Advanced Synthesis of Fluorinated Silylated Derivatives for Commercial Scale-up
The landscape of organofluorine chemistry is constantly evolving, driven by the critical need for efficient methods to introduce fluorine atoms into complex molecular architectures. Patent CN1898251A introduces a transformative approach for the production of fluorinated and silylated derivatives, specifically targeting the synthesis of compounds possessing a bond between a fluorine-carrying carbon and a silicon atom. This technology addresses the longstanding challenges associated with preparing perfluoroalkylated derivatives, which are indispensable intermediates in the development of high-performance pharmaceuticals and agrochemicals. By utilizing a novel base-catalyzed cleavage mechanism, this method circumvents the prohibitive costs and operational complexities traditionally linked to the direct synthesis of Ruppert-type reagents. The core innovation lies in the strategic use of specific precursors of general formula (I), where a fluorinated group is linked to a silyl moiety via a heteroatom bridge, allowing for controlled release of the active fluorinating species under thermal conditions.
Furthermore, the patent delineates a robust framework for generating these reactive intermediates in situ, thereby eliminating the need for isolating unstable or hazardous reagents prior to use. This capability is particularly valuable for industrial applications where safety and process efficiency are paramount. The methodology supports a wide range of fluorinated groups, including trifluoromethyl and pentafluoroethyl moieties, expanding its utility across diverse synthetic pathways. For procurement and supply chain leaders, this represents a significant opportunity to streamline the sourcing of critical fluoroalkylation reagents. Instead of relying on external suppliers for expensive, pre-formed silyl reagents, manufacturers can now produce them on-demand from readily available carboxylic acid derivatives, fundamentally altering the cost structure of fluoroalkylation processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing fluorinated silyl derivatives, such as the renowned Ruppert reagent (CF3-SiMe3), have long been plagued by significant economic and technical hurdles. The conventional synthesis often requires multi-step sequences involving expensive starting materials and harsh reaction conditions that are difficult to control on a large scale. These processes frequently suffer from low atom economy and generate substantial amounts of waste, complicating downstream purification and environmental compliance. Moreover, the isolation and storage of highly reactive silyl reagents pose safety risks due to their sensitivity to moisture and potential for decomposition. For R&D directors, these limitations translate into extended development timelines and increased uncertainty regarding the purity and consistency of the final intermediates. The reliance on specialized, high-cost reagents also creates supply chain vulnerabilities, where disruptions in the availability of a single key component can halt entire production lines.
The Novel Approach
In stark contrast, the method disclosed in CN1898251A offers a streamlined pathway that leverages inexpensive precursors and mild catalytic conditions to achieve superior results. By employing derivatives of general formula Rf-Y-O-D, where Y acts as a leaving group facilitator, the process enables the generation of the desired fluorinated silyl species through a simple thermal activation step in the presence of a base. This approach effectively bypasses the need for complex pre-functionalization, allowing manufacturers to utilize commodity chemicals like trifluoroacetic acid esters as starting points. The ability to tune the reaction by selecting specific weak bases and polar aprotic solvents provides a high degree of control over the reaction kinetics and selectivity. Consequently, this novel approach not only reduces the overall number of synthetic steps but also enhances the safety profile of the operation by minimizing the handling of isolated reactive intermediates, making it an ideal candidate for commercial scale-up of complex polymer additives and pharmaceutical intermediates.
Mechanistic Insights into Base-Catalyzed Silyl Cleavage
The mechanistic foundation of this invention rests on the delicate balance between basicity and silicophilicity of the anionic species involved in the reaction. The patent specifies the use of weak bases, particularly those with anions that exhibit low affinity for silicon, such as trifluoroacetate or halides. This is crucial because a highly silicophilic base would irreversibly bind to the silicon atom, preventing the formation of the desired carbon-silicon bond in the product. Instead, the weak base facilitates the cleavage of the oxygen-silicon bond in the precursor through a coordinated transition state, releasing the fluorinated carbanion equivalent which then attacks the silicon center. This subtle interplay ensures that the reaction proceeds with high conversion efficiency while minimizing side reactions such as desilylation or polymerization. Understanding this mechanism allows chemists to optimize the catalyst loading and solvent system to maximize the yield of the target fluorinated derivative.
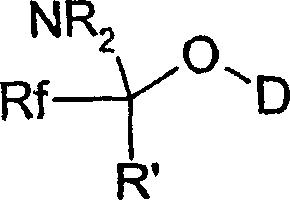
Impurity control is another critical aspect addressed by the mechanistic design of this process. The selection of the Y group in the precursor formula plays a pivotal role in determining the nature of the byproducts formed during the reaction. When Y is a carbonyl group, the resulting byproduct is typically a carboxylate salt, which is easily separable from the organic phase. Alternatively, when Y is an amino alkylidenyl group, as depicted in the structural diagram above, the reaction pathway involves the formation of stable amide byproducts that do not interfere with subsequent fluoroalkylation steps. This inherent selectivity minimizes the formation of difficult-to-remove impurities, thereby simplifying the workup procedure and reducing the burden on purification units. For quality assurance teams, this means achieving higher purity specifications with fewer processing steps, directly contributing to cost reduction in electronic chemical manufacturing and other high-value sectors.
How to Synthesize Fluorinated Silylated Derivatives Efficiently
The implementation of this synthesis route requires careful attention to reaction parameters to ensure optimal performance and safety. The process generally involves dissolving a weak base, such as potassium trifluoroacetate, in a high-boiling polar aprotic solvent like dimethylformamide (DMF) under an inert atmosphere. The silylated precursor is then introduced, and the mixture is heated to temperatures ranging from 130°C to 170°C to drive the cleavage reaction. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Prepare a reaction vessel with a polar aprotic solvent such as DMF or NMP and add a weak base like potassium trifluoroacetate.
- Introduce the silylated precursor of general formula Rf-Y-O-D into the reaction mixture under an inert atmosphere.
- Heat the mixture to temperatures between 130°C and 170°C to facilitate the cleavage of the silicon-oxygen bond and formation of the fluorinated silyl derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented technology offers profound benefits for organizations seeking to optimize their supply chains and reduce manufacturing costs. The primary advantage lies in the substitution of expensive, specialized reagents with commodity chemicals that are widely available in the global market. By shifting the synthesis of the active fluorinating agent to an in-house or near-shore operation, companies can mitigate the risks associated with long lead times and price volatility of imported fine chemicals. This strategic shift enhances supply chain reliability, ensuring consistent production schedules even in the face of global logistical disruptions. Furthermore, the simplified process flow reduces the capital expenditure required for specialized equipment, as the reaction can be conducted in standard stainless steel reactors capable of handling high-temperature organic solvents.
- Cost Reduction in Manufacturing: The elimination of multi-step synthesis sequences for the reagent itself leads to substantial cost savings. By utilizing inexpensive precursors like trifluoroacetic acid esters and simple inorganic salts as catalysts, the raw material cost per kilogram of the active fluorinating species is drastically lowered. Additionally, the high atom economy of the reaction minimizes waste disposal costs, further improving the overall economic viability of the process. This cost efficiency is particularly impactful for high-volume applications in the agrochemical and pharmaceutical sectors, where margin pressure is intense.
- Enhanced Supply Chain Reliability: Relying on a decentralized production model for critical intermediates reduces dependency on single-source suppliers. The precursors required for this method are produced by multiple chemical manufacturers worldwide, ensuring a robust and resilient supply network. This diversification allows procurement managers to negotiate better terms and secure long-term contracts with greater confidence. Moreover, the ability to generate the reagent on-demand reduces the need for large inventory holdings of hazardous materials, lowering storage costs and regulatory compliance burdens associated with dangerous goods.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in both batch and continuous flow configurations. The use of recyclable solvents and the generation of benign byproducts align with modern green chemistry principles, facilitating easier regulatory approval and environmental permitting. The high thermal stability of the reaction system allows for efficient energy integration, where waste heat can be recovered and reused within the plant. These factors collectively support the sustainable expansion of production capacity to meet growing market demand without compromising environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this fluorination technology. These answers are derived directly from the experimental data and embodiments described in the patent literature, providing a reliable basis for decision-making. Understanding these nuances is essential for engineering teams planning to integrate this chemistry into existing production lines.
Q: What is the primary advantage of using weak bases in this fluorination process?
A: Using weak bases with low silicophilicity prevents the irreversible destruction of the silyl group while still facilitating the necessary cleavage of the precursor, leading to higher selectivity and reduced side reactions compared to strong nucleophiles.
Q: Can this method be adapted for continuous flow manufacturing?
A: Yes, the patent explicitly describes embodiments involving continuous charging and distillation, indicating that the process is highly suitable for continuous flow setups to manage volatile products and improve throughput.
Q: What solvents are recommended for high-temperature silylation?
A: Polar aprotic solvents with high boiling points, such as dimethylformamide (DMF), N,N-dimethylacetamide (DMAc), or N-methyl-2-pyrrolidone (NMP), are preferred to maintain reaction stability at temperatures exceeding 130°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Silylated Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis route for the global fine chemical industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity fluorinated intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of complex precursors or large-scale supply of the final fluorinated derivatives, our infrastructure is designed to support your most demanding projects with precision and reliability.
We invite you to collaborate with us to unlock the full commercial potential of your fluorination projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower you to make informed sourcing decisions. Let us help you optimize your supply chain and accelerate your time to market with our superior manufacturing capabilities.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
