Advanced Dehydration Technology for High-Purity Alpha,Beta-Unsaturated Carboxylic Acids
Advanced Dehydration Technology for High-Purity Alpha,Beta-Unsaturated Carboxylic Acids
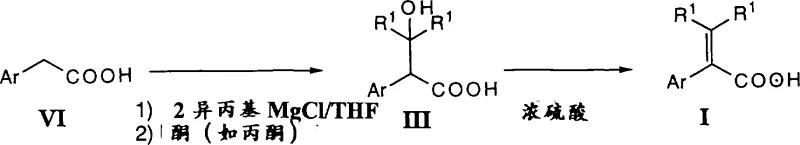
The pharmaceutical and fine chemical industries constantly seek robust synthetic routes that balance high purity with economic viability, particularly for complex intermediates used in cardiovascular therapeutics. Patent CN1162589A discloses a groundbreaking preparation method for alpha,beta-unsaturated organic carboxylic acids, which serve as critical precursors for optically active saturated acids used in drugs like Mibefradil. This technology represents a significant leap forward by replacing traditional, harsh dehydration conditions with a mild, high-yield protocol using concentrated sulfuric acid or polyphosphoric acid. By operating at temperatures between 0°C and 40°C, this method effectively suppresses detrimental side reactions such as decarboxylation and isomerization, which have historically plagued the synthesis of these valuable building blocks. For R&D directors and procurement specialists alike, understanding this process offers a pathway to securing a reliable API intermediate supplier capable of delivering superior quality materials with reduced processing complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha,beta-unsaturated acids from their corresponding beta-hydroxy precursors relied on dehydration agents such as tosic acid or potassium bisulfate, as documented in earlier literature like U.S. Pat. No. 4,409,397. While functional, these conventional approaches suffer from significant thermodynamic and kinetic drawbacks that compromise overall efficiency. Under the conditions required for these weaker acids to function, the reaction environment often promotes competing pathways, specifically the formation of isomeric acids and decarboxylation products. These side reactions not only consume valuable starting materials but also generate difficult-to-remove impurities that complicate downstream purification. Furthermore, to circumvent these issues, some prior art methods necessitated a multi-step sequence involving acid-catalyzed dehydration to an ester followed by saponification, thereby increasing both the operational time and the cost of goods sold. Such inefficiencies create bottlenecks in the supply chain, making it challenging to achieve the consistent high purity required for modern pharmaceutical applications.
The Novel Approach
In stark contrast, the methodology outlined in CN1162589A introduces a direct dehydration strategy utilizing very strong acids, specifically concentrated sulfuric acid or polyphosphoric acid, under remarkably mild thermal conditions. By maintaining the reaction temperature between 0°C and 40°C, preferably around 20-25°C, the process kinetically favors the elimination of water while thermodynamically disfavoring the formation of isomers and decarboxylation byproducts. This approach allows for the direct conversion of the hydroxy acid intermediate into the desired unsaturated acid with exceptional efficiency, reportedly achieving purified yields up to 99% in optimized examples. The use of sulfuric acid serves a dual purpose as both the catalytic proton source and the dehydrating agent, eliminating the need for auxiliary reagents. Additionally, the reaction can be performed at high concentrations, with a ratio of approximately 1 part hydroxy acid to 2-2.5 parts sulfuric acid, which significantly reduces solvent waste and reactor volume requirements. This streamlined protocol not only simplifies the workflow but also enhances the economic feasibility of producing these intermediates on a commercial scale.

Mechanistic Insights into Acid-Catalyzed Dehydration and Precursor Synthesis
The core innovation of this technology lies in the precise control of the dehydration mechanism through the selection of acid strength and temperature. In traditional weak acid systems, higher temperatures are required to drive the elimination of the hydroxyl group, which inadvertently provides the activation energy necessary for double bond migration (isomerization) and loss of carbon dioxide (decarboxylation). By employing a super-strong acid like sulfuric acid, the protonation of the hydroxyl group occurs rapidly even at near-ambient temperatures, facilitating the formation of the carbocation intermediate and subsequent elimination of water without triggering these degradation pathways. The patent data indicates that other strong acids like HCl or HBr are ineffective under these mild conditions, highlighting the unique role of sulfuric acid's dehydrating properties and non-nucleophilic nature in this specific transformation. This mechanistic understanding is crucial for process chemists aiming to replicate the high purity profiles observed in the patent examples, where gas chromatographic purity often exceeds 99.8%.
Furthermore, the synthesis of the requisite beta-hydroxy acid precursor (Formula III) is elegantly achieved through a Grignard addition to arylacetic acids, as illustrated in the reaction schemes provided. The process involves converting the arylacetic acid into a dianion using strong bases like organomagnesium compounds, followed by nucleophilic attack on acetone. This step is highly versatile, accommodating various substituents on the aryl ring, including halogens, alkyl groups, and alkoxy groups, allowing for the generation of a diverse library of intermediates. The ability to use the crude oily product of this Grignard reaction directly in the subsequent dehydration step without intermediate isolation further underscores the process intensification potential of this route. This telescoped approach minimizes material handling and exposure to atmospheric moisture, ensuring that the integrity of the sensitive intermediates is maintained throughout the synthesis.
Beyond the dehydration step, the utility of these unsaturated acids is maximized through their conversion into optically active saturated acids via asymmetric hydrogenation. The patent details the use of sophisticated transition metal catalysts, such as ruthenium complexes with chiral diphosphine ligands like (R)-BIPHEMP or rhodium complexes with ferrocenyl ligands. These catalytic systems enable the highly stereoselective reduction of the double bond, yielding chiral acids with enantiomeric excess values frequently surpassing 96%. This downstream capability transforms the achiral unsaturated acid into a high-value chiral building block essential for the synthesis of bioactive molecules. The robustness of the unsaturated acid precursor produced by the new dehydration method ensures that the hydrogenation catalysts are not poisoned by impurities, thereby maintaining high turnover numbers and consistent stereochemical outcomes.

How to Synthesize 2-(p-Fluorophenyl)-3-methylcrotonic Acid Efficiently
The practical implementation of this synthesis route is demonstrated through detailed examples in the patent, providing a clear roadmap for laboratory and pilot-scale production. The process begins with the formation of the Grignard reagent, typically isopropylmagnesium chloride, which is then reacted with p-fluorophenylacetic acid and acetone to generate the beta-hydroxy acid intermediate. Following aqueous workup, the crude hydroxy acid is subjected to the critical dehydration step using concentrated sulfuric acid under strict temperature control to prevent exotherms. The detailed standardized synthesis steps below outline the specific reagent quantities, addition rates, and workup procedures required to achieve the reported high yields and purity levels.
- Preparation of Hydroxy Acid Precursor: React arylacetic acid with a Grignard reagent (e.g., isopropylmagnesium chloride) and acetone to form the beta-hydroxy acid intermediate.
- Direct Dehydration: Treat the crude hydroxy acid with concentrated sulfuric acid or polyphosphoric acid at mild temperatures (0-40°C) to effect dehydration without isomerization.
- Purification: Quench the reaction mixture into ice water, filter the precipitate, and recrystallize from suitable solvents like hexane or methanol to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel dehydration technology offers substantial strategic benefits that extend beyond simple yield improvements. The elimination of multi-step protection and deprotection sequences, or the avoidance of esterification-saponification detours, translates directly into a simplified manufacturing workflow. This simplification reduces the number of unit operations required, thereby lowering the capital expenditure needed for equipment and decreasing the overall labor intensity of the production process. By streamlining the synthesis, manufacturers can respond more agilely to market demand fluctuations, ensuring a more reliable supply of critical intermediates for downstream drug production.
- Cost Reduction in Manufacturing: The direct dehydration method significantly lowers production costs by removing the need for expensive reagents and additional processing steps associated with conventional methods. By avoiding the formation of isomeric impurities, the process reduces the burden on purification systems, leading to lower solvent consumption and waste disposal costs. The ability to use crude intermediates directly without rigorous intermediate purification further drives down operational expenses, making the final API intermediate more cost-competitive in the global market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sulfuric acid, acetone, and standard arylacetic acids ensures a stable and secure raw material supply base. Unlike processes dependent on specialized or scarce catalysts for the dehydration step, this method utilizes widely available reagents that are less susceptible to supply chain disruptions. The robustness of the reaction conditions also implies a lower risk of batch failures due to minor variations in raw material quality, contributing to a more predictable and consistent delivery schedule for customers.
- Scalability and Environmental Compliance: The high concentration nature of the reaction minimizes the volume of organic solvents required, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. The suppression of decarboxylation side reactions means less gaseous waste generation and higher atom economy, which simplifies compliance with increasingly stringent environmental regulations. Furthermore, the mild temperature profile of the reaction reduces energy consumption for heating and cooling, facilitating easier scale-up from kilogram to multi-ton production scales without significant engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dehydration technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, offering clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary side reactions avoided by this new dehydration method?
A: Conventional methods using tosic acid often lead to isomerization (forming the alpha,beta-unsaturated isomer) and decarboxylation. The novel sulfuric acid method at low temperatures effectively suppresses these pathways.
Q: Can this process be scaled for industrial production of Mibefradil intermediates?
A: Yes, the process utilizes high concentrations (1 part substrate to 2-2.5 parts acid) and common reagents like sulfuric acid and Grignard reagents, making it highly suitable for large-scale commercial manufacturing.
Q: What level of optical purity can be achieved after hydrogenation?
A: The resulting unsaturated acids serve as excellent substrates for asymmetric hydrogenation using Ru or Rh catalysts, achieving enantiomeric excess (ee) values exceeding 96%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Beta-Unsaturated Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to meeting stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of alpha,beta-unsaturated acid meets the exacting standards required for subsequent asymmetric hydrogenation and API synthesis. Our expertise in managing complex chemistries allows us to optimize this specific dehydration route for maximum efficiency and safety.
We invite you to collaborate with us to explore how this advanced synthesis method can enhance your supply chain resilience and cost structure. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project goals, ensuring a partnership built on transparency and technical excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
