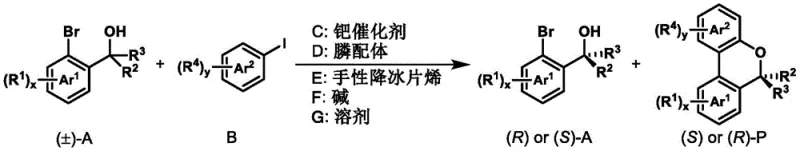
Advanced Kinetic Resolution Strategy for Commercializing Chiral Benzopyran Intermediates
The landscape of asymmetric synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for higher enantiomeric purity and more efficient processes. A significant breakthrough in this domain is documented in patent CN113087691A, which discloses a novel method for synthesizing chiral aryl tertiary alcohols and benzopyran compounds based on a kinetic resolution strategy. This technology addresses a long-standing challenge in organic chemistry: the efficient kinetic resolution of sterically hindered tertiary alcohols, which are critical structural motifs in numerous bioactive molecules such as tramadol, efinaconazole, and voriconazole. By leveraging a sophisticated palladium-catalyzed system involving a chiral norbornene derivative, this method achieves exceptional enantioselectivity and a high selectivity factor, offering a robust pathway for producing high-value chiral building blocks. The ability to simultaneously generate a chiral benzopyran product and recover unreacted chiral tertiary alcohol with high optical purity represents a dual-value proposition for process chemists aiming to optimize atom economy and reduce waste in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral tertiary alcohols has relied on methods that often suffer from significant drawbacks regarding substrate scope and operational complexity. Conventional kinetic resolution strategies frequently employ Lewis base-catalyzed acylation or Bronsted acid-catalyzed acetalization, which can be limited by the steric bulk of the tertiary carbon center. The three non-hydrogen substituents on the alcoholic carbon create a crowded environment that hinders the approach of chiral catalysts, often resulting in low reaction rates and poor selectivity factors. Furthermore, many existing methods require harsh reaction conditions or expensive, difficult-to-prepare chiral resolving agents that are not readily available on a commercial scale. These limitations translate directly into higher production costs and longer development timelines for pharmaceutical companies, as multiple recrystallization steps are often needed to upgrade the enantiomeric excess to acceptable levels for drug substance manufacturing. The inability to efficiently resolve tertiary alcohols has historically been a bottleneck in the supply chain for various central nervous system and antifungal agents.
The Novel Approach
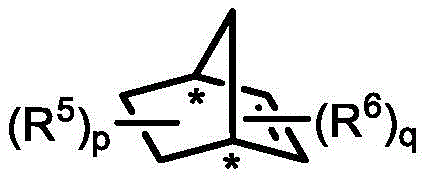
In stark contrast to these traditional limitations, the method disclosed in CN113087691A introduces a transformative palladium-catalyzed C-H activation and C-O coupling sequence that bypasses the steric hurdles of conventional resolution. This novel approach utilizes a synergistic catalytic system comprising a palladium source, a phosphine ligand, and a chiral norbornene derivative to facilitate the reaction under relatively mild thermal conditions. The integration of the chiral norbornene mediator allows for precise stereochemical control during the formation of the new carbon-oxygen bond, leading to the construction of the benzopyran scaffold with high fidelity. This strategy not only resolves the racemic starting material but also constructs a complex heterocyclic ring system in a single operation, effectively telescoping two synthetic steps into one. The result is a streamlined process that delivers both the desired cyclic product and the recovered linear alcohol with excellent yields and enantiomeric ratios, significantly simplifying the downstream purification workflow and enhancing the overall economic viability of the synthesis.
Mechanistic Insights into Pd-Catalyzed Kinetic Resolution
The core of this technological advancement lies in the intricate catalytic cycle mediated by the palladium-norbornene cooperative system. The reaction initiates with the oxidative addition of the aryl iodide to the palladium(0) species, generating an aryl-palladium(II) intermediate. This species then undergoes a crucial norbornene insertion, forming a key alkyl-palladium complex that activates the ortho-C-H bond of the aryl ring. The chiral information is transferred from the norbornene derivative to the substrate during the subsequent coordination and insertion of the tertiary alcohol. The steric environment created by the chiral backbone of the norbornene derivative differentiates between the two enantiomers of the racemic tertiary alcohol, allowing one enantiomer to react much faster than the other. This differential reaction rate is the essence of kinetic resolution, leading to the preferential formation of the (S)-benzopyran product while leaving the (R)-tertiary alcohol largely unreacted. The cycle concludes with a reductive elimination step that releases the final benzopyran product and regenerates the active palladium(0) catalyst, ready to enter another turnover.
Controlling the impurity profile in such complex transformations is paramount for pharmaceutical applications, and this mechanism offers inherent advantages in that regard. The high selectivity factor, reported to be as high as 544 in optimized examples, ensures that cross-reactivity between the wrong enantiomers is minimized, thereby reducing the formation of diastereomeric impurities that are notoriously difficult to separate. The use of specific ligands like tris(2-furyl)phosphine further tunes the electronic properties of the palladium center, suppressing side reactions such as homocoupling of the aryl iodide or beta-hydride elimination. Moreover, the reaction conditions, typically employing potassium carbonate in toluene at 105 °C, are compatible with a wide range of functional groups including halogens, esters, and ethers. This functional group tolerance means that protecting group manipulations can often be avoided, which not only reduces the number of synthetic steps but also minimizes the generation of chemical waste associated with protection and deprotection sequences, aligning the process with green chemistry principles.
How to Synthesize Chiral Benzopyran Efficiently
Implementing this kinetic resolution strategy in a laboratory or pilot plant setting requires careful attention to the stoichiometry and quality of the catalytic components. The standard protocol involves charging a reaction vessel with the racemic aryl tertiary alcohol and the aryl iodide coupling partner in a molar ratio that favors the resolution efficiency, typically around 1.0 to 0.8 equivalents respectively. To this mixture, the palladium catalyst, phosphine ligand, and the chiral norbornene derivative are added along with the inorganic base. The choice of solvent is critical, with toluene being the preferred medium due to its ability to solubilize the organic substrates while maintaining the stability of the catalytic species at elevated temperatures. The detailed standardized synthesis steps, including specific workup procedures and purification parameters to achieve the reported >99% ee, are outlined in the guide below.
- Prepare the reaction mixture by combining racemic aryl tertiary alcohol, aryl iodide, palladium catalyst, phosphine ligand, chiral norbornene derivative, and base in an organic solvent under inert gas.
- Heat the reaction mixture to a temperature between 50-130°C, preferably 105°C, and stir for 5 to 72 hours to allow the kinetic resolution and cyclization to proceed.
- Upon completion, filter the mixture, concentrate under reduced pressure, and purify the crude product via column chromatography to isolate the chiral benzopyran and recovered alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this kinetic resolution technology offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the accessibility of the starting materials; aryl iodides are commoditized reagents available from numerous global suppliers, ensuring a stable and competitive supply chain. Unlike methods that rely on bespoke chiral auxiliaries which may have long lead times, the chiral norbornene derivatives used here can be sourced or synthesized in bulk, mitigating the risk of raw material shortages. Furthermore, the robustness of the reaction conditions allows for flexibility in manufacturing locations, as the process does not require cryogenic temperatures or ultra-high pressure equipment, thereby reducing capital expenditure requirements for facility upgrades. This operational simplicity translates directly into cost reduction in pharmaceutical intermediate manufacturing by lowering energy consumption and minimizing the need for specialized reactor hardware.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by its high atom economy and the dual output of valuable products. By converting a racemic mixture into two distinct chiral entities—the benzopyran and the recovered alcohol—the process effectively doubles the utility of the starting material compared to resolutions that discard one enantiomer. The elimination of expensive transition metal scavengers is another potential cost saver, as the palladium loading is kept relatively low (2.5 mol%) and the heterogeneous workup allows for easy metal removal. Additionally, the high selectivity reduces the burden on downstream purification, meaning less silica gel and solvent are consumed during column chromatography or crystallization, leading to substantial cost savings in waste disposal and solvent recovery operations.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the broad substrate scope of this methodology. The ability to tolerate diverse substituents on both the alcohol and the iodide components means that a single platform technology can be applied to synthesize a library of different intermediates. This versatility reduces the dependency on multiple, specialized synthetic routes for different drug candidates, simplifying inventory management and vendor qualification processes. In the event of a supply disruption for one specific precursor, the flexibility of the chemistry may allow for rapid adaptation to alternative analogues without necessitating a complete process redevelopment. This adaptability ensures reducing lead time for high-purity chiral alcohols and maintains continuity of supply for critical drug programs.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the use of toluene as a solvent and potassium carbonate as a base aligns well with established industrial safety protocols. The reaction generates minimal hazardous byproducts, primarily inorganic salts, which are easy to handle and dispose of in compliance with environmental regulations. The thermal stability of the reaction mixture at 105 °C suggests that the process can be safely scaled from gram to kilogram quantities without exothermic runaway risks. This scalability is crucial for meeting the demands of commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to ramp up production volumes seamlessly as a drug candidate progresses through clinical trials to market launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this kinetic resolution technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic expectation of performance and applicability for process development teams. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing pipelines.
Q: What is the selectivity factor achieved in this kinetic resolution method?
A: The method described in patent CN113087691A achieves a selectivity factor as high as 544, indicating exceptional efficiency in distinguishing between enantiomers of the tertiary alcohol substrate.
Q: Which catalyst system is preferred for this transformation?
A: The preferred catalyst system utilizes Palladium Acetate (Pd(OAc)2) as the metal source, Tris(2-furyl)phosphine (TFP) as the ligand, and a chiral norbornene derivative as the co-catalyst to induce asymmetry.
Q: Can this method be scaled for industrial production of API intermediates?
A: Yes, the process uses commercially available aryl iodides and operates at moderate temperatures (105°C) in common solvents like toluene, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Benzopyran Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing advanced synthetic methodologies like the one described in CN113087691A to maintain a competitive edge in the pharmaceutical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is smooth and efficient. We are committed to delivering high-purity chiral building blocks that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific benzopyran derivatives or the recovery of chiral tertiary alcohols, our infrastructure is designed to support your most demanding projects with reliability and precision.
We invite you to collaborate with us to explore how this kinetic resolution strategy can optimize your supply chain and reduce your overall cost of goods. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, helping you identify opportunities for efficiency gains. We encourage you to reach out to request specific COA data and route feasibility assessments for your target compounds. By partnering with us, you gain access to a wealth of chemical expertise and manufacturing capacity dedicated to accelerating your drug development timeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
