Scalable Production of 2-Hydroxy-5-phenylalkylaminobenzoic Acid Derivatives via Mesylate Chemistry
The pharmaceutical industry continuously seeks robust manufacturing pathways for central nervous system (CNS) therapeutics, particularly for treating complex conditions such as ischemia, Alzheimer's disease, and traumatic brain injuries. A pivotal advancement in this domain is documented in patent CN101874016A, which discloses a highly efficient production method for 2-hydroxy-5-phenylalkylaminobenzoic acid derivatives and their salts. This technology addresses critical bottlenecks in the synthesis of these bioactive scaffolds, specifically targeting the production of 2-hydroxy-5-[2-(4-trifluoromethylphenyl)ethylamino]benzoic acid. As a reliable pharmaceutical intermediates supplier, understanding the nuances of this patented route is essential for ensuring supply chain continuity and product quality. The innovation lies not merely in the final structure but in the strategic selection of leaving groups and purification protocols that enable true commercial scale-up.
This technical insight report analyzes the transformative potential of the mesylate-based synthetic route described in the patent. By shifting away from traditional halide-based alkylation methods, the process mitigates the formation of persistent organic impurities that have historically plagued the quality profile of these intermediates. For R&D directors and procurement managers alike, this represents a significant opportunity for cost reduction in pharmaceutical intermediates manufacturing. The ability to produce high-purity [精准的行业名词] at scale without extensive chromatographic purification translates directly into improved margins and faster time-to-market for downstream drug development projects.
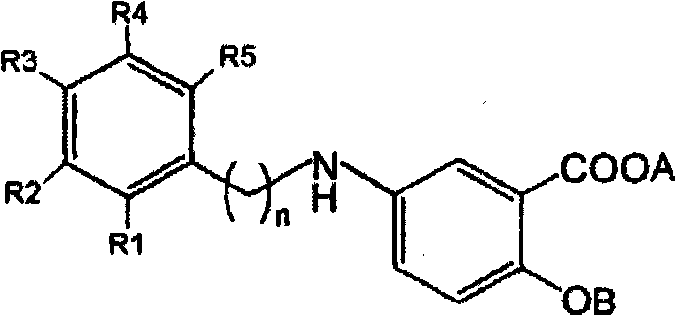
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-hydroxy-5-(substituted)phenylalkylaminobenzoic acids relied heavily on nucleophilic substitution reactions using alkyl halides, such as 2-(4-nitrophenyl)ethyl bromide. While conceptually straightforward, this conventional approach suffers from severe chemical liabilities when attempted on an industrial scale. The primary issue arises from the dual functionality of the starting material, 5-aminosalicylic acid, where the amine moiety can inadvertently act as a base rather than a nucleophile. Under typical reaction conditions, this basicity promotes an elimination reaction of the alkyl halide starting material, leading to the formation of olefinic impurities like 4-nitrostyrene or 1-allyl-4-nitrobenzene.
These elimination byproducts are chemically similar to the desired product and possess solubility profiles that make them notoriously difficult to remove via standard recrystallization techniques. Consequently, manufacturers often face yield losses and extended processing times as they attempt to scrub these impurities from the crude mixture. Furthermore, the use of bromide leaving groups can introduce trace metal contaminants or require expensive scavenging steps to meet regulatory standards for pharmaceutical ingredients. The cumulative effect of these inefficiencies renders the conventional bromide-based route unsuitable for the mass production required by the global market for CNS therapeutics.
The Novel Approach
The methodology outlined in CN101874016A introduces a paradigm shift by utilizing phenylalkyl methanesulfonates (mesylates) as the electrophilic coupling partner instead of alkyl bromides. This strategic substitution fundamentally alters the reaction kinetics and thermodynamics, favoring the desired nucleophilic attack over the competing elimination pathway. The mesylate group is an exceptional leaving group that facilitates smooth displacement by the amine of the 5-aminosalicylic acid alkyl ester, even under milder conditions.
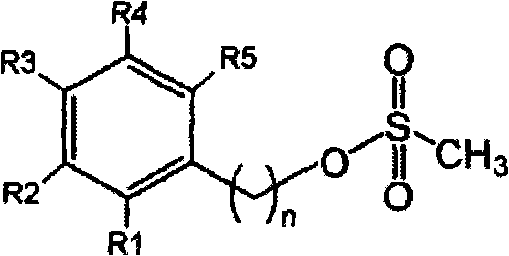
By employing this novel approach, the formation of nitrostyrene-type impurities is drastically suppressed, resulting in a much cleaner reaction profile. The process utilizes alkyl 5-aminosalicylates, where the carboxylic acid is protected as an ester, preventing unwanted side reactions at the acid site during the alkylation step. This protection-deprotection strategy, combined with the superior reactivity of the mesylate, ensures that the substitution proceeds with high fidelity. The result is a robust synthetic sequence that is inherently safer and more predictable, providing a solid foundation for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Mesylate-Mediated Nucleophilic Substitution
The core of this technological breakthrough lies in the mechanistic advantages of the mesylate leaving group within the specific steric and electronic environment of the reaction. In the condensation step, the nitrogen atom of the alkyl 5-aminosalicylate attacks the benzylic carbon of the phenylalkyl methanesulfonate. Unlike bromide ions, which can sometimes participate in reversible equilibria or promote E2 elimination due to their size and basicity of the conjugate acid, the mesylate anion is a very weak base and a stable leaving group. This stability drives the equilibrium forward towards the formation of the C-N bond, effectively outcompeting the beta-elimination pathway that generates olefinic impurities.
Furthermore, the reaction conditions specified in the patent, typically ranging from 60°C to 90°C with triethylamine as a base, are optimized to balance reaction rate with selectivity. Temperatures exceeding this range accelerate side reactions, while lower temperatures result in impractically long cycle times. The use of toluene as a solvent provides an ideal medium for azeotropic removal of water if necessary and ensures good solubility of the organic reactants. Following the condensation, the intermediate ester undergoes hydrolysis in a mixture of sulfuric acid, water, and acetic acid. This acidic hydrolysis cleaves the ester protecting group to reveal the free carboxylic acid, simultaneously forming a sulfate salt which aids in the initial isolation of the product from the reaction matrix.
Impurity control is further refined during the purification stage through precise pH manipulation. The intermediate sulfate salt possesses both amino and carboxyl functionalities, making it amphoteric. By controlling the pH of the aqueous solution to a narrow window between 3.0 and 3.5 during the neutralization step, the process exploits the isoelectric point of the molecule. At this specific pH, the zwitterionic form of the 2-hydroxy-5-phenylalkylaminobenzoic acid has minimal solubility and precipitates out of the solution, while charged ionic impurities and residual salts remain dissolved in the mother liquor. This elegant physicochemical separation technique eliminates the need for column chromatography, which is a critical factor for reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize 2-Hydroxy-5-phenylalkylaminobenzoic Acid Efficiently
The synthesis protocol described in the patent offers a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety. The process begins with the condensation of the mesylate and the aminosalicylate ester in toluene, followed by a workup that involves acidification to isolate the intermediate ester sulfate. This intermediate is then subjected to hydrolysis under reflux conditions to generate the final acid sulfate salt. The final step involves a careful pH adjustment to precipitate the pure free acid.
- Condense phenylalkyl methanesulfonate with alkyl 5-aminosalicylate in toluene with triethylamine at 70-80°C.
- Hydrolyze the resulting ester intermediate using a mixture of sulfuric acid, water, and acetic acid at 95-100°C.
- Purify the sulfate salt by dissolution and subsequent neutralization to pH 3.0-3.5 to precipitate the final free acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this mesylate-based route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage is the significant simplification of the purification train. By minimizing the generation of hard-to-remove elimination byproducts, the process reduces the number of recrystallization cycles required to meet specification. This reduction in unit operations directly correlates to lower manufacturing costs, as it decreases solvent consumption, energy usage for heating and cooling, and labor hours associated with filtration and drying.
- Cost Reduction in Manufacturing: The elimination of expensive and time-consuming purification steps, such as preparative chromatography or multiple iterative recrystallizations, leads to substantial cost savings. The use of readily available starting materials like methanesulfonic acid derivatives and common solvents like toluene and ethanol further optimizes the bill of materials. Additionally, the higher selectivity of the reaction improves the overall yield of the active pharmaceutical ingredient precursor, maximizing the output per batch and reducing the cost of goods sold.
- Enhanced Supply Chain Reliability: The robustness of the mesylate chemistry ensures consistent batch-to-batch quality, which is vital for maintaining regulatory compliance and avoiding production delays. The starting materials are commercially accessible and do not rely on exotic reagents that might be subject to supply shortages. This reliability allows for better production planning and inventory management, ensuring that downstream drug manufacturers receive their intermediates on schedule without unexpected interruptions caused by failed batches or quality deviations.
- Scalability and Environmental Compliance: The process has been demonstrated to be scalable, with the patent detailing operations in 50L reactors, indicating readiness for transfer to larger commercial vessels. The solvent system is relatively benign compared to chlorinated alternatives often used in older methodologies, facilitating easier waste treatment and disposal. The ability to recycle solvents like toluene and ethanol further enhances the environmental profile of the manufacturing process, aligning with modern green chemistry principles and reducing the environmental footprint of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for process evaluation.
Q: Why is the mesylate leaving group preferred over bromide in this synthesis?
A: Using a bromide leaving group often leads to elimination side reactions where the amine moiety acts as a base, generating difficult-to-remove impurities like nitrostyrene. The mesylate group facilitates a cleaner nucleophilic substitution with significantly reduced elimination byproducts.
Q: What are the critical parameters for the hydrolysis step?
A: The hydrolysis requires a specific solvent system of sulfuric acid, water, and acetic acid at temperatures between 95-100°C. Maintaining this temperature range ensures complete conversion of the ester to the acid without degrading the sensitive amino functionality.
Q: How is high purity achieved in the final isolation?
A: High purity is achieved through a precise pH control strategy. The crude product is isolated as a sulfate salt, dissolved, and then carefully neutralized to a pH of 3.0-3.5. This specific pH window allows the zwitterionic free acid to precipitate while keeping ionic impurities in solution.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy-5-phenylalkylaminobenzoic Acid Derivatives Supplier
The synthesis of 2-hydroxy-5-phenylalkylaminobenzoic acid derivatives represents a sophisticated challenge in organic process chemistry, requiring precise control over reaction parameters and purification strategies. NINGBO INNO PHARMCHEM stands ready to support your development needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team of expert chemists is well-versed in the nuances of mesylate chemistry and pH-controlled crystallizations, ensuring that we can deliver these critical CNS intermediates with stringent purity specifications. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for these vital therapeutic building blocks. By leveraging our technical expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of 2-hydroxy-5-phenylalkylaminobenzoic acid derivatives to accelerate your drug development pipeline.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
