Advanced Organocatalytic Synthesis of Complex Pharmaceutical Intermediates via Novel Sugar-Phosphine Thiourea Systems
The landscape of asymmetric synthesis for high-value pharmaceutical intermediates is undergoing a significant transformation driven by the need for greener, more efficient, and highly selective catalytic processes. Patent CN102120749A introduces a groundbreaking class of sugar-containing chiral phosphine thiourea compounds that address critical limitations in the construction of complex chiral scaffolds. This technology specifically targets the intramolecular asymmetric Baylis-Hillman reaction, a powerful tool for generating densely functionalized cyclic structures found in numerous bioactive molecules. By leveraging the unique stereoelectronic properties of carbohydrate backbones combined with bifunctional phosphine-thiourea motifs, this invention achieves exceptional enantioselectivity of up to 99% ee. For R&D directors and process chemists, this represents a paradigm shift away from traditional metal-based or low-selectivity organocatalytic methods, offering a robust pathway to access high-purity chiral building blocks essential for modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the Baylis-Hillman reaction has been a cornerstone of carbon-carbon bond formation, yet its asymmetric variant, particularly in intramolecular settings, has faced persistent challenges regarding reaction rate and stereocontrol. Conventional approaches often rely on L-proline or its simple derivatives as organocatalysts. While these amino acid-based catalysts are inexpensive and readily available, they frequently suffer from modest enantioselectivity when applied to complex intramolecular cyclizations, often failing to exceed 90% ee in difficult substrate classes. Furthermore, alternative strategies employing transition metal complexes, while sometimes effective, introduce significant supply chain and regulatory burdens due to the strict limits on residual heavy metals in active pharmaceutical ingredients (APIs). The removal of these metal traces requires additional purification steps, such as scavenging or recrystallization, which drastically reduce overall yield and increase manufacturing costs. Additionally, many traditional catalysts require harsh conditions or extended reaction times, leading to potential decomposition of sensitive functional groups and the formation of difficult-to-remove impurities that complicate the final drug substance quality profile.
The Novel Approach
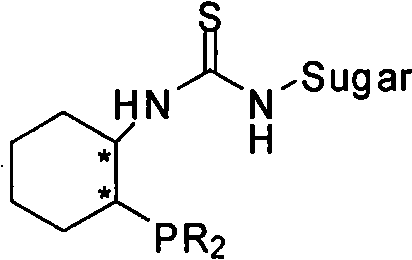
The innovation disclosed in CN102120749A overcomes these hurdles through the rational design of a bifunctional organocatalyst that integrates a chiral sugar moiety with a phosphine-thiourea framework. This novel architecture allows for simultaneous activation of both the nucleophile and the electrophile within the substrate, creating a highly organized transition state that dictates stereochemical outcome with remarkable precision. Unlike the planar or less rigid structures of proline derivatives, the bulky and rigid carbohydrate scaffold provides a well-defined chiral pocket that effectively shields one face of the reacting species, thereby enforcing high facial selectivity. The phosphine group acts as a Lewis base to activate the electron-deficient alkene, while the thiourea moiety engages the carbonyl group through dual hydrogen bonding. This synergistic activation not only accelerates the reaction kinetics, allowing it to proceed efficiently at room temperature, but also ensures that the newly formed chiral center is established with near-perfect fidelity. As demonstrated in the patent data, this approach consistently delivers products with enantiomeric excess values reaching 99% ee, setting a new benchmark for metal-free asymmetric synthesis in this domain.

Mechanistic Insights into Sugar-Phosphine Thiourea Catalysis
The exceptional performance of these catalysts stems from their sophisticated bifunctional mechanism, which mimics the efficiency of enzymatic active sites. The core of the catalytic cycle involves the nucleophilic attack of the phosphine atom on the activated alkene of the substrate, generating a zwitterionic enolate intermediate. Simultaneously, the thiourea functionality, positioned in close proximity due to the rigid cyclohexyl and sugar linkers, forms strong hydrogen bonds with the carbonyl oxygen of the electrophilic aldehyde or ketone moiety. This dual activation brings the reactive centers into close spatial alignment while locking the substrate into a specific conformation dictated by the chiral information encoded in the sugar unit. The steric bulk of the acetyl-protected sugar groups further restricts the rotational freedom of the intermediate, preventing non-productive pathways and minimizing the formation of racemic byproducts. This precise control over the transition state geometry is what enables the system to differentiate between enantiotopic faces with such high accuracy, resulting in the observed superior enantioselectivity compared to simpler monofunctional catalysts.
From an impurity control perspective, this mechanistic pathway offers distinct advantages for process chemistry. Because the reaction proceeds through a well-defined organocatalytic cycle without the involvement of radical species or unstable metal intermediates, the generation of side products is inherently minimized. The absence of transition metals eliminates the risk of metal-induced oxidation or coupling side reactions that often plague traditional methods. Furthermore, the mild reaction conditions (typically 25°C in tert-butanol) prevent thermal degradation of the substrate or product, ensuring a cleaner crude reaction profile. This simplifies the downstream purification process, often allowing for direct crystallization or simple chromatographic separation to achieve pharmaceutical-grade purity. For quality assurance teams, this translates to a more predictable and robust impurity profile, reducing the analytical burden required to characterize and quantify trace degradants in the final API intermediate.
How to Synthesize Sugar-Containing Chiral Phosphine Thiourea Efficiently
The preparation of these high-performance catalysts is designed for operational simplicity and scalability, utilizing readily available chiral starting materials. The synthesis relies on a straightforward condensation reaction between a single-configuration chiral aminophosphine and a protected glycosyl isothiocyanate. This convergent strategy allows for the modular assembly of the catalyst library by varying either the phosphine backbone or the sugar component to fine-tune steric and electronic properties for specific substrates. The reaction conditions are mild, typically proceeding in common organic solvents like dichloromethane at room temperature, which minimizes energy consumption and safety risks associated with high-pressure or high-temperature operations. The detailed standardized synthesis protocol below outlines the critical parameters for achieving high yields and purity, ensuring reproducibility for industrial applications.
- Dissolve single-configuration (R,R)- or (S,S)-1-amino-2-(diarylphosphine)cyclohexane in an organic solvent such as dichloromethane at room temperature.
- Add glycosyl isothiocyanate (Sugar-NCS) dropwise to the solution, maintaining a molar ratio between 1: 2 and 1:4, and stir for 12 to 24 hours.
- Purify the resulting crude mixture using silica gel column chromatography to isolate the high-purity sugar-containing chiral phosphinethiourea compound.
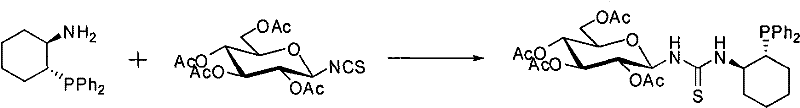
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this organocatalytic technology presents a compelling value proposition centered on cost efficiency, regulatory compliance, and supply security. The shift from metal-based catalysis to metal-free organocatalysis fundamentally alters the cost structure of manufacturing complex chiral intermediates. By eliminating the need for expensive transition metal precursors and the associated downstream metal scavenging resins or specialized filtration equipment, manufacturers can realize substantial cost reductions in raw material and processing expenses. Furthermore, the simplified purification workflow reduces solvent consumption and waste generation, aligning with increasingly stringent environmental regulations and sustainability goals. This streamlined process not only lowers the direct cost of goods sold (COGS) but also shortens the overall production cycle time, enhancing the agility of the supply chain to respond to market demands.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a significant variable cost component and mitigates the financial risk associated with volatile metal prices. Additionally, the high selectivity of the catalyst reduces the formation of diastereomeric impurities, which often require costly and yield-loss-inducing recrystallization steps to remove. This improvement in first-pass yield directly contributes to a more economical manufacturing process, allowing for competitive pricing of the final pharmaceutical intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: The starting materials for this catalyst, such as chiral cyclohexyl amines and protected sugars, are derived from abundant renewable feedstocks or established commodity chemical streams. This contrasts sharply with supply chains dependent on scarce platinum group metals, which are subject to geopolitical instability and mining constraints. By diversifying the raw material base towards bio-derived and synthetic organic components, manufacturers can secure a more stable and resilient supply chain, reducing the risk of production disruptions due to raw material shortages.
- Scalability and Environmental Compliance: The reaction operates under mild conditions using benign solvents like tert-butanol, which facilitates safe scale-up from laboratory to commercial production volumes. The absence of toxic heavy metals simplifies waste treatment protocols and reduces the environmental footprint of the manufacturing facility. This ease of compliance with environmental, health, and safety (EHS) regulations accelerates the regulatory approval process for new drug filings, as the impurity profile is cleaner and easier to justify to health authorities, ultimately speeding up time-to-market for new therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic technology in pharmaceutical manufacturing. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is critical for making informed decisions about process development and vendor selection.
Q: What is the primary advantage of this sugar-containing catalyst over traditional L-proline catalysts?
A: The novel sugar-containing chiral phosphine thiourea catalysts described in CN102120749A achieve significantly higher enantioselectivity, reaching up to 99% ee in intramolecular Baylis-Hillman reactions, whereas traditional L-proline systems often yield lower selectivity for these specific transformations.
Q: Are there heavy metal residues associated with this catalytic process?
A: No, this process utilizes purely organic organocatalysts. Unlike transition metal-catalyzed routes, this method eliminates the risk of heavy metal contamination, simplifying downstream purification and ensuring compliance with stringent pharmaceutical impurity guidelines.
Q: What are the typical reaction conditions for the intramolecular Baylis-Hillman reaction using this catalyst?
A: The reaction proceeds under mild conditions, typically at 25°C in tert-butanol (t-BuOH) with a low catalyst loading of approximately 2 mol%, demonstrating high efficiency and operational simplicity suitable for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sugar-Containing Chiral Phosphine Thiourea Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in accelerating drug development timelines. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like the sugar-phosphine thiourea system can be seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral intermediate delivered meets the highest global regulatory standards. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our dedicated technical team is committed to supporting your projects from early-stage route scouting through to commercial launch.
We invite you to explore how this cutting-edge catalytic technology can optimize your synthesis of complex chiral intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can drive efficiency and reliability in your supply chain. Let us partner with you to turn challenging synthetic problems into commercial successes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
