for the fine-tuning of biological activity and physicochemical properties. The alkylene bridge (Alk) provides necessary spacing between the aromatic ring and the aminooxy functionality, a structural motif frequently found in potent herbicidal agents. The ability to access these diverse analogues through a unified synthetic platform significantly enhances the speed of process development for new active ingredients, positioning this technology as a cornerstone for next-generation agricultural chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of O-substituted hydroxylamines has been plagued by inefficiencies associated with protecting group chemistry. Conventional routes often rely on the formation of N-hydroxyphthalimide derivatives, which require subsequent cleavage steps to reveal the desired aminooxy functionality. This deprotection step is notoriously problematic, frequently leading to the formation of intractable coupling byproducts such as phthalohydrazide and hydrazine derivatives that are difficult to separate from the target molecule. Furthermore, the recovery of the phthalimide protecting group is generally economically unfeasible, leading to significant atom economy losses and increased waste disposal costs. Alternative methods utilizing direct alkylation with alkyl halides often demand the use of strong, moisture-sensitive bases like sodium hydride or organolithium reagents under strictly anhydrous conditions, posing severe safety risks and operational complexities for large-scale production facilities.
The Novel Approach
The methodology disclosed in CN1095708A represents a paradigm shift by utilizing alkyl sulfonates (Formula III) as superior alkylating agents. Unlike volatile and reactive alkyl halides, sulfonic acid esters exhibit enhanced stability and reactivity profiles that allow the reaction to proceed efficiently with milder bases such as alkali metal hydroxides, alcoholates, or carbonates. This innovation eliminates the necessity for rigorous anhydrous environments and expensive pyrophoric reagents, thereby drastically simplifying the reactor setup and operational protocols. The process facilitates the direct condensation of the oximido compound (Formula II) with the sulfonate, yielding the oximido derivative (Formula IV) in high purity. Subsequent acid hydrolysis cleanly converts this intermediate into the target azanol ether salt, avoiding the generation of stubborn organic byproducts and enabling a streamlined isolation procedure that is ideally suited for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Sulfonate-Mediated O-Alkylation
The core of this technological advancement lies in the nucleophilic substitution mechanism where the oxygen anion of the oximido compound attacks the electrophilic carbon of the sulfonate ester. The preparation of the critical alkylating agent begins with the reduction of phenoxy alkanoic acid esters to the corresponding phenoxy alkanols (Formula V), a transformation that can be achieved using standard reducing agents like lithium aluminum hydride or sodium borohydride. This step ensures the availability of the hydroxyl precursor from commodity starting materials, securing the supply chain against volatility in specialized reagent markets. The subsequent activation of the alcohol involves reaction with sulfonyl chlorides, such as methanesulfonyl chloride, in the presence of a tertiary amine base to form the mesylate (Formula III).
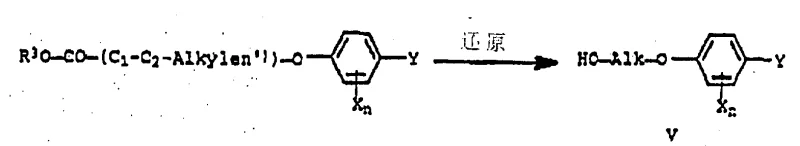
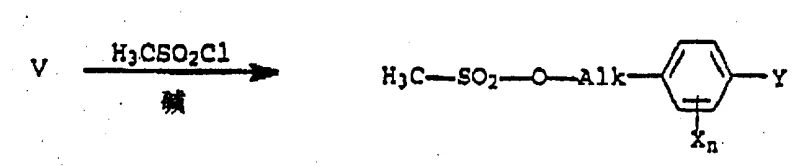
This activation step is crucial as it converts a poor leaving group (hydroxyl) into an excellent leaving group (mesylate), facilitating the subsequent coupling reaction under mild thermal conditions (typically 60-100°C). The use of dipolar aprotic solvents such as N-methylpyrrolidone (NMP) or dimethylformamide (DMF) is instrumental in stabilizing the transition state and solubilizing the ionic intermediates without participating in side reactions. Unlike halide-based alkylations which may suffer from elimination side reactions or require cryogenic temperatures to control exotherms, this sulfonate-based route proceeds smoothly with excellent chemoselectivity. The mechanistic robustness ensures that impurities arising from solvent degradation or reagent decomposition are minimized, resulting in a crude product profile that often meets stringent purity specifications without the need for extensive chromatographic purification, a key factor for commercial viability.
How to Synthesize Azanol Ether Efficiently
The synthesis of these valuable intermediates follows a logical sequence designed for industrial robustness, beginning with the preparation of the sulfonate alkylating agent and concluding with the hydrolytic cleavage of the oxime protecting group. The process is characterized by its tolerance to various functional groups and its adaptability to different scales of operation. Detailed standard operating procedures regarding stoichiometry, addition rates, and specific workup protocols are essential for maximizing yield and ensuring operator safety during the handling of alkaline mixtures and acidic hydrolysis steps.
- Preparation of the alkylating agent (Formula III) by converting phenoxy alkanols into sulfonic acid esters using sulfonyl chlorides.
- Reaction of the oximido compound (Formula II) with the alkylating agent in the presence of a base such as sodium methylate or potassium hydroxide in solvents like DMF or NMP.
- Hydrolysis of the resulting oximido derivative (Formula IV) using mineral acids like hydrochloric acid to yield the final azanol ether salt or free base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The elimination of hazardous reagents like sodium hydride and butyllithium removes significant logistical burdens related to the storage and handling of pyrophoric materials, thereby reducing insurance premiums and facility compliance costs. Furthermore, the use of stable sulfonate intermediates allows for batch preparation and quality verification prior to the coupling step, decoupling the critical path of the synthesis and enhancing overall production scheduling flexibility. This modularity ensures that supply disruptions in one area do not necessarily halt the entire manufacturing campaign, providing a level of resilience that is increasingly valued in global chemical supply chains.
- Cost Reduction in Manufacturing: The process achieves significant cost savings by replacing expensive, single-use protecting groups with a catalytic or stoichiometric base system that generates benign salt byproducts. The avoidance of complex purification steps to remove phthalimide residues reduces solvent consumption and energy usage during distillation and crystallization. Additionally, the ability to recover and recycle high-boiling solvents like NMP further drives down the variable cost per kilogram, making the final azanol ether intermediate highly competitive in price-sensitive agrochemical markets.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as methanesulfonyl chloride and common alkali bases, the process mitigates the risk of supply shortages associated with specialized fluorinated or organometallic reagents. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without extensive re-validation, ensuring continuity of supply even in the face of regional disruptions. This geographic flexibility is a strategic asset for multinational corporations seeking to diversify their supplier base and minimize dependency on single-source vendors.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and atmospheric pressure, eliminating the need for specialized high-pressure reactors or cryogenic cooling systems. This simplicity facilitates rapid scale-up from pilot plant to multi-ton commercial production with minimal engineering hurdles. From an environmental perspective, the absence of heavy metal catalysts and the generation of water-soluble inorganic salts simplify wastewater treatment processes, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals without compromising output volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this azanol ether synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application requirements.
Q: What are the primary advantages of using sulfonate alkylating agents over traditional halides in this synthesis?
A: The use of sulfonic acid esters (Formula III) eliminates the need for harsh, anhydrous conditions and expensive bases like sodium hydride or butyllithium required for halide alkylation. This significantly simplifies the process safety profile and reduces raw material costs while maintaining high reactivity.
Q: How does this novel route improve impurity control compared to protecting group methods?
A: Traditional methods utilizing N-hydroxyphthalimide protecting groups often generate difficult-to-remove coupling byproducts like phthalohydrazide upon deprotection. The direct O-alkylation strategy described in CN1095708A avoids these protecting groups entirely, resulting in a cleaner crude product profile and simplified downstream purification.
Q: Is this process suitable for large-scale commercial manufacturing of agrochemical intermediates?
A: Yes, the process utilizes common industrial solvents like dimethylformamide (DMF) and N-methylpyrrolidone (NMP) and operates at moderate temperatures (60-100°C). The ability to recover and recycle solvents and the avoidance of pyrophoric reagents make it highly scalable for multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azanol Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final product formulations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering azanol ether intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to process excellence means that every batch is manufactured with a focus on consistency, safety, and regulatory compliance, giving you the confidence to integrate our materials into your most demanding synthesis campaigns.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can optimize your current supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to verify that our capabilities align perfectly with your project timelines and quality standards.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
