Advanced Synthesis of N-Benzoylcarbamates for Scalable Agrochemical Production
Advanced Synthesis of N-Benzoylcarbamates for Scalable Agrochemical Production
The global demand for high-performance insecticides, particularly benzoylurea derivatives like Teflubenzuron and Flufenoxuron, necessitates robust and safe supply chains for their key precursors. Patent CN1045579A introduces a transformative methodology for the preparation of N-benzoylcarbamates, which serve as critical intermediates in this value chain. This technology addresses long-standing inefficiencies in traditional synthetic routes by replacing hazardous reagents with safer, more cost-effective alternatives. By leveraging the reaction between benzamide alkali metal salts and dialkyl carbonates, manufacturers can achieve superior yields while mitigating the environmental and safety risks associated with isocyanate chemistry. For procurement leaders and R&D directors seeking a reliable agrochemical intermediate supplier, understanding this patented pathway is essential for optimizing production costs and ensuring supply continuity.
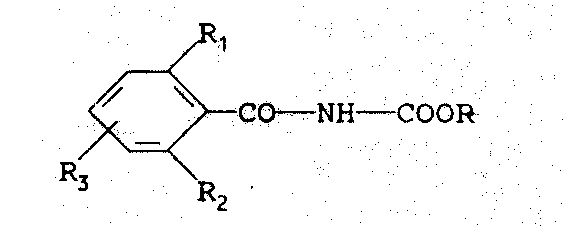
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-benzoylcarbamates has relied heavily on the reaction of benzoyl isocyanates with alcohols or the interaction of benzamides with chloroformates. These conventional pathways present significant operational challenges for large-scale commercial scale-up of complex agrochemical intermediates. Benzoyl isocyanates are notoriously unstable, moisture-sensitive, and highly toxic, requiring stringent containment measures and specialized equipment that drive up capital expenditure. Furthermore, the use of chloroformates introduces corrosive byproducts and necessitates complex waste neutralization protocols. Alternative methods involving O-ethyl carbamates often require harsh conditions, such as boiling xylene at temperatures exceeding 100°C, which can lead to thermal degradation of sensitive functional groups and lower overall process efficiency. These factors collectively contribute to higher production costs and increased regulatory scrutiny, creating bottlenecks for companies aiming for cost reduction in insecticide manufacturing.
The Novel Approach
The innovative process disclosed in the patent data offers a paradigm shift by utilizing dialkyl carbonates, such as dimethyl carbonate or diethyl carbonate, as the carbamoylating agent. This method reacts the alkali metal salt of a substituted benzamide with the carbonate in an inert solvent, eliminating the need for dangerous isocyanates entirely. The reaction proceeds smoothly at temperatures ranging from 0°C to the distillation temperature of the mixture, offering remarkable flexibility in process control. As illustrated in the reaction scheme below, this approach not only simplifies the synthetic sequence but also generates benign byproducts like alkali metal salts that are easily removed by filtration. This streamlined workflow significantly enhances the economic viability of producing high-purity N-benzoylcarbamates, making it an attractive option for modern chemical manufacturing facilities focused on sustainability and safety.
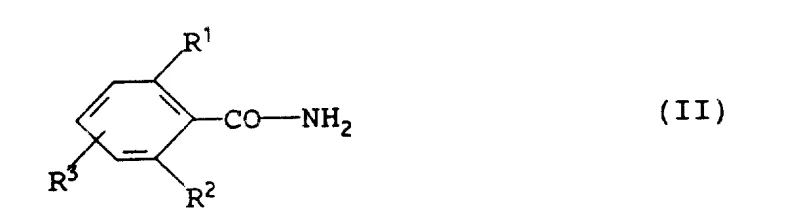
Mechanistic Insights into Alkali Metal Salt Mediated Carbamoylation
The core of this technological advancement lies in the generation and reactivity of the benzamide alkali metal salt intermediate. By treating the substituted benzamide with a strong base such as sodium hydride, potassium hydroxide, or lithium hydroxide, the amide nitrogen is deprotonated to form a highly nucleophilic anion. This anion then attacks the electrophilic carbonyl carbon of the dialkyl carbonate in a nucleophilic substitution mechanism. The choice of base and solvent plays a pivotal role in the reaction kinetics; for instance, using sodium hydride in acetone or tetrahydrofuran allows for rapid hydrogen evolution, serving as a convenient visual indicator of reaction progress. The presence of secondary alcohols like isopropanol can further facilitate the initiation of the reaction, ensuring consistent conversion rates even in less polar solvent systems. This mechanistic clarity allows process chemists to fine-tune reaction parameters for optimal throughput.
Furthermore, the isolation of the intermediate alkali metal salt of the N-benzoylcarbamate provides a unique opportunity for quality control before final acidification. The patent demonstrates that these salts, such as the sodium or potassium salts, can be isolated as stable solids with high purity, as evidenced by the spectral data provided in the examples. This stability allows for potential storage or transport of the intermediate, adding flexibility to the supply chain. Subsequent neutralization with mild acids like acetic acid or hydrochloric acid releases the free N-benzoylcarbamate in quantitative yields. This two-stage process (salt formation followed by neutralization) effectively separates the exothermic salt formation from the final product isolation, enhancing process safety and enabling better impurity profiling for reducing lead time for high-purity agrochemical intermediates.
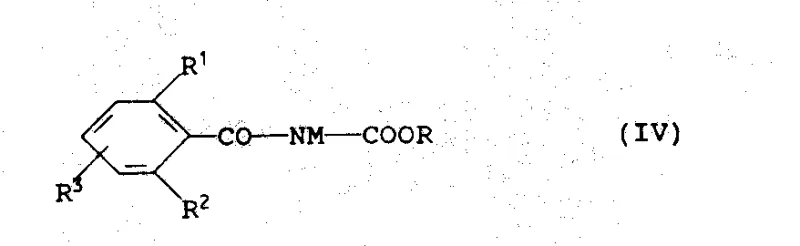
How to Synthesize N-Benzoylcarbamates Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and solvent selection to maximize yield and minimize side reactions. The process begins with the suspension of the benzamide in an inert solvent, followed by the controlled addition of the base to generate the reactive salt. Once the salt is formed, the dialkyl carbonate is introduced, and the mixture is stirred until gas evolution ceases, indicating complete consumption of the base. The detailed standardized synthesis steps, including specific molar ratios and workup procedures derived from the patent examples, are outlined below to guide your technical team in replicating this high-efficiency protocol.
- Prepare the alkali metal salt of the substituted benzamide by reacting the benzamide with a base like sodium hydride or potassium hydroxide in an inert solvent.
- React the resulting benzamide salt with a dialkyl carbonate (e.g., dimethyl carbonate) at temperatures ranging from 0°C to the solvent's distillation temperature.
- Neutralize the resulting alkali metal salt of the carbamate with an acid such as acetic acid or hydrochloric acid to isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this carbonate-based synthesis route offers substantial strategic benefits beyond mere technical feasibility. The elimination of benzoyl isocyanates and chloroformates removes a major category of hazardous raw materials from the procurement list, thereby simplifying vendor qualification and reducing the costs associated with hazardous material handling and storage. The use of commodity chemicals like dimethyl carbonate and common solvents such as acetone or toluene ensures a stable and competitive pricing structure for raw materials, shielding the production budget from the volatility often seen with specialty reagents. Additionally, the mild reaction conditions reduce energy consumption compared to high-temperature reflux processes, contributing to lower utility costs and a smaller carbon footprint for the manufacturing site.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous isocyanates with inexpensive dialkyl carbonates directly lowers the bill of materials. Furthermore, the simplified workup procedure, which often involves simple filtration of inorganic salts followed by solvent evaporation, reduces the need for complex purification columns or extensive washing steps. This operational simplicity translates into reduced labor hours and lower waste disposal costs, as the byproducts are primarily non-toxic inorganic salts. The high crude yields reported in the patent examples suggest that material loss is minimized, further enhancing the overall cost-effectiveness of the process for large-volume production runs.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials like substituted benzamides and dialkyl carbonates mitigates the risk of supply disruptions caused by the limited availability of specialty isocyanates. The robustness of the reaction across various solvents (acetone, ethyl acetate, THF, toluene) provides manufacturing flexibility, allowing production to continue even if specific solvent supplies are constrained. This adaptability ensures a consistent flow of intermediates to downstream customers, reinforcing the reliability of the supply chain for critical agrochemical active ingredients.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of reactions on multi-gram scales in the patent examples without loss of efficiency. The absence of toxic isocyanates simplifies environmental compliance and permitting, as the facility does not need to maintain strict controls for airborne toxicants associated with isocyanate handling. The generation of solid inorganic waste that can be easily filtered and disposed of aligns with modern green chemistry principles, facilitating easier regulatory approval and community acceptance of the manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-benzoylcarbamate synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of adopting this route for their specific production needs and ensures alignment with quality and safety standards.
Q: What are the safety advantages of this new carbamate synthesis route?
A: Unlike conventional methods that utilize toxic benzoyl isocyanates or corrosive chloroformates, this process employs stable dialkyl carbonates and benzamide salts, significantly reducing handling risks and waste treatment costs.
Q: Can this process be scaled for commercial insecticide production?
A: Yes, the reaction operates under mild conditions (0°C to reflux) using common industrial solvents like acetone or toluene, making it highly suitable for large-scale commercial manufacturing of intermediates like Teflubenzuron.
Q: What purity levels can be achieved with this method?
A: Experimental data indicates high crude yields (up to 97%) with purities exceeding 97% after simple purification steps, ensuring consistent quality for downstream pharmaceutical or agrochemical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Benzoylcarbamates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the production of high-value agrochemical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1045579A are fully realized in practical manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-benzoylcarbamates meets the exacting standards required for downstream insecticide synthesis. Our commitment to process optimization allows us to deliver consistent quality while adhering to the highest safety and environmental protocols.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our optimized manufacturing capabilities can support your long-term production goals and enhance your competitive position in the global agrochemical market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
