Optimizing 2,3-Dichloropyridine Production for Global Pharmaceutical and Agrochemical Supply Chains
Optimizing 2,3-Dichloropyridine Production for Global Pharmaceutical and Agrochemical Supply Chains
The global demand for high-purity heterocyclic intermediates continues to surge, driven by the expanding markets for novel pharmaceuticals and advanced crop protection agents. Among these critical building blocks, 2,3-dichloropyridine stands out as a pivotal precursor, particularly for the synthesis of diamide insecticides such as chlorantraniliprole. A significant technological breakthrough in the manufacturing of this compound is detailed in patent CN103570609A, which outlines a refined synthetic pathway designed to overcome the limitations of traditional methods. This innovation focuses on maximizing yield and purity while minimizing environmental impact through a streamlined sequence involving Hofmann degradation, selective chlorination, and a concurrent diazotization-Sandmeyer reaction. For procurement leaders and technical directors seeking a reliable pharma intermediates supplier, understanding the nuances of this improved process is essential for securing a stable and cost-effective supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2,3-dichloropyridine has relied on a multi-step sequence that presents significant engineering and safety challenges. The traditional route typically initiates with the Hofmann degradation of nicotinamide to yield 3-aminopyridine, followed by an oxychlorination step that often utilizes elemental chlorine gas. This reliance on chlorine gas introduces severe safety hazards, requiring specialized containment infrastructure and rigorous monitoring to prevent leaks and exposure. Furthermore, the conventional oxychlorination process is prone to generating multiple isomeric by-products, such as 2,6-dichloropyridine, which complicates downstream purification and lowers the overall yield of the desired 2,3-isomer. The subsequent steps involving separate diazotization and Sandmeyer reactions add further complexity, necessitating the isolation and drying of unstable diazonium intermediates, which increases the risk of thermal runaway and reduces process efficiency.
The Novel Approach
In contrast, the methodology disclosed in the referenced patent introduces a series of strategic modifications that fundamentally enhance the reaction profile. Instead of using hazardous chlorine gas for the chlorination step, the new process employs hydrogen peroxide in a hydrochloric acid medium to effect the transformation of 3-aminopyridine to 2-chloro-3-aminopyridine. This shift not only mitigates safety risks but also improves regioselectivity, ensuring that the chlorine atom is introduced predominantly at the 2-position. Additionally, the process innovates by combining the diazotization and Sandmeyer reaction into a single operational unit. By adding sodium nitrite directly to the reaction mixture containing cuprous chloride, the unstable diazonium salt is consumed immediately as it forms, thereby preventing decomposition and side reactions. This telescoped approach eliminates the need for intermediate isolation, resulting in a simpler operation that is highly amenable to large-scale commercial production.
Mechanistic Insights into the Modified Synthetic Pathway
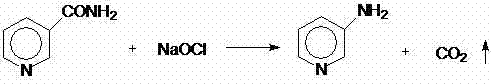
The core of this synthesis begins with the Hofmann degradation, where nicotinamide reacts with sodium hypochlorite under alkaline conditions. As illustrated in the reaction scheme, the amide nitrogen is excised as carbon dioxide, leaving behind the primary amine functionality on the pyridine ring. The patent specifies maintaining a temperature range of 0-5°C during the addition of sodium hypochlorite to control the exothermic nature of the reaction and prevent over-oxidation. Following this, the 3-aminopyridine is extracted and converted to its hydrochloride salt, which serves as the substrate for the subsequent chlorination. The use of hydrogen peroxide as the oxidant in the presence of hydrochloric acid generates reactive chlorine species in situ, which act as electrophiles to attack the electron-rich pyridine ring. This mechanism avoids the harsh conditions associated with gaseous chlorine, leading to a cleaner reaction profile with fewer poly-chlorinated impurities.
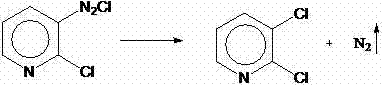
The final transformation involves the conversion of the amino group to a chloro substituent via a diazonium intermediate. In traditional protocols, this requires low temperatures and careful handling of the isolated diazonium salt. However, the improved method performs this step concurrently with the Sandmeyer substitution. Upon the addition of sodium nitrite to the acidic solution containing 2-chloro-3-aminopyridine and cuprous chloride, the diazonium ion is generated and immediately intercepted by the copper catalyst. This rapid succession prevents the accumulation of the energetic diazonium species, significantly enhancing process safety. The evolution of nitrogen gas drives the reaction forward, and the resulting 2,3-dichloropyridine is recovered through extraction and distillation. This mechanistic efficiency is crucial for achieving the reported molar yields of over 60% from the starting nicotinamide, a substantial improvement over older techniques.
How to Synthesize 2,3-Dichloropyridine Efficiently
Implementing this synthesis route requires precise control over reaction parameters to ensure optimal yield and safety. The process begins with the careful preparation of the 3-aminopyridine intermediate, followed by the critical chlorination step where temperature and oxidant addition rates must be strictly monitored. The final stage involves the simultaneous addition of nitrite and copper catalyst to effect the substitution. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below which outlines the specific conditions validated in the patent examples.
- Perform Hofmann degradation on nicotinamide using sodium hypochlorite at 0-5°C to generate 3-aminopyridine with high recovery.
- Execute electrophilic chlorination of 3-aminopyridine using hydrogen peroxide in hydrochloric acid solution to form 2-chloro-3-aminopyridine.
- Conduct simultaneous diazotization and Sandmeyer reaction using sodium nitrite and cuprous chloride to finalize the 2,3-dichloropyridine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refined manufacturing process offers tangible benefits that extend beyond mere chemical yield. The elimination of hazardous chlorine gas handling reduces the regulatory burden and insurance costs associated with production facilities, translating into a more resilient supply base. Furthermore, the ability to skip the purification of the 3-aminopyridine intermediate significantly cuts down on solvent usage and waste generation, aligning with modern sustainability goals and reducing disposal costs. These operational efficiencies contribute to a more stable pricing structure and reliable delivery schedules for downstream customers seeking cost reduction in pharma intermediates manufacturing.
- Cost Reduction in Manufacturing: The process design inherently lowers production costs by removing unit operations. By avoiding the isolation and refining of the 3-aminopyridine intermediate, the manufacturer saves on energy consumption for distillation and reduces the volume of organic solvents required for extraction. Additionally, the substitution of chlorine gas with hydrogen peroxide simplifies the reactor setup, removing the need for specialized corrosion-resistant alloys and gas scrubbing systems. These factors collectively drive down the capital expenditure and operating expenses, allowing for a more competitive market price for the final 2,3-dichloropyridine product without compromising on quality standards.
- Enhanced Supply Chain Reliability: Safety is a cornerstone of supply chain continuity. By mitigating the risks associated with handling toxic gases and unstable intermediates, the likelihood of unplanned shutdowns due to safety incidents is drastically reduced. The robustness of the simultaneous diazotization-Sandmeyer step ensures consistent batch-to-batch quality, minimizing the rejection rates that can disrupt inventory levels. For buyers, this means a dependable source of high-purity 2,3-dichloropyridine that can support long-term production planning for critical drug substances and agrochemical formulations, ensuring that project timelines are met without delay.
- Scalability and Environmental Compliance: The simplified workflow is inherently easier to scale from pilot plant to commercial tonnage. The reduction in waste streams, particularly the avoidance of heavy metal contamination from excessive copper usage and the minimization of chlorinated organic by-products, facilitates easier compliance with environmental regulations. The process generates less hazardous waste, lowering the cost of treatment and disposal. This environmental stewardship is increasingly important for multinational corporations aiming to reduce their carbon footprint and adhere to strict green chemistry principles, making this supply route a preferred choice for sustainable commercial scale-up of complex agrochemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,3-dichloropyridine. These answers are derived from the technical specifications and beneficial effects described in the underlying patent literature, providing clarity on the process capabilities and quality assurances available to potential partners.
Q: How does the new process improve safety compared to traditional chlorination methods?
A: The novel methodology replaces hazardous chlorine gas oxychlorination with a controlled hydrogen peroxide oxidation system in acidic media, significantly reducing operational risks and equipment corrosion while maintaining high regioselectivity for the 2-position.
Q: What are the purity specifications achievable with this synthesis route?
A: By eliminating intermediate refining steps and utilizing a simultaneous diazotization-Sandmeyer protocol, the process consistently achieves product content exceeding 98.0% as verified by liquid chromatography external standard methods.
Q: Does this method require purification of the 3-aminopyridine intermediate?
A: No, a key advantage of this technology is the ability to use the crude 3-aminopyridine hydrochloride solution directly in the subsequent chlorination step, which drastically reduces solvent waste and energy consumption associated with isolation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dichloropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patents like CN103570609A is translated into robust industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,3-dichloropyridine meets the exacting standards required by the global pharmaceutical and agrochemical industries. Our commitment to technical excellence ensures that you receive a product that facilitates efficient downstream synthesis without unexpected impurities.
We invite you to collaborate with us to optimize your supply chain for this essential building block. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project milestones. Let us be your partner in driving efficiency and quality in your manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
