Advanced Manufacturing of COX/LOX Inhibitor Intermediates via Optimized Hydrogenation and Cyclization
The pharmaceutical landscape for dual cyclo-oxygenase and 5-lipoxygenase inhibitors has been significantly advanced by the development of efficient synthetic routes for key intermediates like 6-(4-chlorophenyl)-2,2-dimethyl-7-phenyl-2,3-dihydro-1H-pyrrolizin-5-ylacetic acid, commonly referred to as ML3000. Patent CN1545516A discloses a groundbreaking methodology that fundamentally restructures the synthesis of this potent anti-inflammatory agent, moving away from hazardous and operationally complex Grignard reactions towards a more robust catalytic hydrogenation strategy. This technological shift addresses critical pain points in process chemistry, including equipment corrosion, cryogenic energy demands, and difficult purification protocols associated with prior art methods. By leveraging specific catalytic conditions and optimized intermediate handling, the disclosed process achieves superior purity profiles and operational safety, positioning it as a preferred pathway for the reliable pharmaceutical intermediates supplier seeking to modernize their production capabilities. The core innovation lies in the direct conversion of a nitrile-ketone precursor into the pyrroline ring system, streamlining the overall synthetic sequence.
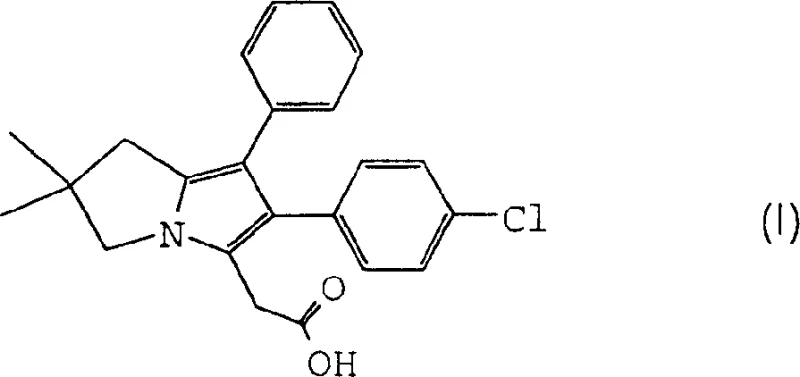
Historically, the construction of the pyrrolizine core for ML3000 relied heavily on organometallic chemistry that posed significant challenges for industrial scale-up. Conventional methods, as illustrated in the prior art reaction schemes, typically necessitated the use of benzylmagnesium chloride in a Grignard reaction to install the benzyl group, requiring stringent temperature control often reaching cryogenic levels of -78°C to manage exotherms and selectivity. Furthermore, these legacy processes involved multiple distillation steps to purify intermediates like 4-chloro-3,3-dimethylnitrile, which not only increased energy consumption but also introduced risks of thermal decomposition and yield loss. The reliance on strong acids and corrosive reagents in subsequent steps further exacerbated equipment maintenance costs and limited the longevity of reactor vessels. In contrast, the novel approach detailed in the patent circumvents these limitations by utilizing a catalytic hydrogenation of 2,2-dimethyl-4-oxo-5-phenylpentanenitrile, which proceeds under much milder thermal conditions and eliminates the need for moisture-sensitive organometallic reagents entirely.

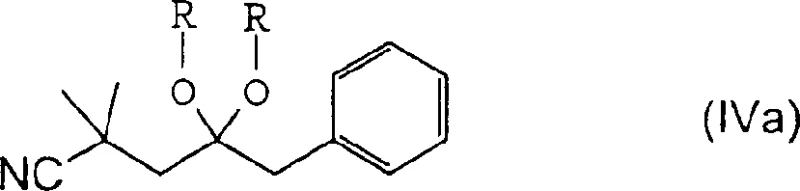
The mechanistic elegance of the new process is rooted in the selective reduction of the nitrile functionality in the presence of a ketone, followed by spontaneous intramolecular cyclization to form the pyrroline ring. A critical aspect of this transformation is the management of over-hydrogenation, which can lead to unwanted by-products such as tetramethyleneimine derivatives. The patent elucidates that the purity of the starting nitrile-ketone compound is paramount; utilizing precursors with purity exceeding 95% ensures a smooth reaction trajectory with minimal side reactions. Additionally, the choice of catalyst plays a pivotal role, with anhydrous Raney nickel demonstrating superior performance compared to moist variants or noble metals like platinum. To further enhance selectivity, the process allows for the temporary protection of the ketone group as a ketal, as shown in the formation of Formula IVa, which shields the carbonyl from reduction during the nitrile hydrogenation step. This strategic protection-deprotection sequence ensures that the cyclization occurs exclusively at the desired stage, yielding the 2-benzyl-4,4-dimethyl-1-pyrroline intermediate with high fidelity.
Mechanistic Insights into Catalytic Hydrogenation and Cyclization
The hydrogenation step represents the cornerstone of this improved synthesis, where the interplay between solvent composition, catalyst state, and substrate purity dictates the overall success of the transformation. When employing Raney nickel in a toluene and methanol mixture, the reaction kinetics are significantly accelerated, allowing the reduction to proceed efficiently at moderate temperatures between 50°C and 60°C. The mechanism involves the initial reduction of the nitrile group to a primary amine, which then immediately condenses with the adjacent ketone to form a cyclic imine, effectively driving the equilibrium towards the desired pyrroline product. However, without careful control, the catalyst may further reduce the cyclic imine to a saturated pyrrolidine or reduce the ketone directly, leading to complex impurity profiles that are difficult to separate. The patent highlights that maintaining the catalyst in an anhydrous state prior to reaction is crucial for suppressing these over-reduction pathways, as water can alter the surface activity of the nickel and promote non-selective hydrogen uptake. Furthermore, the use of ketal protection offers an alternative mechanistic pathway where the ketone is rendered inert, allowing the nitrile to be reduced under more forcing conditions if necessary, followed by acidic hydrolysis to reveal the ketone and trigger cyclization.
Impurity control is rigorously addressed through the optimization of reaction parameters and workup procedures, ensuring that the final intermediate meets the stringent specifications required for pharmaceutical applications. One of the primary impurities of concern is the isomeric 5-(4-chlorophenyl) derivative, which can arise during the subsequent cyclization with omega-bromo-4-chloroacetophenone if reaction conditions are not tightly regulated. The novel process mitigates this by conducting the cyclization in methanol with solid sodium bicarbonate as a base scavenger, which effectively neutralizes the generated hydrogen bromide without introducing excessive water that could hydrolyze sensitive intermediates. The resulting crude product is purified through a simple aqueous wash at elevated temperatures, which removes inorganic salts and residual starting materials while retaining the organic product in high yield. This streamlined purification protocol stands in stark contrast to the laborious chromatographic or distillation methods often required in older syntheses, thereby reducing the environmental footprint and operational time. The ability to achieve purity levels greater than 97% with minimal isomer content demonstrates the robustness of this mechanistic design.
How to Synthesize 6-(4-chlorophenyl)-2,2-dimethyl-7-phenyl-2,3-dihydro-1H-pyrrolizine Efficiently
The execution of this synthesis requires precise adherence to the optimized conditions regarding catalyst preparation, solvent ratios, and temperature gradients to maximize yield and minimize by-product formation. Operators must ensure that the Raney nickel catalyst is thoroughly washed with anhydrous methanol to remove any residual water before being introduced to the reaction vessel, as moisture content is a critical variable affecting selectivity. The reaction mixture should be maintained under a positive pressure of hydrogen, typically between 4 to 6 bar, to ensure sufficient gas-liquid mass transfer for the reduction to proceed to completion within a reasonable timeframe of 3 to 4 hours. Following the hydrogenation, the catalyst is removed via filtration, and the solvent is exchanged or adjusted to facilitate the subsequent cyclization step with the bromo-acetophenone derivative. Detailed standardized operating procedures for each unit operation, including specific quenching protocols and crystallization parameters, are essential for reproducibility.
- Prepare 2-benzyl-4,4-dimethyl-1-pyrroline via catalytic hydrogenation of 2,2-dimethyl-4-oxo-5-phenylpentanenitrile using Raney nickel.
- React the resulting pyrroline intermediate with omega-bromo-4-chloroacetophenone in a polar solvent like methanol to form the pyrrolizine core.
- Introduce the acetic acid side chain at the 5-position through reaction with oxalyl chloride followed by hydrazine reduction and hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers substantial benefits for procurement managers and supply chain directors focused on cost reduction in pharmaceutical intermediates manufacturing. By eliminating the requirement for cryogenic cooling systems and expensive Grignard reagents, the process significantly lowers both capital expenditure on specialized equipment and operational expenditure on raw materials and energy. The avoidance of strong acids and corrosive by-products extends the lifecycle of reactor vessels and piping, reducing maintenance downtime and replacement costs associated with equipment degradation. Furthermore, the simplified purification steps, which rely on aqueous washes and crystallization rather than complex distillations or chromatography, translate into faster batch cycles and higher throughput capacity. These efficiencies collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The transition from Grignard chemistry to catalytic hydrogenation removes the need for hazardous and costly organometallic reagents, which are often subject to volatile pricing and strict handling regulations. Additionally, the elimination of cryogenic operations reduces energy consumption drastically, as maintaining reactors at -78°C is far more resource-intensive than operating at ambient or moderately elevated temperatures. The simplified workup procedure, which avoids tedious distillation of intermediates, further cuts down on utility costs and solvent usage, leading to a leaner and more economical production model. These factors combined ensure a competitive cost structure that enhances the overall profitability of the final API production.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials such as nitriles and ketones, rather than moisture-sensitive organometallics, greatly improves the reliability of the supply chain. The robustness of the catalytic hydrogenation step means that the process is less susceptible to variations in raw material quality or minor fluctuations in reaction conditions, ensuring consistent output quality. Moreover, the reduced generation of hazardous waste simplifies disposal logistics and regulatory compliance, minimizing the risk of supply disruptions due to environmental violations. This stability allows for more accurate forecasting and inventory management, securing a steady flow of high-purity intermediates for downstream synthesis.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard hydrogenation reactors and filtration equipment that are common in fine chemical manufacturing facilities. The reduction in hazardous waste streams, particularly the avoidance of magnesium salts and halogenated by-products associated with Grignard reactions, aligns with increasingly stringent environmental regulations. The ability to recycle solvents like toluene and methanol further enhances the sustainability profile of the process. This combination of scalability and environmental stewardship makes the technology an attractive option for companies looking to expand production capacity while adhering to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this advanced synthesis method for ML3000 intermediates. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating the feasibility of technology transfer and for quality assurance personnel establishing control strategies. The answers reflect the specific advantages of the hydrogenation route over traditional methods, highlighting improvements in safety, efficiency, and product quality.
Q: How does the novel hydrogenation route improve upon traditional Grignard methods?
A: The novel route eliminates the need for cryogenic temperatures (-78°C) and hazardous Grignard reagents, replacing them with catalytic hydrogenation under milder conditions, which significantly reduces equipment corrosion and purification complexity.
Q: What strategies are used to prevent over-hydrogenation in this process?
A: Over-hydrogenation is controlled by using anhydrous Raney nickel catalysts and optimizing solvent systems, such as toluene-methanol mixtures, or by employing ketal protection groups on the ketone functionality prior to reduction.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process avoids complex distillation steps for intermediates and utilizes robust catalytic hydrogenation, making it highly scalable and economically viable for industrial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-(4-chlorophenyl)-2,2-dimethyl-7-phenyl-2,3-dihydro-1H-pyrrolizin-5-ylacetic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of next-generation anti-inflammatory therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. By leveraging the advanced hydrogenation technologies described in recent patents, we can offer our partners a secure and cost-effective supply of complex heterocyclic building blocks essential for the synthesis of dual COX/LOX inhibitors.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how our manufacturing capabilities can optimize your supply chain. Request a Customized Cost-Saving Analysis today to understand the specific economic benefits of switching to our optimized process. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your development timelines are met with the highest standards of quality and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
