Advanced Aqueous-Phase Synthesis of Polyhaloalkylaryls for High-Performance Agrochemical and Pharmaceutical Intermediates
Advanced Aqueous-Phase Synthesis of Polyhaloalkylaryls for High-Performance Agrochemical and Pharmaceutical Intermediates
The global demand for high-performance agrochemicals and pharmaceuticals continues to drive the need for sophisticated intermediate synthesis technologies that balance efficiency, cost, and environmental sustainability. Patent CN1498881A introduces a groundbreaking methodology for the preparation of polyhaloalkylaryls, a critical class of compounds known for enhancing the lipophilicity and membrane permeability of bioactive molecules. This invention specifically addresses the longstanding challenges associated with introducing perfluoroalkyl or polyhaloalkyl groups onto aromatic rings, a transformation that is essential for creating next-generation aroylurea type sterilants and other potent active ingredients. By shifting away from traditional, solvent-intensive protocols, this technology offers a pathway to more sustainable and economically viable manufacturing processes.
The core innovation lies in the utilization of a heterogeneous reaction medium comprising water and an organic phase, catalyzed by phase-transfer agents and initiated by reducing conditions or UV irradiation. This approach fundamentally alters the reaction landscape, allowing for the use of less expensive polyhaloalkyl bromides or chlorides instead of the traditionally required, costly iodides. For R&D directors and process chemists, this represents a significant opportunity to optimize synthetic routes for complex fluorinated intermediates. The ability to operate under mild conditions while achieving high yields positions this technology as a cornerstone for modern fine chemical synthesis, particularly for reliable agrochemical intermediate suppliers seeking to enhance their portfolio capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of perfluoroalkylaryl compounds has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex polymer additives and agrochemical precursors. Traditional methods often rely on aprotic polar solvents such as dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) to facilitate the reaction between aromatic substances and perfluoroalkyl halides. While these solvents can dissolve the reactants, their high boiling points make them exceptionally difficult to remove from the final product, leading to energy-intensive distillation processes and potential product contamination. Furthermore, the recovery and recycling of these solvents are often technically challenging and economically unviable, creating a substantial burden on waste management systems and increasing the overall carbon footprint of the manufacturing process.
Another critical bottleneck in conventional synthesis is the reliance on perfluoroalkyl iodides as the halogenating agent. These reagents are not only prohibitively expensive due to the high cost of iodine but also suffer from poor atom economy because of their high molecular weight. The use of iodides necessitates stringent handling procedures and often results in lower overall process efficiency. Additionally, older methodologies involving metal and sulfur dioxide or alkali-metal hyposulfite in homogeneous systems often yield only moderate productivity, requiring extensive purification steps to meet the rigorous purity specifications demanded by the pharmaceutical and agrochemical industries. These cumulative inefficiencies create a pressing need for a more robust, cost-effective, and environmentally friendly alternative.
The Novel Approach
The methodology disclosed in CN1498881A presents a paradigm shift by employing a heterogeneous reaction system that elegantly bypasses the limitations of homogeneous polar solvents. By utilizing a biphasic mixture of water and an organic solvent—such as t-butyl methyl ether, toluene, or chlorobenzene—the process ensures that the reaction occurs at the interface or within the organic phase, facilitated by a phase-transfer catalyst. This setup allows for the effective use of polyhaloalkyl bromides or chlorides, which are significantly cheaper and more atom-economical than their iodide counterparts. The reaction is driven by the presence of a reducing agent, such as sodium dithionite, or by irradiation with light of wavelengths 400nm or shorter, which generates the necessary radical species to propagate the substitution reaction on the aromatic ring.
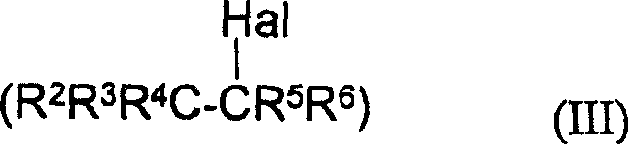
This novel approach not only simplifies the reaction workup—since water can be easily separated from the organic product phase—but also drastically reduces the thermal load required for solvent removal. The use of common, inexpensive phase-transfer catalysts like tetrabutylammonium salts further enhances the economic viability of the process. Moreover, the reaction conditions are remarkably mild, typically operating between 0°C and 70°C at atmospheric or slightly elevated pressure, which minimizes safety risks and equipment corrosion. For procurement managers, this translates to a supply chain that is less dependent on volatile, high-cost reagents and more resilient to market fluctuations in raw material pricing, ensuring a steady flow of high-purity intermediates for downstream applications.
Mechanistic Insights into Radical-Mediated Phase Transfer Catalysis
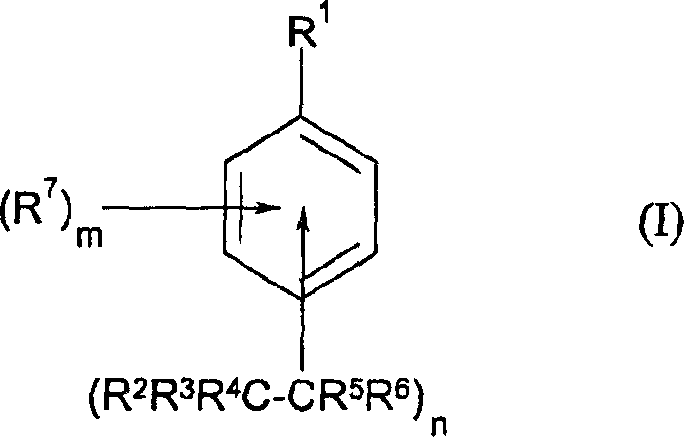
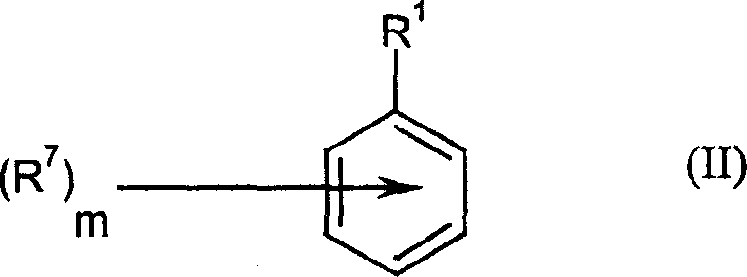
The mechanistic underpinning of this synthesis involves a complex interplay between radical chemistry and phase transfer catalysis, offering deep insights for researchers focused on optimizing reaction kinetics and impurity profiles. The process initiates with the generation of polyhaloalkyl radicals from the halide precursor (Formula III). This homolytic cleavage of the carbon-halogen bond is facilitated either by the single-electron transfer from the reducing agent (e.g., dithionite) or by direct photolysis under UV irradiation. Once formed, these highly reactive radicals attack the aromatic ring of the substrate (Formula II), leading to the formation of a cyclohexadienyl radical intermediate. The subsequent loss of a proton or hydrogen atom restores aromaticity, yielding the desired polyhaloalkylaryl product. The phase-transfer catalyst plays a pivotal role by shuttling anionic species or facilitating the interaction between the aqueous reducing agent and the organic-soluble substrates, thereby accelerating the rate of radical generation and propagation.
From an impurity control perspective, this mechanism offers distinct advantages over ionic pathways. The radical nature of the reaction tends to be highly selective for the formation of the carbon-carbon bond between the polyhaloalkyl group and the aromatic ring, minimizing side reactions such as poly-substitution or rearrangement that are common in harsher ionic conditions. The heterogeneous nature of the system also acts as a kinetic filter; by controlling the agitation speed and the concentration of the phase-transfer catalyst, chemists can fine-tune the mass transfer rates to favor the desired mono-substituted product. Furthermore, the use of water as a co-solvent helps to quench any ionic byproducts or inorganic salts, keeping them in the aqueous phase and preventing them from contaminating the organic product layer. This inherent separation capability simplifies the purification process, ensuring that the final API intermediate meets stringent quality standards with minimal downstream processing.
How to Synthesize Polyhaloalkylaryls Efficiently
The practical implementation of this synthesis route requires careful attention to the selection of solvents, catalysts, and reaction parameters to maximize yield and purity. The patent outlines a versatile protocol that can be adapted for various substrates, including anilines and other substituted aromatics, making it a valuable tool for the commercial scale-up of complex fluorinated intermediates. The key to success lies in maintaining the integrity of the biphasic system and ensuring efficient mixing to maximize the interfacial area for the phase-transfer catalysis. Detailed standard operating procedures regarding the addition rates of reagents and temperature control are critical to preventing runaway exotherms and ensuring consistent batch-to-batch reproducibility.
- Prepare a heterogeneous reaction medium containing water and an organic solvent such as t-butyl methyl ether or toluene.
- Add the aromatic substrate (Formula II), a phase-transfer catalyst (e.g., tetrabutylammonium salts), and a reducing agent like sodium dithionite.
- Introduce the polyhaloalkyl halide (Formula III) and maintain the reaction at 0-70°C under stirring, optionally utilizing UV irradiation to initiate radical formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers compelling strategic advantages that extend beyond mere technical feasibility. The shift from expensive perfluoroalkyl iodides to more abundant and cost-effective bromides or chlorides represents a direct reduction in raw material expenditure. This substitution does not compromise yield; in fact, the patent examples demonstrate high theoretical yields, such as 90% for the preparation of 2-methyl-4-(heptafluoro-2-propyl)aniline. By eliminating the need for high-boiling polar solvents like DMF and DMSO, the process also removes the associated costs of solvent recovery, waste disposal, and energy consumption for distillation. These factors combine to create a significantly leaner cost structure for the manufacturing of critical agrochemical and pharmaceutical building blocks.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the substitution of high-cost reagents with commodity chemicals. Perfluoroalkyl iodides are notoriously expensive and subject to supply volatility, whereas polyhaloalkyl bromides and chlorides are produced on a much larger industrial scale, ensuring price stability. Furthermore, the elimination of difficult-to-remove solvents reduces the operational expenditure (OPEX) associated with utility consumption and waste treatment. The use of inexpensive inorganic reducing agents like sodium dithionite and common phase-transfer catalysts further drives down the variable cost per kilogram of product. This comprehensive cost optimization strategy allows manufacturers to offer more competitive pricing for high-purity agrochemical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available raw materials and a robust reaction system. The reliance on water as a primary reaction medium reduces the dependency on specialized anhydrous solvents, which can be logistics-heavy and prone to supply disruptions. The mild reaction conditions (0-70°C) mean that the process can be run in standard glass-lined or stainless steel reactors without the need for exotic high-pressure or cryogenic equipment. This accessibility ensures that production can be scaled up rapidly to meet surging demand for active ingredients, reducing lead time for high-purity agrochemical precursors. Additionally, the simplified workup procedure shortens the overall cycle time, allowing for faster turnover and improved inventory management.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles. The avoidance of persistent organic pollutants like DMF and the reduction of halogenated waste streams simplify regulatory compliance and reduce the risk of environmental liabilities. The heterogeneous system allows for easier containment and treatment of aqueous waste, which typically contains inorganic salts that are easier to manage than complex organic solvent mixtures. The scalability of the process is proven by its operation in standard biphasic conditions, which are well-understood in chemical engineering. This makes the transition from pilot plant to multi-ton commercial production seamless, ensuring a continuous and compliant supply of vital intermediates for the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polyhaloalkylaryl synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN1498881A, providing a factual basis for decision-making. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing lines or for procurement specialists assessing the long-term viability of suppliers utilizing this method.
Q: What are the primary advantages of this aqueous-phase method over traditional DMF/DMSO processes?
A: The primary advantage is the elimination of high-boiling polar aprotic solvents like DMF and DMSO, which are difficult to remove and recover. This new method uses a water-organic biphasic system that simplifies downstream processing, reduces solvent costs, and improves environmental compliance by avoiding persistent polar waste streams.
Q: Why is the use of polyhaloalkyl bromides preferred over iodides in this synthesis?
A: Polyhaloalkyl iodides are significantly more expensive and have higher molecular weights, leading to lower atom economy. The patented process successfully utilizes cheaper and more readily available bromides or chlorides, driven by a radical mechanism facilitated by reducing agents or UV light, resulting in substantial raw material cost savings.
Q: Can this process be scaled for commercial production of agrochemical intermediates?
A: Yes, the process is highly scalable. It operates under mild temperatures (0-70°C) and atmospheric or slightly elevated pressure in a heterogeneous system. The use of common phase-transfer catalysts and inexpensive inorganic reducing agents makes it robust for large-scale manufacturing, ensuring consistent supply for agrochemical active ingredient production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyhaloalkylaryl Supplier
The technological advancements detailed in CN1498881A underscore the immense potential of aqueous-phase radical chemistry in modernizing the production of fluorinated intermediates. At NINGBO INNO PHARMCHEM, we recognize the critical importance of such innovations in delivering high-value solutions to the global agrochemical and pharmaceutical sectors. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like this are translated into reliable, industrial realities. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that guarantee every batch meets the exacting standards required for active ingredient synthesis.
We invite industry leaders to collaborate with us to leverage these advanced synthetic capabilities for their specific product pipelines. Whether you are looking to optimize an existing process or develop a new supply chain for fluorinated building blocks, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest degree of confidence and efficiency. Together, we can drive the next generation of agricultural and pharmaceutical innovation through superior chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
