Advanced Lithiation Strategy for Commercial Scale Production of High-Purity 2-Formyl Oxazole
Advanced Lithiation Strategy for Commercial Scale Production of High-Purity 2-Formyl Oxazole
The pharmaceutical and fine chemical industries constantly seek robust synthetic routes for critical heterocyclic building blocks, and the methodology disclosed in patent CN102532049B represents a significant leap forward in the preparation of 2-formyl oxazole. This innovative approach utilizes commercially available oxazole as a starting material, subjecting it to controlled lithiation followed by formylation with N-formylpiperidine to yield a highly pure solid lithium salt intermediate. The subsequent hydrolysis of this stable intermediate provides the target aldehyde with exceptional efficiency. By shifting away from traditional formylating agents that often result in complex oily mixtures, this process ensures a nuclear magnetic resonance (NMR) internal standard purity stabilizing above 98.0%, with isolated yields consistently ranging between 75.0% and 85.0%. 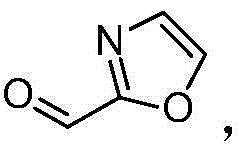 This technological advancement not only simplifies the operational workflow but also drastically reduces environmental pollution, offering a sustainable and scalable solution for the production of high-value oxazole derivatives essential for modern drug discovery and agrochemical applications.
This technological advancement not only simplifies the operational workflow but also drastically reduces environmental pollution, offering a sustainable and scalable solution for the production of high-value oxazole derivatives essential for modern drug discovery and agrochemical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-formyl oxazole has been plagued by significant downstream processing challenges that hinder efficient commercial production. Traditional organolithium methods typically employ N,N-dimethylformamide (DMF) or N-formylmorpholine as the formylating source, both of which present distinct disadvantages in an industrial setting. When DMF is utilized, the resulting reaction mixture often necessitates rigorous purification via silica gel column chromatography to separate the product from byproducts, a technique that is notoriously difficult to scale and results in substantial material loss due to the product's high water solubility and low boiling point. Alternatively, the use of N-formylmorpholine frequently yields the product as an inseparable oily mixture rather than a crystalline solid, complicating storage, transportation, and quality control. These conventional pathways suffer from low overall yields and generate significant waste, making them economically unviable for large-scale manufacturing where cost efficiency and reproducibility are paramount concerns for procurement and supply chain managers.
The Novel Approach
In stark contrast to these legacy methods, the novel protocol described in the patent introduces N-formylpiperidine as a superior formylating agent, fundamentally altering the physical state of the reaction intermediate to facilitate easier handling. By reacting the lithiated oxazole species with N-formylpiperidine under controlled thermal conditions, the process generates a solid lithium salt intermediate that can be isolated via simple filtration and washing. This solid intermediate exhibits remarkable stability and high purity, effectively bypassing the need for complex chromatographic separation techniques. The ability to isolate a discrete, high-purity solid species before the final hydrolysis step allows for rigorous quality assurance prior to product release, ensuring that only materials meeting stringent specifications proceed to the final stage. This strategic modification transforms a previously cumbersome liquid-liquid extraction or chromatography-heavy process into a streamlined solid-phase isolation workflow, dramatically enhancing the feasibility of commercial scale-up and reducing the operational burden on production facilities.
Mechanistic Insights into Low-Temperature Lithiation and Formylation
The core of this synthetic strategy relies on the precise generation of the C2-lithiated oxazole species through a cryogenic lithiation process. The reaction initiates by dissolving oxazole in an anhydrous ether solvent, such as tetrahydrofuran (THF) or 2-methyltetrahydrofuran, and cooling the system to a temperature range of -90°C to -60°C. At these low temperatures, n-butyllithium is added dropwise to effect the deprotonation at the C2 position of the oxazole ring, a step that requires strict thermal control to prevent side reactions or decomposition of the sensitive organolithium species. Following the formation of the lithiated intermediate, N-formylpiperidine is introduced to the reaction mixture, where the nucleophilic carbon at the C2 position attacks the carbonyl carbon of the formyl group. 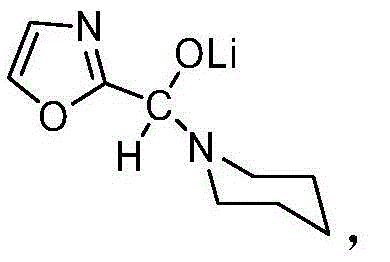 The reaction mixture is then allowed to warm gradually to 20°C to 30°C and maintained at this temperature for an extended period, typically between 32 to 72 hours, to ensure complete conversion to the lithium salt of the hemiaminal ether. This prolonged incubation at moderate temperatures is critical for driving the equilibrium towards the formation of the stable solid intermediate, which precipitates out of the solution upon cooling, thereby leveraging Le Chatelier's principle to maximize yield.
The reaction mixture is then allowed to warm gradually to 20°C to 30°C and maintained at this temperature for an extended period, typically between 32 to 72 hours, to ensure complete conversion to the lithium salt of the hemiaminal ether. This prolonged incubation at moderate temperatures is critical for driving the equilibrium towards the formation of the stable solid intermediate, which precipitates out of the solution upon cooling, thereby leveraging Le Chatelier's principle to maximize yield.
Impurity control in this process is inherently managed through the physical properties of the intermediate lithium salt. Unlike the oily mixtures produced by DMF-based methods, which trap impurities within a viscous matrix, the crystallization of the solid lithium salt acts as a self-purification step. As the salt precipitates from the ether solvent upon cooling to -5°C to 10°C, soluble impurities and excess reagents remain in the mother liquor and are removed during the suction filtration and washing steps. The use of methyl tert-butyl ether (MTBE) for washing the filter cake further enhances purity by removing organic contaminants without dissolving the desired lithium salt. Finally, the hydrolysis step using dilute acid (such as 5-20% hydrochloric acid or acetic acid) cleaves the hemiaminal ether linkage to release the free aldehyde. Because the precursor salt is already of high purity (>98.0% by NMR), the final hydrolysis yields 2-formyl oxazole with minimal byproduct formation, eliminating the need for further purification and ensuring a clean impurity profile suitable for pharmaceutical applications.
How to Synthesize 2-Formyl Oxazole Efficiently
The synthesis of 2-formyl oxazole via this lithiation pathway is designed for operational simplicity and robustness, making it an ideal candidate for technology transfer from laboratory to pilot plant. The process begins with the preparation of the reaction vessel under an inert atmosphere, followed by the addition of oxazole and the selected ether solvent. Critical to the success of the reaction is the maintenance of low temperatures during the addition of the pyrophoric n-butyllithium reagent to ensure safety and selectivity. Once the lithiation is complete, the addition of N-formylpiperidine and the subsequent warming phase require careful monitoring to optimize the formation of the solid intermediate. The detailed standardized synthesis steps, including specific molar ratios, stirring rates, and crystallization parameters, are outlined in the guide below to assist process engineers in replicating these results accurately.
- Dissolve oxazole in an ether solvent such as THF and cool the mixture to between -90°C and -60°C before slowly adding n-butyllithium to generate the lithiated species.
- Introduce N-formylpiperidine to the reaction system while maintaining low temperatures, then allow the mixture to warm to 20-30°C to facilitate the formylation reaction.
- Cool the system to induce crystallization of the solid lithium salt intermediate, filter and wash the solid, and finally hydrolyze with dilute acid to obtain the pure 2-formyl oxazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical yield, addressing critical pain points related to cost, logistics, and scalability. The primary advantage lies in the drastic simplification of the workup procedure; by eliminating the need for silica gel column chromatography, the process removes a major bottleneck that typically consumes significant amounts of solvents, stationary phase, and labor hours. This reduction in processing complexity translates directly into lower manufacturing costs and a smaller environmental footprint, aligning with modern green chemistry initiatives. Furthermore, the generation of a stable solid intermediate mitigates the risks associated with transporting unstable oily compounds, thereby enhancing supply chain reliability and reducing the potential for product degradation during transit between manufacturing sites or to external customers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and time-consuming purification steps. Traditional methods relying on chromatography incur high costs due to the consumption of silica gel and large volumes of elution solvents, which also require costly disposal or recycling. By contrast, this method relies on crystallization and filtration, unit operations that are significantly cheaper and faster to execute on a large scale. Additionally, the higher yield range of 75.0% to 85.0% means that less raw material is required to produce the same amount of final product, effectively lowering the cost of goods sold (COGS). The use of commercially available and relatively inexpensive reagents like N-formylpiperidine further contributes to a favorable cost structure, making the final 2-formyl oxazole more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the instability of intermediates and the complexity of synthesis. The ability to isolate and store the solid lithium salt intermediate provides a strategic buffer; manufacturers can produce the intermediate in batches and store it for later hydrolysis based on demand, decoupling the upstream lithiation step from the final product delivery. This flexibility allows for better inventory management and reduces the risk of batch failures affecting immediate customer deliveries. Moreover, the robustness of the reaction conditions, which tolerate a range of temperatures and solvent choices (THF, MTBE, ether), ensures that production is less susceptible to minor fluctuations in utility availability or raw material quality, guaranteeing a steady flow of high-purity material to downstream users.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but this route is explicitly designed for large-scale production, with patent examples demonstrating successful execution in reactors as large as 1500L. The avoidance of chromatographic purification removes a significant barrier to scale-up, as column chromatography is rarely feasible for multi-ton production. From an environmental perspective, the process generates less hazardous waste due to the reduced solvent usage and the absence of silica waste streams. The use of dilute acid for hydrolysis and the potential for solvent recovery (ether, THF, MTBE) further supports compliance with increasingly stringent environmental regulations. This scalability ensures that the supply of 2-formyl oxazole can grow in tandem with the demands of the pharmaceutical industry without requiring disproportionate increases in infrastructure or waste treatment capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-formyl oxazole using this advanced lithiation methodology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on process capabilities and product quality. Understanding these details is crucial for R&D teams evaluating this route for their own synthetic programs and for procurement specialists assessing the reliability of the supply source.
Q: Why is N-formylpiperidine preferred over DMF for this synthesis?
A: Unlike DMF which often requires complex silica gel chromatography and yields oily mixtures difficult to purify, N-formylpiperidine allows for the isolation of a high-purity solid lithium salt intermediate (>98% NMR purity), significantly simplifying downstream processing and improving overall yield to 75-85%.
Q: What are the storage advantages of the intermediate lithium salt?
A: The process generates a stable solid lithium salt that can be isolated, dried under nitrogen protection, and stored or transported safely. This contrasts with traditional methods that produce unstable oily mixtures, thereby enhancing supply chain reliability and reducing degradation risks during logistics.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly demonstrates scalability with examples ranging from 100L to 1500L reactors. The use of stable process conditions, mild hydrolysis steps, and the elimination of difficult purification techniques like column chromatography makes it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Formyl Oxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We are committed to delivering 2-formyl oxazole with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the highest standards required for pharmaceutical synthesis. Our capability to implement the advanced lithiation process described in CN102532049B allows us to offer a product with superior consistency and lower impurity levels compared to traditional sources, providing our partners with a distinct advantage in their own drug development timelines.
We invite global pharmaceutical and agrochemical companies to collaborate with us to optimize their supply chains for oxazole derivatives. By leveraging our technical expertise and manufacturing capacity, we can help you achieve significant cost reduction in API manufacturing while ensuring a secure and reliable supply of critical building blocks. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced production methods can support your project goals and accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
