Advanced Chiral Resolution Strategies for High-Purity Antihistamine Intermediates
Introduction to Advanced Chiral Separation Technologies
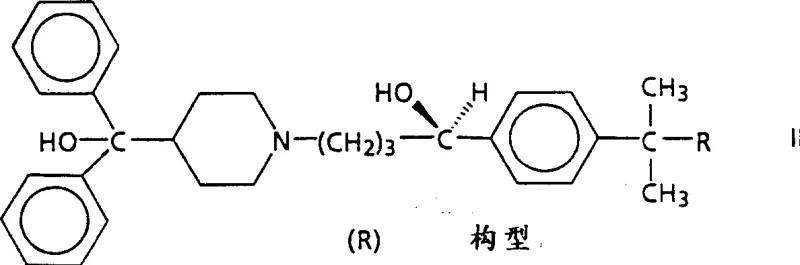
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients (APIs) and their precursors, particularly for chiral molecules where biological activity is strictly dependent on stereochemistry. Patent CN1309121A presents a significant technological breakthrough in the optical resolution of racemic α-[4-(1,1-dimethylethyl)phenyl]-4-(hydroxydiphenylmethyl)-1-piperidinebutanol, commonly known in the industry as RMI 9918. This compound and its derivatives serve as critical intermediates for potent antihistaminic and anti-allergic agents. The disclosed methodology addresses the longstanding economic and technical challenges associated with separating enantiomers on an industrial scale, moving away from labor-intensive chromatographic methods toward efficient crystallization-based techniques. By leveraging specific chiral resolving agents such as di-p-toluoyltartaric acid (DPTTA) and mandelic acid, this innovation enables manufacturers to achieve exceptional optical purity with streamlined processing steps. For global procurement teams and R&D directors, understanding this resolution pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality materials consistently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the resolution of complex piperidine derivatives like RMI 9918 relied heavily on the use of expensive and cumbersome resolving agents such as (-)-dinaphthyl phosphoric acid, as described in earlier United States patents. These traditional processes were plagued by inefficiencies, often requiring excessive recrystallization cycles—sometimes up to seven distinct crystallization steps—to achieve acceptable enantiomeric excess. Such repetitive processing not only drastically reduced the overall chemical yield, often dropping below 25%, but also increased the consumption of solvents and energy, thereby inflating production costs. Furthermore, the physical properties of the diastereomeric salts formed with dinaphthyl phosphoric acid often resulted in poor filtration characteristics and difficult handling during scale-up. These operational bottlenecks made the conventional approach less viable for modern cost reduction in API manufacturing, where speed and efficiency are paramount competitive advantages.
The Novel Approach
The innovative method detailed in the patent data introduces a robust alternative utilizing (+)-di-p-toluoyltartaric acid (DPTTA) or (-)-mandelic acid as the primary chiral selectors. This novel approach capitalizes on the distinct solubility differences between diastereomeric salts in common organic solvents like acetone and methanol. By optimizing the stoichiometry and temperature profiles, the process allows for the selective precipitation of the target enantiomer with high diastereomeric purity in the initial crystallization step. For instance, experimental data demonstrates that using DPTTA in acetone can yield diastereomeric salts with over 90% de initially, which can be upgraded to nearly 100% ee with just a single recrystallization. This drastic reduction in processing complexity translates directly into higher throughput and lower operational expenditure. The ability to recover the resolving agent and recycle the mother liquor further enhances the sustainability and economic feasibility of this route for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Diastereomeric Salt Crystallization
The core mechanism driving this resolution process is the formation of diastereomeric salts through acid-base interactions between the basic racemic amine and the acidic chiral resolving agent. When the racemic mixture interacts with a single enantiomer of the resolving agent, two distinct diastereomeric salts are formed: one comprising the (+)-enantiomer of the substrate and the (+)-resolving agent, and the other comprising the (-)-enantiomer and the (+)-resolving agent. Although these salts share similar chemical connectivity, their three-dimensional spatial arrangements differ, leading to unique physical properties such as solubility, lattice energy, and crystal habit. In the specific case of RMI 9918 resolution with DPTTA, the salt formed between the target (+)-(R)-enantiomer and the (+)-DPTTA exhibits significantly lower solubility in acetone at reduced temperatures compared to the mismatched pair. This thermodynamic preference drives the selective nucleation and growth of the desired salt crystals while leaving the unwanted isomer in the solution phase.
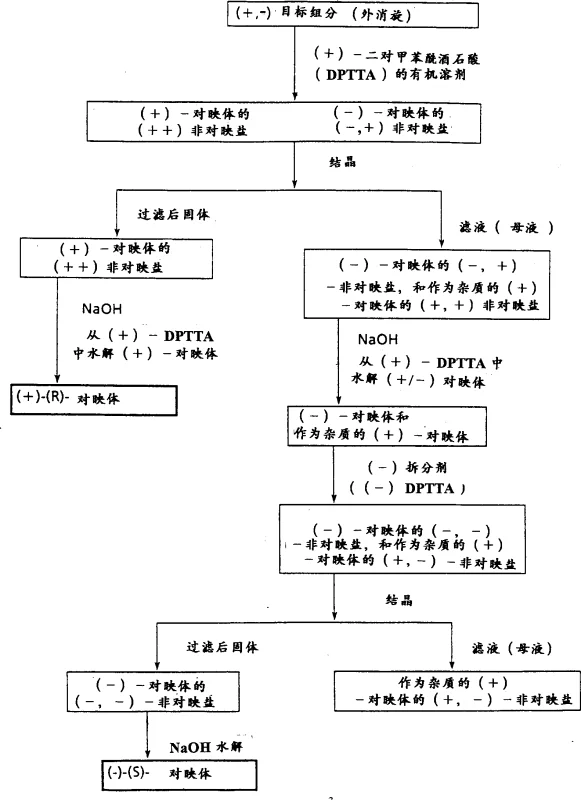
Following the crystallization, the isolation of the pure enantiomer is achieved through a hydrolysis step, typically employing aqueous sodium hydroxide. This step breaks the ionic bond between the chiral acid and the amine, liberating the free base form of the target molecule. The patent data highlights that this hydrolysis can be performed under mild conditions, preserving the integrity of the sensitive hydroxyl and piperidine functionalities within the molecule. Crucially, the process allows for the recovery of the resolving agent from the aqueous phase, which can be regenerated and reused, minimizing waste generation. The impurity profile is tightly controlled throughout this sequence; by carefully managing the cooling rates and solvent ratios, the inclusion of the undesired enantiomer within the crystal lattice is minimized. This mechanistic precision ensures that the final product meets stringent purity specifications required for downstream pharmaceutical synthesis without the need for extensive chromatographic purification.
How to Synthesize RMI 9918 Efficiently
To implement this resolution strategy effectively, manufacturers must adhere to precise operational parameters regarding solvent selection, temperature control, and molar ratios. The process begins with dissolving the racemic substrate and the chiral acid in a polar organic solvent, followed by a controlled heating phase to ensure homogeneity. Subsequent cooling induces supersaturation, triggering the selective crystallization of the target diastereomer. The solid is then filtered, washed, and subjected to base hydrolysis to release the optically pure amine. Detailed standardized synthetic steps see the guide below.
- Dissolve the racemic compound and an equimolar amount of chiral resolving agent (such as (+)-DPTTA) in a suitable organic solvent like acetone.
- Heat the solution to approximately 50-55°C to ensure complete dissolution and formation of soluble diastereomeric salts.
- Cool the solution slowly to room temperature and then further to 4°C to induce selective crystallization of the target diastereomeric salt.
- Filter the precipitated crystals and subject them to hydrolysis using sodium hydroxide to liberate the optically pure free base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resolution technology offers transformative benefits in terms of cost stability and supply reliability. The shift from multi-step recrystallization protocols to a streamlined one or two-step crystallization process significantly reduces the manufacturing cycle time. This acceleration allows for faster response to market demand fluctuations and shorter lead times for high-purity pharmaceutical intermediates. Moreover, the replacement of exotic resolving agents with commercially available and cost-effective acids like DPTTA and mandelic acid mitigates the risk of raw material shortages. The use of common solvents such as acetone and methanol further simplifies the supply chain logistics, as these materials are readily available in bulk quantities globally, reducing the dependency on specialized chemical vendors.
- Cost Reduction in Manufacturing: The elimination of excessive recrystallization steps directly correlates to substantial savings in utility costs, solvent consumption, and labor hours. By achieving high optical purity early in the process, the need for resource-intensive purification techniques is removed, leading to a leaner production model. Additionally, the high recovery yield of the resolving agent means that the effective cost per kilogram of the chiral acid is drastically lowered over multiple batches. This efficiency creates a more competitive pricing structure for the final intermediate, allowing partners to optimize their overall cost reduction in API manufacturing budgets without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of this crystallization-based method ensures consistent batch-to-batch reproducibility, which is critical for maintaining uninterrupted supply lines. Unlike biochemical or enzymatic methods that may suffer from variability due to catalyst sensitivity, this chemical resolution relies on stable physical properties that are easier to control at scale. The ability to process large volumes in standard reactor equipment enhances the scalability of the operation, ensuring that suppliers can meet large-scale orders reliably. This stability reduces the risk of production delays and provides a secure foundation for long-term procurement contracts.
- Scalability and Environmental Compliance: From an environmental perspective, the process generates less waste due to higher yields and solvent recyclability. The reduced solvent usage per unit of product lowers the burden on waste treatment facilities and aligns with increasingly strict environmental regulations. The simplicity of the unit operations—dissolution, crystallization, filtration, and hydrolysis—facilitates easy scale-up from pilot plants to multi-ton commercial production facilities. This scalability ensures that the technology remains viable and efficient regardless of the production volume, supporting sustainable growth in the specialty chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this resolution process. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on solvent choices, resolving agent efficacy, and purity outcomes. Understanding these nuances is vital for technical teams evaluating the feasibility of this route for their specific manufacturing contexts.
Q: What are the advantages of using DPTTA over dinaphthyl phosphoric acid for RMI 9918 resolution?
A: According to patent CN1309121A, using (+)-di-p-toluoyltartaric acid (DPTTA) significantly reduces the number of recrystallization steps required compared to the traditional dinaphthyl phosphoric acid method. While the older method required up to seven recrystallizations to achieve high purity, the DPTTA method achieves comparable or superior optical purity (up to 100% ee) with only one or two recrystallizations, leading to substantially higher overall chemical yields.
Q: Which solvents are most effective for this chiral resolution process?
A: The patent data indicates that acetone is the preferred solvent when using DPTTA as the resolving agent, providing excellent solubility differentials between the diastereomeric salts at varying temperatures. For resolutions involving mandelic acid, methanol or ethanol serves as an effective medium. The choice of solvent is critical for maximizing the diastereomeric excess during the initial crystallization phase.
Q: Can both (R) and (S) enantiomers be recovered from this process?
A: Yes, the process is designed to recover both enantiomers efficiently. After the primary target enantiomer is crystallized and filtered, the mother liquor retains the opposite enantiomer. This remaining material can be treated with the opposite enantiomer of the resolving agent or recycled to isolate the second enantiomer, ensuring minimal waste and maximizing atom economy in the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable RMI 9918 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation antihistamines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated resolution techniques described in patent CN1309121A are executed with precision. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to verify enantiomeric excess at every stage of production. Our commitment to quality assurance guarantees that every batch of RMI 9918 delivered meets the exacting standards required by global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your supply chain goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient resolution strategies can lower your total cost of ownership. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless integration of our high-quality intermediates into your pharmaceutical pipeline.
