Advanced Synthesis of Stable Ruthenium Catalyst Ligands for Industrial Hydrogenation
Advanced Synthesis of Stable Ruthenium Catalyst Ligands for Industrial Hydrogenation
The landscape of homogeneous catalysis is undergoing a significant transformation driven by the need for more robust and scalable ligand systems. Patent CN108290910B introduces a groundbreaking methodology for the production of N,N-bis(2-dialkylphosphinoethyl)amine-borane complexes, which serve as critical precursors for high-performance ruthenium catalysts. These catalysts are indispensable in the hydrogenation of carbonyl compounds and the dehydrogenation of alcohols, reactions that are foundational to the synthesis of active pharmaceutical ingredients (APIs) and fine chemicals. The core innovation lies in the stabilization of the phosphine moiety through borane coordination, coupled with a novel synthetic route utilizing oxazolidinone derivatives. This approach effectively circumvents the historical challenges associated with air-sensitive phosphines, offering a pathway to high-purity intermediates that can be reliably sourced for industrial applications. By shifting from unstable silyl-protected intermediates to air-stable borane complexes, this technology addresses critical pain points in both research and development and large-scale manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N-bis(2-dialkylphosphinoethyl)amines has been plagued by significant operational hurdles that hindered their widespread industrial adoption. Traditional methods often relied on the protection of the central amine nitrogen using trimethylsilyl groups to prevent unwanted side reactions during the introduction of phosphino groups. However, the resulting N,N-bis(2-chloroethyl)trimethylsilylamine intermediates are notoriously unstable, particularly during purification processes such as distillation where thermal decomposition frequently occurs. Furthermore, these synthetic routes typically demand extremely low temperature conditions to maintain reaction control, necessitating specialized cryogenic equipment that drives up capital expenditure and operational complexity. The inherent air sensitivity of the free phosphine ligands also requires rigorous degassing operations and inert atmosphere handling throughout the entire supply chain, increasing the risk of batch failure and compromising the consistency of the final catalyst performance. These factors collectively result in low yields, high production costs, and significant safety concerns that make conventional methods unsuitable for reliable commercial scale-up.
The Novel Approach
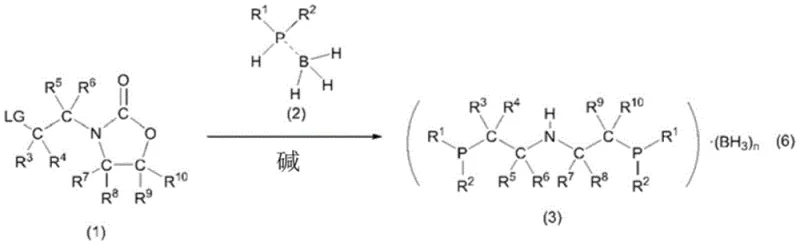
The methodology disclosed in the patent represents a paradigm shift by introducing an oxazolidinone-based synthetic strategy that fundamentally alters the stability profile of the intermediates. Instead of relying on fragile silyl protections, the process utilizes 3-(2-chloroethyl)-2-oxazolidinone, a robust cyclic carbamate that acts as an efficient electrophile for nucleophilic substitution. This compound reacts smoothly with dialkylphosphine-borane complexes in the presence of a base, such as alkyllithium reagents, to form the target N,N-bis(2-dialkylphosphinoethyl)amine-borane complexes. The presence of the borane group coordinated to the phosphorus atoms provides exceptional stability against oxidation, allowing the intermediates to be handled in air without immediate degradation. This stability translates directly into simplified purification protocols, where standard techniques like column chromatography or recrystallization can be employed effectively. The elimination of cryogenic requirements and the use of stable, isolable intermediates significantly streamline the manufacturing workflow, making the production of these sophisticated ligands feasible for reliable pharmaceutical intermediate supplier networks aiming for high throughput and consistency.
Mechanistic Insights into Oxazolidinone-Mediated Ligand Assembly
The chemical elegance of this process lies in the precise orchestration of nucleophilic substitution and coordination chemistry. The reaction initiates with the deprotonation of the dialkylphosphine-borane complex by a strong base, typically n-butyllithium, generating a highly nucleophilic phosphido-borane species. This anionic phosphorus center then attacks the electrophilic carbon adjacent to the chlorine atom in the oxazolidinone ring system. Unlike open-chain chloroamines which might undergo polymerization or elimination, the oxazolidinone structure directs the substitution cleanly, opening the pathway for the formation of the P-C bonds while maintaining the integrity of the central nitrogen framework. The borane moiety plays a dual role here: electronically, it moderates the nucleophilicity of the phosphorus to prevent over-reaction or degradation, and sterically, it shields the phosphorus lone pair from atmospheric oxygen. Following the substitution, the borane groups remain coordinated, locking the ligand in a protected state. This mechanistic pathway ensures that the final product is obtained as a stable solid, often as a mixture of di- or tri-borane adducts depending on the stoichiometry, which can be easily converted to the free ligand upon coordination with the ruthenium metal center in the subsequent step.
Impurity control is inherently built into this mechanism due to the crystallinity of the intermediates. In conventional syntheses, oily and unstable intermediates trap impurities that are difficult to remove without destructive distillation. In contrast, the borane-protected ligands described in this patent exhibit favorable crystallization properties. By selecting appropriate solvent systems, such as toluene or hexane mixtures, unwanted byproducts and unreacted starting materials can be washed away, leaving behind a high-purity lattice of the target complex. This ability to purify via recrystallization is a critical quality attribute for R&D directors focused on impurity profiles, as it ensures that the final ruthenium catalyst is free from phosphine oxides or chlorinated organic residues that could poison the catalytic cycle. The robust nature of the oxazolidinone leaving group also minimizes the formation of elimination byproducts, further enhancing the chemical purity of the stream before it ever reaches the metalation stage.
How to Synthesize N,N-Bis(2-Dialkylphosphinoethyl)amine-Borane Complex Efficiently
The practical implementation of this synthesis involves a sequence of well-defined unit operations that balance reactivity with safety. The process begins with the preparation of the oxazolidinone precursor, followed by the generation of the phosphine-borane nucleophile, and concludes with the coupling reaction. Each step is designed to maximize yield while minimizing exposure to hazardous conditions. The detailed standardized synthesis steps are outlined below to guide process chemists in replicating this high-efficiency route.
- Synthesize 3-(2-chloroethyl)-2-oxazolidinone by reacting N,N-bis(2-chloroethyl)amine hydrochloride with carbon dioxide and triethylamine in methanol.
- Prepare dialkylphosphine-borane complexes by reacting secondary phosphines with borane-THF solution under inert atmosphere.
- Perform nucleophilic substitution by reacting the oxazolidinone with lithiated phosphine-borane species in THF/hexane to yield the stable amine-borane ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage is the drastic simplification of logistics and storage requirements. Because the N,N-bis(2-dialkylphosphinoethyl)amine-borane complexes are air-stable, they do not require the expensive and cumbersome inert gas packaging typically mandated for free phosphines. This stability reduces the risk of spoilage during transit and allows for longer shelf lives, enabling companies to maintain leaner inventory levels without fear of material degradation. Furthermore, the elimination of cryogenic reaction conditions means that production can be carried out in standard glass-lined reactors available in most multipurpose chemical plants, removing the need for specialized low-temperature infrastructure. This compatibility with existing assets significantly lowers the barrier to entry for contract manufacturing organizations looking to offer these high-value intermediates.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the removal of costly reagents and energy-intensive steps. Traditional routes rely on expensive silylating agents like trimethylsilyl chloride and require significant energy input to maintain sub-zero temperatures over extended periods. By replacing these with readily available carbon dioxide and operating at near-ambient temperatures, the variable cost of goods sold is substantially reduced. Additionally, the ability to purify the product via recrystallization rather than complex chromatographic separations or risky distillations lowers solvent consumption and waste disposal costs. The higher isolated yields reported in the patent examples further contribute to cost efficiency by maximizing the output per batch, ensuring that raw material investments are fully realized in the final product.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the sensitivity of key intermediates. The air-stable nature of the borane complexes mitigates the risk of batch rejection due to accidental exposure to oxygen during handling or sampling. This robustness allows for more flexible scheduling and reduces the dependency on highly specialized operators who are trained exclusively in air-free techniques. Moreover, the starting materials, such as secondary phosphines and chloroethyl amines, are commodity chemicals with established supply chains, reducing the risk of raw material shortages. This reliability makes the technology an attractive option for securing long-term supply agreements for critical catalyst components used in continuous manufacturing processes.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this route is inherently designed for expansion. The reaction exotherms are manageable, and the solvents used, such as THF and toluene, are standard industrial solvents with well-established recovery and recycling protocols. The avoidance of silicon-based byproducts simplifies waste treatment, as siloxanes can be difficult to degrade in wastewater streams. The overall atom economy is improved by the direct coupling strategy, which generates fewer stoichiometric byproducts compared to multi-step protection-deprotection sequences. This alignment with green chemistry principles facilitates regulatory approval and supports corporate sustainability goals, making it a preferred choice for environmentally conscious manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: Why is the borane protection strategy superior to silyl protection for PNP ligands?
A: Unlike silyl-protected intermediates which decompose during distillation and require extreme low temperatures, borane-protected phosphines are air-stable solids that can be purified via simple recrystallization, drastically improving handling safety and yield.
Q: What are the key advantages of the oxazolidinone route for large-scale production?
A: The oxazolidinone route eliminates the need for expensive silylating agents and cryogenic conditions. It utilizes stable starting materials and allows for purification through crystallization, making it highly suitable for multi-kilogram commercial scale-up.
Q: Can this ligand system be adapted for different alkyl groups on phosphorus?
A: Yes, the patent demonstrates successful synthesis with various dialkylphosphine variants including dicyclohexyl, di-tert-butyl, and diisopropyl groups, allowing for fine-tuning of steric and electronic properties for specific catalytic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N-Bis(2-Dialkylphosphinoethyl)amine-Borane Complex Supplier
The technological potential of the N,N-bis(2-dialkylphosphinoethyl)amine-borane complex system is immense, offering a pathway to more efficient and sustainable catalytic processes for the global pharmaceutical industry. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to tonnage manufacturing is seamless and robust. We understand that the efficacy of a ruthenium catalyst is entirely dependent on the purity of its ligand; therefore, our stringent purity specifications and rigorous QC labs are dedicated to delivering intermediates that meet the highest standards of quality required for GMP-grade API synthesis.
We invite you to collaborate with us to optimize your catalyst supply chain. Whether you are looking to reduce costs in catalyst manufacturing or secure a stable source of high-purity ligands, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this advanced synthesis method can be integrated into your existing production workflows.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
