Advanced Catalytic Hydrogenation for High-Purity Toluene Derivatives and Pharmaceutical Intermediates
Advanced Catalytic Hydrogenation for High-Purity Toluene Derivatives and Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the need for more sustainable and cost-effective synthetic routes. A pivotal development in this sector is detailed in patent CN1622926A, which outlines a robust method for producing toluene derivatives through the catalytic hydrogenation of benzaldehydes and benzyl alcohols. This technology represents a strategic shift away from reliance on scarce and expensive noble metal catalysts, offering a viable pathway for the large-scale production of critical pharmaceutical intermediates and agrochemical building blocks. By leveraging base metal catalysis, specifically utilizing cobalt, nickel, and copper systems, this process addresses long-standing inefficiencies in traditional hydrogenation protocols. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for optimizing supply chains and reducing the overall cost of goods sold (COGS) for complex aromatic compounds.
The core innovation lies in the specific composition of the catalyst, which is engineered to maintain high activity and selectivity without the need for platinum group metals. The process accommodates a wide range of substrates where substituents can include halogens, alkyl groups, hydroxyls, or alkoxy groups, making it highly versatile for diverse synthetic applications. This flexibility ensures that manufacturers can produce a broad spectrum of high-purity intermediates required for modern drug discovery and development. As we delve deeper into the technical specifics, it becomes clear that this methodology not only enhances chemical efficiency but also aligns with global trends towards greener chemistry and reduced environmental impact in industrial operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of benzaldehydes and benzyl alcohols to their corresponding toluene derivatives has relied heavily on noble metal catalysts such as palladium on carbon (Pd/C) or platinum-based systems. While effective, these traditional methods suffer from significant economic and operational drawbacks that hinder scalability. A major issue is the formation of corrosive solvent mixtures; for instance, literature often cites the use of acetic acid as a solvent, which, when combined with water produced during the reaction, creates an aggressive environment that accelerates equipment degradation and increases maintenance costs. Furthermore, the reliance on precious metals introduces volatility into the supply chain, as the prices of palladium and rhodium are subject to extreme market fluctuations and geopolitical instability. Additionally, alternative methods utilizing copper chromite catalysts pose severe environmental and safety hazards due to the toxicity of hexavalent chromium, necessitating costly waste treatment and disposal procedures that burden the overall production economics.
The Novel Approach
The methodology described in CN1622926A offers a compelling solution to these entrenched problems by introducing a novel catalyst system based on non-noble metals. This approach utilizes a composite catalyst comprising cobalt, nickel, or copper, potentially promoted by other transition metals and stabilized with alkali or alkaline earth components. 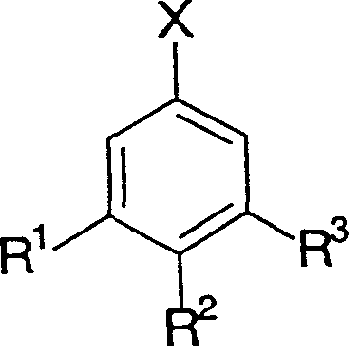 As illustrated in the reaction scheme, this system facilitates the direct conversion of Formula II compounds (benzaldehydes/benzyl alcohols) to Formula I (toluene derivatives) with remarkable efficiency. The elimination of acetic acid as a mandatory solvent allows for the use of milder, less corrosive media such as alcohols, ethers, or even solvent-free conditions in the gas phase. This shift not only preserves reactor integrity but also simplifies the workup procedure, leading to substantial operational savings. By replacing toxic chromium and expensive noble metals with abundant base metals, this technology provides a sustainable and economically superior alternative for the commercial scale-up of complex aromatic intermediates.
As illustrated in the reaction scheme, this system facilitates the direct conversion of Formula II compounds (benzaldehydes/benzyl alcohols) to Formula I (toluene derivatives) with remarkable efficiency. The elimination of acetic acid as a mandatory solvent allows for the use of milder, less corrosive media such as alcohols, ethers, or even solvent-free conditions in the gas phase. This shift not only preserves reactor integrity but also simplifies the workup procedure, leading to substantial operational savings. By replacing toxic chromium and expensive noble metals with abundant base metals, this technology provides a sustainable and economically superior alternative for the commercial scale-up of complex aromatic intermediates.
Mechanistic Insights into Base Metal-Catalyzed Hydrogenation
The success of this hydrogenation process hinges on the intricate synergy between the primary metal components and the various promoters within the catalyst matrix. The active phase typically consists of reduced cobalt, nickel, or copper species dispersed on a support or existing as a bulk solid. The addition of promoters such as molybdenum, zirconium, or manganese plays a critical role in modulating the electronic properties of the active sites, thereby enhancing the adsorption of hydrogen and the substrate while inhibiting undesirable side reactions. For example, the presence of zirconium oxide in the catalyst structure can improve thermal stability and prevent the sintering of active metal particles during the high-temperature activation phase, ensuring consistent performance over extended operation cycles. This precise engineering of the catalyst surface is what allows the process to achieve high conversion rates, often exceeding 90%, while maintaining exceptional selectivity for the methyl group formation over ring saturation.
From an impurity control perspective, the mechanism is designed to suppress the hydrogenolysis of ether bonds and the reduction of the aromatic ring, which are common pitfalls in aggressive hydrogenation environments. Traditional noble metal catalysts often lack the necessary discrimination, leading to the formation of cyclohexane derivatives or demethylated byproducts that are difficult to separate and reduce the overall yield of the desired API intermediate. The base metal catalysts described in this patent exhibit a unique chemoselectivity that preserves sensitive functional groups such as methoxy and halo substituents. This specificity is crucial for pharmaceutical synthesis, where the integrity of the molecular scaffold is paramount. By minimizing the generation of structural impurities, the process reduces the burden on downstream purification units, such as crystallization or chromatography, thereby streamlining the entire manufacturing workflow and improving the final product's quality profile.
How to Synthesize Toluene Derivatives Efficiently
Implementing this synthesis route requires careful attention to catalyst activation and reaction parameters to maximize throughput and yield. The process is adaptable to both batch and continuous flow operations, offering flexibility for different production scales. Whether operating in the liquid phase with solvents like methanol or tetrahydrofuran, or utilizing a solvent-free gas phase approach, the key lies in the proper pre-treatment of the catalyst to ensure the metal oxides are fully reduced to their active metallic states. The following guide outlines the standardized procedural framework derived from the patent examples, providing a roadmap for technical teams to replicate these high-efficiency results in a pilot or commercial setting.
- Preparation of the Catalyst System: Synthesize or procure a heterogeneous catalyst comprising Cobalt, Nickel, or Copper oxides promoted with metals like Molybdenum or Zirconium, ensuring the absence of expensive noble metals.
- Reaction Setup and Activation: Load the catalyst into a fixed-bed or slurry reactor and activate under a hydrogen stream at elevated temperatures (150-400°C) to reduce metal oxides to their active metallic states.
- Hydrogenation Process: Introduce the benzaldehyde or benzyl alcohol substrate (either as a melt, in solution, or gas phase) and react with hydrogen at 100-260°C and 20-250 bar pressure to achieve high conversion to the toluene derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this base-metal catalytic technology translates into tangible strategic benefits that extend beyond simple chemical yield. The most immediate impact is seen in the reduction of raw material costs, as the substitution of palladium and platinum with cobalt, nickel, and copper eliminates the exposure to volatile precious metal markets. This stability allows for more accurate long-term budgeting and pricing strategies for downstream customers. Moreover, the ability to operate without corrosive acetic acid significantly lowers the total cost of ownership for manufacturing assets by reducing the frequency of reactor replacements and maintenance downtime. These factors combine to create a more resilient and cost-efficient supply chain capable of meeting the rigorous demands of the global pharmaceutical and agrochemical industries.
- Cost Reduction in Manufacturing: The transition to non-noble metal catalysts fundamentally alters the cost structure of toluene derivative production. By removing the dependency on expensive platinum group metals, manufacturers can achieve significant savings on catalyst procurement and regeneration. Additionally, the high selectivity of the process minimizes the loss of valuable starting materials to byproducts, effectively increasing the mass balance efficiency. The potential for solvent-free operation further reduces costs by eliminating the need for solvent purchase, recovery, and disposal, leading to a leaner and more profitable manufacturing process that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: Relying on base metals such as copper and nickel mitigates the supply risks associated with geographically concentrated precious metal mining. These base metals are widely available and possess stable supply chains, ensuring continuity of production even during periods of geopolitical tension. Furthermore, the robustness of the catalyst system allows for longer campaign lengths without frequent change-outs, reducing the logistical burden of catalyst logistics and waste management. This reliability is critical for maintaining consistent delivery schedules to key clients in the pharmaceutical sector, where supply interruptions can have cascading effects on drug development timelines.
- Scalability and Environmental Compliance: The process is inherently scalable, with demonstrated efficacy in both gas-phase continuous reactors and liquid-phase batch systems. This versatility facilitates easy scale-up from pilot plant to multi-ton commercial production without the need for extensive process re-engineering. From an environmental standpoint, the avoidance of chromium-containing catalysts and corrosive acids aligns with increasingly stringent global environmental regulations. The reduction in hazardous waste generation simplifies compliance reporting and lowers the costs associated with environmental health and safety (EHS) management, positioning the manufacturer as a responsible and sustainable partner in the chemical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this hydrogenation method over conventional palladium-catalyzed processes?
A: The primary advantage is the elimination of expensive noble metals like palladium and platinum, significantly reducing raw material costs. Furthermore, this method avoids the use of corrosive acetic acid solvents often required in traditional methods, thereby reducing equipment maintenance costs and extending reactor lifespan.
Q: Does this process prevent the unwanted hydrogenation of the aromatic ring?
A: Yes, the specific formulation of the catalyst, particularly the inclusion of promoters and the controlled reaction conditions, is designed to exhibit high chemoselectivity. This effectively suppresses side reactions such as ring saturation (forming cyclohexane derivatives) or demethylation, ensuring high purity of the final toluene derivative.
Q: Can this technology be adapted for solvent-free production?
A: Absolutely. The patent data confirms that the hydrogenation can be successfully conducted in the gas phase or using the substrate melt without additional solvents. This capability drastically simplifies downstream processing by eliminating solvent recovery steps and reducing waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Toluene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern chemical industry. Our team of expert engineers and chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1622926A are fully realized in practical application. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle complex catalytic hydrogenations safely and efficiently, providing a secure foundation for your supply chain needs.
We invite you to collaborate with us to optimize your production costs and enhance your supply chain resilience. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to reach out to request specific COA data and route feasibility assessments to determine how our advanced manufacturing capabilities can support your next project. Let us help you navigate the complexities of chemical sourcing with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
