Revolutionizing Asymmetric Suzuki Coupling for Scalable Pharmaceutical Intermediate Production
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral centers, particularly within the realm of non-steroidal anti-inflammatory drugs (NSAIDs) and bioactive molecules. A significant technological breakthrough has been documented in patent CN111848680B, which introduces a novel class of bidentate phosphine-phosphine oxide ligands designed to overcome historical limitations in asymmetric synthesis. This innovation specifically targets the Suzuki-Miyaura coupling reaction, a cornerstone transformation in modern organic chemistry, enabling the efficient construction of alpha-aryl carbonyl compounds with exceptional stereocontrol. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing pathways for high-value pharmaceutical intermediates. The technology addresses critical pain points such as catalyst loading, functional group tolerance, and the suppression of unwanted side reactions, positioning it as a vital tool for next-generation process development.
Furthermore, the implications of this patent extend beyond mere academic interest, offering tangible benefits for supply chain stability and commercial viability. By utilizing arylboronic acids, which are commercially available, stable, and environmentally benign compared to organozinc or Grignard reagents, the process aligns with green chemistry principles while maintaining high operational efficiency. The ability to achieve high enantiomeric excess (ee) values without resorting to expensive chiral resolution steps post-synthesis drastically reduces waste and processing time. As we delve deeper into the technical specifics, it becomes evident that this ligand system provides a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and purity required by global regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of alpha-aryl carbonyl structures has relied heavily on transition metal-catalyzed cross-coupling reactions involving nickel, iron, or cobalt systems. While these methods have demonstrated utility in specific contexts, they are plagued by inherent inefficiencies that hinder large-scale adoption. A primary drawback is the requirement for high catalyst loading, often exceeding 5 mol%, which not only inflates raw material costs but also complicates the removal of residual heavy metals from the final active pharmaceutical ingredient (API). Additionally, many conventional protocols necessitate the use of highly reactive organometallic reagents such as Grignard or organozinc species, which are sensitive to moisture and air, demanding stringent anhydrous conditions and specialized equipment that increase operational complexity and safety risks.
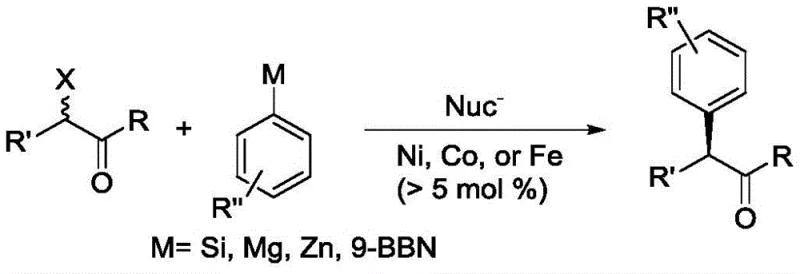
Moreover, the substrate scope for these traditional methods is frequently limited by steric hindrance and functional group compatibility issues. The presence of sensitive moieties often leads to decomposition or side reactions, resulting in lower yields and difficult purification processes. Another significant challenge is the tendency for self-coupling byproducts to form, which competes with the desired cross-coupling pathway and diminishes the overall atom economy of the reaction. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks for procurement managers seeking to optimize supply chains for high-purity pharmaceutical intermediates. The industry urgently requires a solution that mitigates these risks while enhancing reaction efficiency and selectivity.
The Novel Approach
In stark contrast to conventional methodologies, the novel approach detailed in the patent utilizes a specially designed bidentate phosphine-phosphine oxide ligand in conjunction with a palladium catalyst to facilitate asymmetric Suzuki-Miyaura coupling. This system operates under remarkably mild conditions, typically between 40°C and 80°C, and employs stable arylboronic acids as coupling partners, eliminating the need for hazardous organometallic reagents. The unique electronic and steric properties of the ligand allow for drastic reductions in catalyst loading, with palladium usage potentially as low as 0.001 mol%, significantly lowering the burden of metal impurity control. This advancement not only streamlines the synthetic route but also enhances the safety profile of the manufacturing process, making it highly attractive for industrial applications.
Crucially, this novel methodology effectively suppresses the formation of self-coupling byproducts, a persistent issue in alpha-arylation reactions, thereby ensuring higher selectivity for the desired cross-coupled product. The broad substrate compatibility allows for the introduction of diverse functional groups, including halogens, esters, and heterocycles, without compromising reaction performance. For supply chain heads, this translates to greater flexibility in sourcing starting materials and a more robust production schedule less susceptible to raw material variability. The combination of high yield, exceptional enantioselectivity, and operational simplicity positions this technology as a superior alternative for the commercial production of chiral building blocks essential for modern drug discovery and development.
Mechanistic Insights into Bidentate Phosphine-Phosphine Oxide Ligand Catalysis
The exceptional performance of this catalytic system stems from the unique structural features of the bidentate phosphine-phosphine oxide ligand, which creates a highly defined chiral environment around the palladium center. The ligand coordinates to the metal through both the phosphine and phosphine oxide moieties, forming a rigid five-membered ring structure that stabilizes the active catalytic species throughout the reaction cycle. This bidentate coordination mode prevents ligand dissociation, a common cause of catalyst deactivation in monodentate systems, thereby maintaining high turnover numbers even at ultra-low catalyst loadings. The steric bulk provided by substituents on the ligand framework, such as tert-butyl or adamantyl groups, effectively shields the metal center, directing the approach of substrates to favor the formation of one enantiomer over the other with high precision.
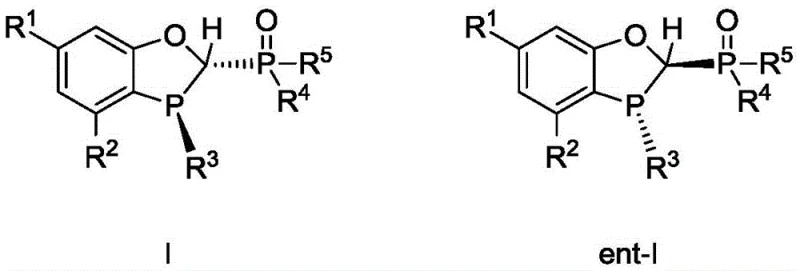
From an impurity control perspective, the mechanism inherently disfavors homocoupling pathways due to the specific electronic tuning of the palladium complex. The ligand facilitates rapid transmetallation with the arylboronic acid while simultaneously retarding the oxidative addition of the alpha-haloamide to itself, thus minimizing self-coupling side reactions. This selectivity is critical for R&D teams focused on purity profiles, as it reduces the complexity of downstream purification and ensures that the final product meets stringent regulatory specifications for chiral drugs. Furthermore, the stability of the ligand under reaction conditions means that it can withstand the presence of various bases and solvents without degradation, contributing to the overall robustness and reproducibility of the process across different batches and scales.
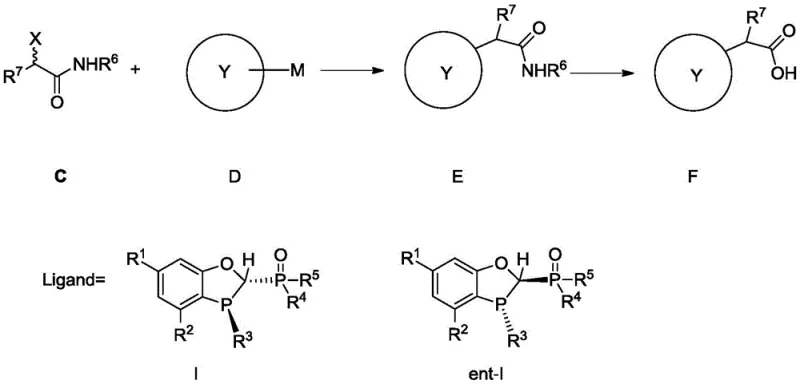
How to Synthesize Chiral Alpha-Aryl Amides Efficiently
The implementation of this advanced coupling technology involves a straightforward yet precise sequence of operations designed to maximize yield and enantioselectivity. The process begins with the preparation of the chiral ligand, followed by its application in the palladium-catalyzed coupling of alpha-bromoamides with arylboronic acids. Detailed standard operating procedures regarding stoichiometry, temperature control, and workup protocols are essential for achieving the reported high performance metrics. The following guide outlines the strategic framework for executing this synthesis, ensuring that technical teams can replicate the success demonstrated in the patent data while adhering to best practices for safety and quality assurance in a GMP environment.
- Preparation of the chiral bidentate phosphine-phosphine oxide ligand through reduction of phosphine oxide precursors using halosilane agents.
- Execution of the Suzuki-Miyaura coupling reaction between alpha-haloamides and arylboronic acids using palladium catalyst and the novel ligand.
- Hydrolysis of the resulting alpha-aryl amide intermediates to obtain high-purity chiral carboxylic acids such as NSAIDs.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology offers substantial strategic advantages that directly impact the bottom line and operational resilience. The most significant benefit lies in the drastic reduction of catalyst consumption; by lowering palladium loading to trace levels, manufacturers can achieve significant cost savings on precious metal inputs, which are often subject to volatile market pricing. Additionally, the elimination of expensive and hazardous organometallic reagents in favor of stable boronic acids simplifies logistics, reduces storage requirements, and minimizes waste disposal costs associated with quenching reactive species. These factors collectively contribute to a more economical and sustainable manufacturing process that enhances competitiveness in the global marketplace.
- Cost Reduction in Manufacturing: The ability to operate with ultra-low catalyst loading translates directly into reduced raw material expenses and lower costs associated with metal scavenging during purification. By avoiding the use of sensitive reagents that require specialized handling and infrastructure, facilities can further optimize their operational expenditures. The high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, improving overall atom economy and yield. Consequently, the total cost of goods sold (COGS) for the final intermediate is significantly lowered, providing a clear financial incentive for adopting this technology over legacy methods.
- Enhanced Supply Chain Reliability: Utilizing commercially available and stable arylboronic acids ensures a consistent and reliable supply of key starting materials, reducing the risk of production delays caused by reagent instability or scarcity. The mild reaction conditions reduce the dependency on specialized high-pressure or cryogenic equipment, allowing for production in a wider range of facilities with standard capabilities. This flexibility enhances supply chain agility, enabling manufacturers to respond more quickly to market demands and scale production up or down as needed without significant capital investment. The robustness of the process also ensures consistent quality across batches, fostering trust with downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from gram to kilogram scales without loss of performance, making it ideal for commercial production. The use of less hazardous reagents and the generation of fewer toxic byproducts align with increasingly strict environmental regulations and corporate sustainability goals. Reduced solvent usage and energy consumption due to mild reaction temperatures further decrease the environmental footprint of the manufacturing process. This compliance not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible and forward-thinking partner in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bidentate ligand technology in pharmaceutical synthesis. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing workflows. The information below serves as a foundational reference for decision-makers assessing the potential impact on their product portfolios and manufacturing strategies.
Q: How does this ligand improve upon traditional nickel or iron catalysis?
A: Traditional nickel or iron catalysis often requires high catalyst loading (>5 mol%) and harsh conditions. This novel phosphine-phosphine oxide ligand enables palladium catalysis with significantly lower loading (down to 0.001 mol%) and milder conditions, reducing metal contamination risks.
Q: What is the primary advantage regarding byproduct formation?
A: The specific bidentate structure effectively suppresses the generation of self-coupling byproducts, a common issue in alpha-arylation, thereby simplifying downstream purification and improving overall yield.
Q: Is this technology scalable for industrial API production?
A: Yes, the reaction conditions are mild (40-80°C) and utilize stable arylboronic acids, making the process highly suitable for commercial scale-up with robust safety profiles and environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ligand Supplier
The technological potential of this bidentate phosphine-phosphine oxide ligand system represents a significant opportunity for pharmaceutical companies seeking to optimize their synthesis of chiral intermediates. NINGBO INNO PHARMCHEM stands ready to support your development efforts as a trusted CDMO partner with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team possesses the expertise to adapt this cutting-edge chemistry to your specific target molecules, ensuring that stringent purity specifications are met through our rigorous QC labs and advanced analytical capabilities. We understand the critical importance of consistency and quality in the supply of high-purity pharmaceutical intermediates and are committed to delivering solutions that meet the highest industry standards.
We invite you to engage with our technical procurement team to discuss how this innovative coupling technology can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the specific economic benefits applicable to your projects. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your needs. Let us collaborate to transform your synthetic challenges into commercial successes, leveraging our deep technical knowledge and robust manufacturing infrastructure to accelerate your path to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
