Revolutionizing 6α-Methylprednisolone Production: A Technical Analysis of Patented Biotransformation Routes
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value corticosteroids, and the technology disclosed in patent CN1978457B represents a significant leap forward in the production of 6α-methylprednisolone and its derivatives. This proprietary method utilizes cortisone acetate and its derivatives as the foundational starting materials, offering a streamlined alternative to traditional routes that often begin with progesterone. By strategically introducing a methine group at the 6-position and subsequently transposing it into an α-methyl group, the process achieves exceptional position specificity. The core innovation lies in the integration of Arthrobacter biotransformation to effectuate 1,2-dehydrogenation, followed by selective reduction of the 11-ketone to the 11β-hydroxyl configuration. This approach not only secures higher yields and superior quality but also addresses critical environmental and safety concerns associated with legacy manufacturing protocols.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6α-methylprednisolone has been plagued by convoluted reaction sequences that compromise both economic efficiency and operational safety. Prominent legacy methods, such as those pioneered by Upjohn, rely on progesterone as the raw material, necessitating a cumbersome series of transformations including 11-position hydroxylation, ketalization at the 3,20-positions, and peracid oxidation. These routes frequently demand the use of highly hazardous reagents such as Grignard reagents, dichromic acid, liquid bromine, and selenium dioxide. The reliance on selenium dioxide for dehydrogenation and DDQ for aromatization introduces significant toxicity risks and creates complex waste streams that are costly to treat. Furthermore, the multiplicity of steps inherently accumulates yield losses, resulting in suboptimal overall production efficiency and inconsistent product quality that fails to meet the stringent purity standards required for modern pharmaceutical applications.
The Novel Approach
In stark contrast, the methodology outlined in CN1978457B circumvents these historical bottlenecks by leveraging the structural advantages of cortisone acetate, which already possesses the requisite oxygenation at the C11 position. This strategic choice eliminates the need for difficult 11-hydroxylation steps entirely. The novel route employs mild etherification conditions to protect the 3-ketone while shifting the double bond, followed by versatile alkylation strategies to introduce the critical 6α-methyl group. Crucially, the process substitutes dangerous chemical oxidants with a highly specific microbial fermentation step using Arthrobacter simplex. This biocatalytic dehydrogenation not only enhances regioselectivity but also operates under benign aqueous conditions. The culmination of the synthesis involves a controlled reduction and deamination sequence that preserves the stereochemical integrity of the molecule, ensuring a high-purity final product suitable for sensitive therapeutic uses without the burden of heavy metal residues.
Mechanistic Insights into Arthrobacter-Catalyzed Dehydrogenation and Alkylation
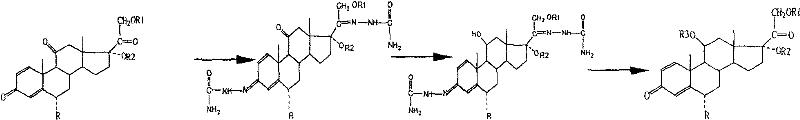
The chemical elegance of this synthesis is best understood through the detailed mechanistic progression from the protected intermediate to the final active pharmaceutical ingredient. The initial phase involves the etherification of the 3-ketone of cortisone acetate derivatives using orthoformates in an anhydrous ether solvent system catalyzed by strong acids like p-toluenesulfonic acid. This step concurrently shifts the Δ4 double bond to the Δ3,5 position, activating the molecule for subsequent alkylation. The introduction of the 6-methyl group can be achieved through three distinct pathways, providing flexibility depending on available reagents. One prominent route involves a Mannich-type reaction with N-alkylaniline and formaldehyde, followed by elimination and reduction. Another pathway utilizes halogenation with carbon tetrabromide followed by dehydrohalogenation. Each pathway is meticulously designed to minimize byproduct formation, ensuring that the steric environment around the steroid nucleus remains conducive to high-yield transformations.
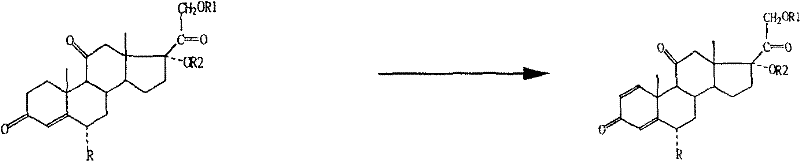
Following the chemical functionalization, the process relies on a sophisticated biotransformation step that serves as the cornerstone of its commercial viability. The intermediate compound is subjected to fermentation with Arthrobacter simplex, a microorganism selected for its ability to produce specific steroid dehydrogenases. The culture medium, optimized with corn steep liquor, glucose, and peptone, supports a conversion rate exceeding 85% within 48 hours. This enzymatic action specifically targets the 1,2-positions to introduce the conjugated diene system essential for glucocorticoid activity. The final chemical steps involve the formation of a semicarbazone at the 20-position, selective reduction of the 11-ketone using potassium borohydride, and subsequent deamination with sodium nitrite in an acidic medium. This sequence effectively restores the 11β-hydroxyl group while maintaining the newly formed 1,2-double bond and the 6α-methyl substituent, delivering the target 6α-methylprednisolone structure with high fidelity.
How to Synthesize 6α-Methylprednisolone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the patented route. The process begins with the preparation of the etherified starting material under strictly anhydrous conditions to prevent hydrolysis of the enol ether. Subsequent alkylation steps must be monitored closely to ensure complete conversion before proceeding to the fermentation stage, as residual chemical reagents can inhibit microbial activity. The biotransformation phase demands sterile technique and careful pH management to maintain enzyme activity throughout the 48-hour cycle. Finally, the workup procedures involving recrystallization from solvents like dichloromethane or acetone are critical for removing trace impurities and achieving the necessary pharmaceutical grade purity. For a comprehensive breakdown of the standardized operating procedures and specific stoichiometric ratios required for each stage, please refer to the technical guide below.
- Etherify cortisone acetate derivatives using orthoformate and anhydrous acid to shift the double bond.
- Introduce the 6-alpha methyl group via alkylation using N-alkylaniline or halogenation pathways.
- Perform microbial dehydrogenation using Arthrobacter simplex to create the 1,2-diene system.
- Complete the synthesis through semicarbazone formation, borohydride reduction, and nitrous acid deamination.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers substantial advantages that directly address the core concerns of cost management and supply chain resilience. By shifting the starting material to cortisone acetate, manufacturers can leverage a globally abundant and price-stable feedstock, mitigating the volatility often associated with specialized steroid precursors. The elimination of hazardous reagents such as selenium dioxide and liquid bromine translates into significant reductions in waste disposal costs and regulatory compliance burdens. Furthermore, the high conversion efficiency of the microbial step reduces the load on downstream purification units, lowering energy consumption and solvent usage. These factors collectively contribute to a more sustainable and economically predictable manufacturing model, allowing procurement teams to negotiate better long-term contracts with confidence in the supplier's ability to maintain consistent pricing and availability.
- Cost Reduction in Manufacturing: The streamlined synthetic route significantly lowers the cost of goods sold by reducing the total number of unit operations required to reach the final API intermediate. By avoiding expensive and dangerous oxidizing agents, the process eliminates the need for specialized corrosion-resistant equipment and extensive safety containment systems, thereby reducing capital expenditure and maintenance overheads. Additionally, the high yield of the biotransformation step minimizes raw material waste, ensuring that a greater proportion of the input cortisone acetate is converted into saleable product. This efficiency gain allows for substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final pharmaceutical formulation in price-sensitive markets.
- Enhanced Supply Chain Reliability: Dependence on readily available starting materials like cortisone acetate ensures a robust supply chain that is less susceptible to disruptions caused by the scarcity of exotic reagents. The use of fermentation technology adds a layer of scalability that chemical synthesis alone often struggles to match, as bioreactors can be easily scaled up to meet surging demand without proportional increases in complexity. Moreover, the avoidance of reagents with strict regulatory controls, such as certain brominating agents, simplifies logistics and customs clearance processes. This operational fluidity guarantees shorter lead times and more reliable delivery schedules, which is critical for pharmaceutical companies managing just-in-time inventory systems for life-saving medications.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, with fermentation parameters that are well-understood and easily controlled in large-scale industrial settings. The replacement of toxic chemical oxidants with biological catalysts aligns perfectly with modern green chemistry principles, significantly reducing the environmental footprint of the manufacturing facility. This compliance with stringent environmental regulations minimizes the risk of production shutdowns due to non-compliance issues. The simplified waste profile, characterized by biodegradable fermentation byproducts rather than heavy metal sludge, facilitates easier and cheaper waste treatment, ensuring long-term operational sustainability and protecting the manufacturer's corporate social responsibility standing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific technical disclosures within the patent documentation and aim to clarify the feasibility and benefits of adopting this technology. Understanding these details is crucial for R&D and procurement teams evaluating the potential for technology transfer or outsourcing partnerships. The answers reflect the proven capabilities of the method as described in the intellectual property, providing a factual basis for decision-making.
Q: What are the primary advantages of using cortisone acetate over progesterone for this synthesis?
A: Using cortisone acetate as the starting material significantly simplifies the synthetic route by eliminating the need for complex 11-position hydroxylation steps required when starting from progesterone. This reduces the total number of reaction steps and avoids the use of hazardous oxidizing agents typically associated with introducing oxygen functionality at the C11 position.
Q: How does the microbial dehydrogenation step impact overall process safety?
A:
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent describes fermentation conditions and purification steps that are highly amenable to scale-up. The use of readily available raw materials like cortisone acetate combined with robust microbial conversion rates exceeding 85% ensures consistent supply continuity and economic viability for industrial API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6α-Methylprednisolone Supplier
The technical potential of the CN1978457B pathway underscores the importance of partnering with a manufacturer who possesses both the chemical expertise and the infrastructure to execute complex steroid syntheses at scale. NINGBO INNO PHARMCHEM stands as a premier CDMO partner with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of this biotransformation process, including state-of-the-art fermentation suites and rigorous QC labs capable of verifying stringent purity specifications. We understand that consistency is paramount in the pharmaceutical supply chain, and our quality management systems are designed to ensure that every batch of 6α-methylprednisolone meets the highest international standards for identity, strength, and purity.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain security. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project's unique volume and timeline requirements. Let us help you engineer a more efficient and resilient supply chain for your critical corticosteroid intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
