Advanced Synthetic Route for ZD6126 Phenol: Enhancing Purity and Scalability for Global Pharmaceutical Supply Chains
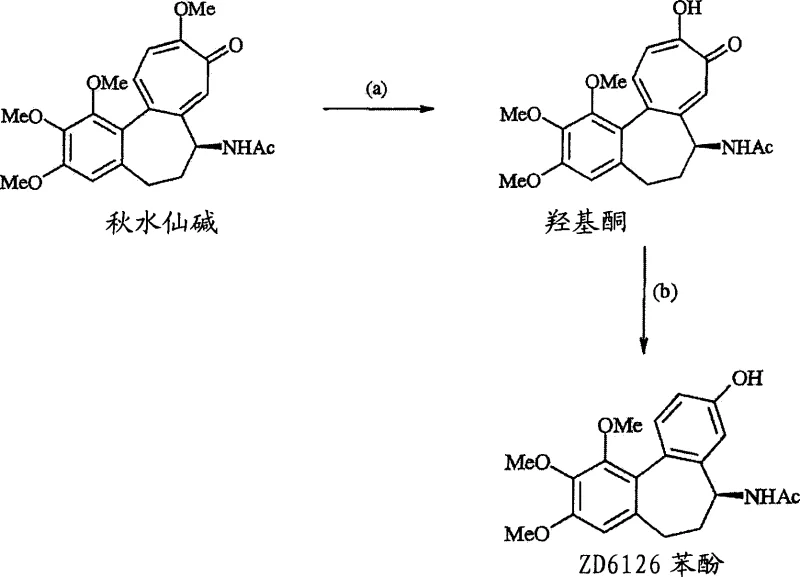
The pharmaceutical industry constantly seeks robust synthetic pathways for potent vascular targeting agents, and patent CN1910140A presents a transformative approach to producing ZD6126 phenol, also known as N-acetylcolchinol. This compound serves as a critical intermediate for synthesizing ZD6126, a phosphate prodrug designed to selectively disrupt tumor vasculature. The disclosed technology addresses long-standing inefficiencies in prior art by introducing a novel acid-catalyzed oxidative rearrangement strategy. Unlike conventional methods that rely on harsh hydrolysis conditions resulting in poor mass balance, this invention utilizes isocolchicine esters or ZD6126 alcohol precursors to achieve surprisingly high yields. By leveraging specific organometallic additions followed by controlled oxidation, the process ensures a reliable supply of high-purity intermediates essential for oncology drug development. This technical breakthrough not only enhances chemical efficiency but also aligns with modern green chemistry principles by minimizing waste and avoiding excessive thermal stress on sensitive molecular structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ZD6126 phenol from colchicine involved a cumbersome two-step sequence described in earlier literature, such as WO 99/02166. This traditional pathway, often referred to as Process A, necessitates acidolysis using hydrochloric acid at temperatures approaching 100°C, followed by treatment with basic hydrogen peroxide. The inherent flaws of this method are stark, with reported synthetic yields for the initial acidolysis step hovering around 79% and the subsequent oxidation step plummeting to merely 25%. Consequently, the overall yield stagnates at a commercially unviable 19%, rendering it unsuitable for large-scale production. Furthermore, the requirement for strong mineral acids at elevated temperatures poses significant safety risks and equipment corrosion challenges, while the low throughput exacerbates raw material costs. Such inefficiencies create bottlenecks in the supply chain, making it difficult for procurement teams to secure consistent volumes of the intermediate without incurring prohibitive expenses.
The Novel Approach
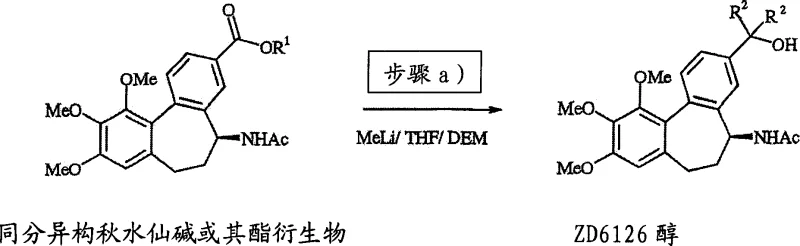
In sharp contrast, the methodology outlined in CN1910140A introduces a sophisticated route that bypasses the limitations of direct acidolysis. By first converting isocolchicine or its ester derivatives into ZD6126 alcohol intermediates using organometallic reagents, the process establishes a robust foundation for the subsequent transformation. The core innovation lies in the oxidative rearrangement of these alcohol intermediates using a combination of acid catalysts and oxidizing agents under mild conditions. This novel approach has demonstrated overall yields of approximately 75% from colchicine, representing a nearly four-fold improvement over traditional techniques. The ability to operate at lower temperatures, typically between 30°C and 70°C, significantly reduces energy consumption and thermal degradation risks. Moreover, the flexibility to use various acid catalysts, such as methanesulfonic acid or p-toluenesulfonic acid, allows manufacturers to optimize reaction kinetics and impurity profiles, ensuring a more stable and predictable manufacturing process that meets stringent regulatory standards for pharmaceutical intermediates.
Mechanistic Insights into Acid-Catalyzed Oxidative Rearrangement
The chemical elegance of this synthesis lies in the mechanistic pathway converting ZD6126 alcohol to the corresponding phenol. The reaction proceeds through an acid-catalyzed oxidative rearrangement where the benzylic alcohol functionality is activated by a proton source, facilitating the departure of a leaving group or the formation of a carbocation-like species. Simultaneously, the presence of an oxidizing agent, preferably hydrogen peroxide, drives the aromatization of the seven-membered ring system. Detailed studies within the patent reveal that this transformation is not a simple single-step event but involves a complex equilibrium of intermediates. Specifically, the reaction mixture may transiently contain ZD6126 alkenes, hydroperoxides, and even reactive dimers. Understanding this mechanistic landscape is crucial for R&D directors aiming to control the impurity profile. The patent explicitly demonstrates that these potential by-products are not dead ends; rather, they can be effectively recycled or converted back into the desired ZD6126 phenol under the same reaction conditions, thereby maximizing atom economy and minimizing the formation of persistent organic pollutants that would otherwise require costly removal steps.
Controlling the impurity spectrum is paramount for ensuring the safety and efficacy of the final API. The formation of side products like ZD6126 alkenes occurs primarily when at least one R2 group on the alcohol intermediate is a C1-4 alkyl, leading to elimination reactions. Similarly, ZD6126 hydroperoxides can accumulate if the reaction temperature is kept too low, favoring their stability over rearrangement. The patent provides critical guidance on managing these species by adjusting reaction parameters such as temperature and catalyst concentration. For instance, maintaining the reaction at approximately 50°C with a dual addition of acid and oxidant ensures that hydroperoxides and dimers are rapidly consumed to form the phenol. This dynamic control mechanism prevents the accumulation of genotoxic alerts or unstable peroxides in the final product. By rigorously monitoring these mechanistic pathways through HPLC analysis, manufacturers can guarantee a high-purity output that simplifies downstream purification, ultimately reducing the burden on quality control laboratories and accelerating batch release times for clinical supply.
How to Synthesize ZD6126 Phenol Efficiently
Implementing this advanced synthetic route requires precise adherence to the operational parameters defined in the patent to achieve optimal results. The process begins with the preparation of the ZD6126 alcohol intermediate, typically achieved by reacting isocolchicine esters with methyllithium in ethereal solvents like tetrahydrofuran at sub-ambient temperatures. Once the alcohol is secured, either as an isolated solid or in solution, the critical oxidative rearrangement step commences. The detailed standardized synthesis steps below outline the specific reagent stoichiometry, solvent choices, and workup procedures necessary to replicate the high yields reported in the intellectual property. Following these guidelines ensures that the complex interplay between the acid catalyst and oxidizing agent is managed safely, preventing runaway exotherms while driving the reaction to completion. This structured approach allows technical teams to scale the process from laboratory benchtop to pilot plant with confidence, knowing that the critical quality attributes of the intermediate are maintained throughout the campaign.
- React isocolchicine or its ester derivatives with an organometallic reagent like methyllithium in ethereal solvents at low temperatures to form ZD6126 alcohol.
- Perform an acid-catalyzed oxidative rearrangement using methanesulfonic acid and hydrogen peroxide in aromatic solvents like toluene at elevated temperatures.
- Quench the reaction with sodium thiosulfate, adjust pH, and crystallize the final ZD6126 phenol product using heptane and butyl acetate mixtures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers substantial strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly translates to cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for harsh hydrolysis conditions and multiple isolation steps associated with older methods, the new process reduces utility consumption and minimizes the wear and tear on reactor vessels. The ability to achieve high conversion rates in aromatic solvents like toluene or chlorobenzene means that solvent recovery systems can be optimized for these common industrial chemicals, further lowering operational expenditures. Additionally, the robustness of the reaction against variations in raw material quality ensures a more consistent supply, mitigating the risk of batch failures that often disrupt production schedules. This reliability is essential for maintaining continuous operations in a highly regulated environment where downtime can result in significant financial penalties and delayed market entry for new therapies.
- Cost Reduction in Manufacturing: The transition to this high-yield pathway eliminates the massive material losses inherent in the legacy 19% yield process, effectively quadrupling the output per unit of starting colchicine. Since colchicine is a natural product with fluctuating market prices, maximizing its conversion efficiency is a critical lever for controlling COGS. Furthermore, the process avoids the use of expensive transition metal catalysts or exotic reagents, relying instead on commodity chemicals like methanesulfonic acid and hydrogen peroxide. This reliance on readily available inputs stabilizes the supply chain against geopolitical disruptions or raw material shortages. The reduction in waste generation also lowers disposal costs, contributing to a leaner and more economically sustainable production model that enhances the overall margin profile of the final drug product.
- Enhanced Supply Chain Reliability: The scalability of this process is evidenced by its tolerance for a wide range of solvents and its ability to proceed without isolating the unstable alcohol intermediate in a telescoped manner. This flexibility allows manufacturers to adapt to local solvent availability or regulatory restrictions without compromising product quality. The dual addition technique for acid and oxidant enhances safety by preventing the pre-mixing of hazardous components, thereby reducing the risk of plant incidents that could halt production. Moreover, the high purity of the crude product reduces the need for extensive chromatographic purification, shortening the cycle time from raw material intake to finished goods. These factors collectively build a resilient supply chain capable of meeting the rigorous demands of global pharmaceutical clients who require just-in-time delivery of critical oncology intermediates.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with modern sustainability goals by utilizing aqueous hydrogen peroxide, which decomposes into water and oxygen, leaving no toxic heavy metal residues. The use of aromatic solvents, while requiring careful handling, allows for efficient recycling through distillation, minimizing the volume of organic waste sent for incineration. The ability to convert side products like dimers and hydroperoxides back into the main product stream further enhances the green chemistry profile by improving atom economy. This comprehensive approach to waste minimization simplifies the permitting process for new manufacturing facilities and ensures compliance with increasingly stringent environmental regulations. Consequently, companies adopting this technology can position themselves as responsible partners in the pharmaceutical value chain, appealing to investors and customers who prioritize ESG criteria in their supplier selection processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific experimental data and embodiments described in the patent documentation, providing clarity on reaction conditions, impurity management, and scalability. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure. The answers reflect the proven capabilities of the method to deliver high-quality intermediates consistently, addressing the key concerns of R&D and operations stakeholders alike.
Q: What are the critical impurities formed during ZD6126 phenol synthesis?
A: The process may generate ZD6126 alkenes, hydroperoxides, and reactive dimers as side products, all of which can be converted back to the desired phenol under optimized conditions.
Q: Why is the new oxidative rearrangement method superior to traditional acidolysis?
A: Traditional methods using hydrochloric acid at high temperatures yield only 19% overall, whereas the new acid-catalyzed oxidative route achieves yields up to 75% from colchicine.
Q: Which solvents are preferred for the oxidative rearrangement step?
A: Aromatic solvents such as toluene, chlorobenzene, or xylene are preferred due to their ability to provide high conversion rates despite the relatively low solubility of the alcohol intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ZD6126 Phenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex chemistry described in CN1910140A can be executed with precision and safety. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch. Our commitment to excellence means that we do not just supply chemicals; we provide solutions that de-risk your drug development timeline. By leveraging our expertise in acid-catalyzed oxidative rearrangements and organometallic transformations, we can deliver ZD6126 phenol that meets the exacting standards required for clinical and commercial applications.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need specific COA data to validate our material against your internal standards, we are ready to assist. Our experts can provide detailed route feasibility assessments to ensure seamless technology transfer and rapid scale-up. Partnering with us means gaining access to a reliable supply of complex pharmaceutical intermediates backed by a decade of proven performance in the fine chemical sector. Let us help you optimize your production costs and secure your supply chain for the future.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
