Advanced Boric Acid Catalysis for Commercial Scale-up of Chiral Diethyl Tartrate Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for producing high-value chiral intermediates. A pivotal advancement in this domain is documented in patent CN101100428B, which outlines a novel method for utilizing boric acid to catalyze the synthesis of chiral L- or D-diethyl tartrate. This technology represents a significant departure from traditional esterification protocols by replacing corrosive mineral acids with a mild, environmentally benign Lewis acid catalyst. For R&D directors and procurement specialists alike, this patent offers a compelling blueprint for reducing operational expenditures while maintaining the stringent purity profiles required for chiral resolution agents and asymmetric synthesis building blocks. The method not only ensures high optical purity but also introduces a closed-loop system where solvents and catalysts are recoverable, aligning perfectly with modern green chemistry mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of diethyl tartrate has relied heavily on the Fischer esterification process using strong mineral acids such as concentrated sulfuric acid or p-toluenesulfonic acid as catalysts. While effective in driving the equilibrium towards ester formation, these conventional methods suffer from severe drawbacks that impact both the bottom line and environmental compliance. The highly corrosive nature of sulfuric acid necessitates the use of expensive, specialized reactor linings and piping, leading to substantial capital expenditure (CAPEX) for plant infrastructure. Furthermore, the post-reaction workup is notoriously cumbersome; the acidic catalyst must be neutralized, generating large volumes of saline wastewater that require costly treatment before discharge. This creates a significant burden on the supply chain regarding waste management and regulatory adherence, often resulting in unpredictable lead times due to environmental inspections.
The Novel Approach
In stark contrast, the methodology described in CN101100428B utilizes boric acid ($H_3BO_3$) as a non-toxic, non-corrosive catalyst, fundamentally altering the economic and ecological footprint of the synthesis. By operating under mild conditions with boric acid, the process eliminates the need for corrosion-resistant alloy reactors, allowing for the use of standard glass-lined or even stainless steel equipment, thereby drastically reducing equipment investment. The innovation extends to the reaction medium, employing carbon tetrachloride as a solvent which facilitates an azeotropic removal of water, driving the esterification to completion without the need for excessive reagent loading. Most critically for supply chain heads, this approach enables the recycling of both the catalyst and the solvent, creating a near-zero discharge process that minimizes raw material consumption and virtually eliminates secondary pollution, ensuring a stable and compliant production continuity.
Mechanistic Insights into Boric Acid-Catalyzed Esterification
To fully appreciate the technical robustness of this synthesis, one must understand the unique mechanistic role boric acid plays compared to protonic acids. Unlike sulfuric acid which donates protons directly to the carbonyl oxygen, boric acid acts primarily as a Lewis acid. In the reaction mixture, boric acid interacts with the hydroxyl groups of both the tartaric acid and the ethanol, forming transient borate ester intermediates. This coordination activates the carboxylic acid group of the tartaric acid towards nucleophilic attack by the ethanol. The presence of carbon tetrachloride is crucial here; it forms a low-boiling azeotrope with the water produced during the esterification, allowing for continuous dehydration via a Dean-Stark trap or similar water separator. This physical removal of water shifts the chemical equilibrium strongly towards the product side according to Le Chatelier's principle, ensuring high conversion rates without requiring a massive excess of alcohol.
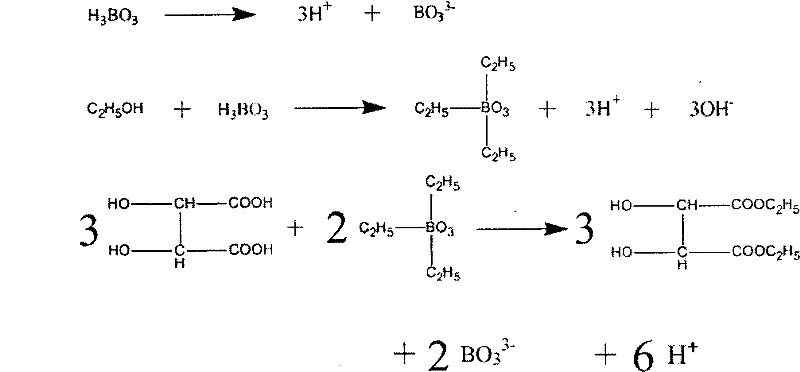
Furthermore, the mild acidity of boric acid is instrumental in preserving the stereochemical integrity of the chiral centers in tartaric acid. Strong acidic conditions at elevated temperatures can sometimes lead to racemization or degradation of sensitive chiral molecules, but the gentle catalytic environment provided by boric acid mitigates this risk. The reaction proceeds through a coordinated transition state where the boron atom stabilizes the developing negative charge on the carbonyl oxygen during the nucleophilic attack. Following the formation of the tetrahedral intermediate, the elimination of water regenerates the carbonyl group and releases the boron species, allowing it to re-enter the catalytic cycle. This mechanism explains the high esterification rate of 96.68% reported in the patent, demonstrating that high efficiency does not require harsh conditions.
How to Synthesize Chiral Diethyl Tartrate Efficiently
Implementing this synthesis on a pilot or commercial scale requires precise control over stoichiometry and thermal parameters to maximize yield and purity. The process begins with the careful mixing of chiral tartaric acid and absolute ethanol, followed by the introduction of the solvent and catalyst system. The reaction is then driven by controlled heating and continuous water removal, a critical step that dictates the final conversion. Once the reaction reaches completion, indicated by the cessation of water separation, the mixture undergoes a streamlined purification sequence involving solvent recovery and vacuum distillation. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis protocol below.
- Mix chiral L- or D-tartaric acid with absolute ethanol in a molar ratio optimized for esterification, typically around 1: 5.
- Add carbon tetrachloride as the solvent and introduce boric acid catalyst (approximately 1/15th of the tartaric acid mass) into the reaction vessel.
- Heat the mixture in an oil bath to 70°C and reflux using a water separator to remove generated water until no more water separates.
- Raise temperature to 80°C to distill off excess ethanol and solvent, then purify the crude product via vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this boric acid catalyzed route offers tangible strategic advantages beyond mere technical feasibility. The shift away from corrosive reagents translates directly into reduced maintenance schedules and extended equipment lifecycles, lowering the total cost of ownership for manufacturing facilities. Additionally, the ability to recycle the carbon tetrachloride solvent and recover the boric acid catalyst creates a circular economy within the production process, significantly insulating the operation from volatility in raw material pricing. This self-sufficiency enhances supply chain reliability, ensuring that production batches are not delayed by the sourcing of fresh catalysts or the disposal of hazardous waste.
- Cost Reduction in Manufacturing: The elimination of strong mineral acids removes the necessity for expensive neutralization agents and the associated waste treatment costs. By utilizing a non-corrosive catalyst, the process allows for the use of standard industrial reactors rather than specialized Hastelloy or glass-lined vessels, representing a substantial reduction in capital expenditure. Furthermore, the high atom economy and the recovery of valuable solvents mean that the variable cost per kilogram of the final API intermediate is significantly lower compared to traditional acid-catalyzed methods.
- Enhanced Supply Chain Reliability: The simplicity of the workup procedure—primarily involving phase separation and distillation—reduces the number of unit operations required, thereby shortening the overall batch cycle time. This efficiency allows for faster turnaround times and increased throughput capacity without the need for additional infrastructure. The robustness of the boric acid catalyst also means that the process is less sensitive to minor fluctuations in feedstock quality, ensuring consistent batch-to-batch reproducibility which is critical for maintaining long-term supply contracts with multinational pharmaceutical clients.
- Scalability and Environmental Compliance: As global regulations on industrial emissions tighten, the zero-discharge characteristic of this process becomes a major competitive advantage. The ability to recycle the solvent and catalyst internally minimizes the environmental footprint, simplifying the permitting process for new production lines. This eco-friendly profile not only reduces the risk of regulatory fines but also aligns with the sustainability goals of major downstream customers, making the manufacturer a preferred partner for green supply chain initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boric acid catalyzed synthesis. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and technical evaluators.
Q: Why is boric acid preferred over sulfuric acid for this esterification?
A: Boric acid is non-toxic and non-corrosive, significantly reducing equipment maintenance costs and eliminating the need for complex neutralization steps required with strong mineral acids.
Q: Can the solvent and catalyst be recovered in this process?
A: Yes, the patent specifies that both the carbon tetrachloride solvent and the boric acid catalyst can be recycled and reused, contributing to a zero-discharge process.
Q: What is the typical yield for this boric acid catalyzed method?
A: Under optimized conditions (70°C, 8-9 hours), the process achieves an esterification rate of approximately 96.68% with an isolated yield of around 90%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Diethyl Tartrate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN101100428B can be realized on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and GC-MS systems to guarantee that every batch of chiral diethyl tartrate meets the exacting standards required for asymmetric synthesis and chiral resolution applications.
We invite you to collaborate with us to leverage this advanced catalytic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this greener route can optimize your bill of materials. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can secure your supply chain for the future.
