Advanced Manufacturing of Electronic Grade Triglycidyl Isocyanurate for High-Performance Applications
The global demand for high-performance electronic materials has necessitated a paradigm shift in the synthesis of key curing agents, specifically focusing on the production of electronic grade triglycidyl isocyanurate (TGIC). As detailed in the pioneering patent CN102174040B, a novel preparation method has been developed that addresses the critical limitations of conventional manufacturing processes, effectively elevating product purity from standard levels of approximately 92% to an exceptional electronic grade exceeding 99%. This technological breakthrough is particularly significant for industries requiring ultra-high purity standards, such as the production of electronic inks, sealants for photosensitive materials, and photoimaging solder resist inks, where even trace impurities can compromise the performance of the final electronic assembly. By leveraging a sophisticated combination of phase transfer catalysis and advanced separation technologies, this method not only enhances chemical purity but also drastically reduces chlorine content, thereby meeting the stringent specifications demanded by top-tier multinational electronics manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of TGIC has relied on traditional two-step methods that suffer from inherent inefficiencies and quality control challenges, particularly during the critical cyclization stage. In these legacy processes, the management of water generated during the reaction is notoriously difficult, often leading to intensified side reactions that introduce unwanted by-products and degrade the overall quality of the final compound. Furthermore, prior art methods, such as those described in Japanese Patent JP-B-45-22751 and US Patent 4,395,542, typically employ water washing or multi-stage stripping techniques that result in relatively low yields and fail to achieve the high purity levels required for modern electronic applications. These conventional approaches often leave behind significant amounts of unreacted epichlorohydrin and chlorinated impurities, rendering the product unsuitable for high-end uses like solder resist inks, and they are generally ill-suited for large-scale industrial production due to their inability to consistently control reaction parameters and impurity profiles.
The Novel Approach
In stark contrast to these outdated techniques, the method disclosed in patent CN102174040B introduces a refined synthetic route that optimizes every stage of the reaction to ensure maximum conversion and minimal impurity formation. The process begins with a precise addition of epichlorohydrin, cyanuric acid, a quaternary ammonium salt phase transfer catalyst, and a controlled amount of water, heated to a specific range of 80-120°C to facilitate the initial synthesis of the intermediate compound. This careful control of reactant ratios, specifically maintaining a cyanuric acid to epichlorohydrin molar ratio between 1:11 and 1:17, ensures that the reaction proceeds efficiently towards the desired intermediate without excessive side reactions. The resulting mixture gradually becomes clear and transparent, indicating a high degree of conversion and setting the stage for the subsequent cyclization step, which is the cornerstone of achieving electronic grade purity.
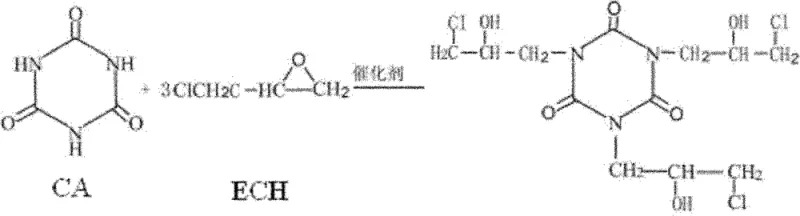
Mechanistic Insights into Phase Transfer Catalyzed Cyclization
The core of this innovative synthesis lies in the sophisticated application of phase transfer catalysis combined with a controlled alkaline cyclization process that effectively drives the formation of the epoxide rings while minimizing hydrolysis. In the second stage of the reaction, caustic soda is introduced to the system at a strictly controlled low temperature of 15-20°C, maintaining a molar ratio of caustic soda to cyanuric acid between 1:3 and 1:3.5 to ensure complete dehydrochlorination. This low-temperature environment is crucial as it suppresses the hydrolysis of the newly formed epoxide groups, a common side reaction in traditional high-temperature processes that leads to the formation of diols and a subsequent drop in epoxy equivalent weight. The use of specific quaternary ammonium salts, such as tetramethylammonium chloride or benzyltriethylammonium chloride, facilitates the transport of hydroxide ions into the organic phase, accelerating the cyclization rate and ensuring a more thorough removal of chlorine atoms, which is evidenced by the significantly lower chlorine content in the final cyclization liquid.
Following the cyclization, the purification strategy employs a dual-distillation approach that is critical for preserving the thermal stability of the TGIC molecule. After the solid salts are removed via pressure filtration, the crude product undergoes reduced pressure distillation followed immediately by high-vacuum thin-film evaporation to strip away unreacted epichlorohydrin. Unlike traditional kettle distillation where the material is subjected to heat for prolonged periods, the thin-film evaporator exposes the liquid to heat for only a few seconds, drastically reducing the risk of thermal decomposition which can otherwise lead to discoloration and polymerization of the product. This mechanistic understanding of thermal sensitivity allows for the production of a crude product that retains its structural integrity, ready for the final crystallization and cryogenic pulverization steps that define the physical properties of the electronic grade material.
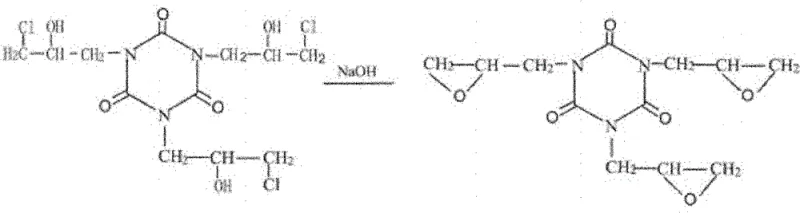
How to Synthesize Electronic Grade Triglycidyl Isocyanurate Efficiently
To replicate the high-purity results demonstrated in the patent examples, operators must adhere to a rigorous protocol that balances reaction kinetics with thermal management throughout the synthesis and purification phases. The process requires precise metering of the phase transfer catalyst, typically between 4-10% of the cyanuric acid mass, and strict temperature control during both the initial synthesis at 80-120°C and the subsequent cyclization at 15-20°C to prevent runaway exotherms. The detailed standardized synthesis steps involve specific timing windows, such as stirring the initial mixture for 4-6 hours and allowing the cyclization to proceed for 100-150 minutes, ensuring that the reaction reaches completion before moving to the separation stages. For a comprehensive breakdown of the operational parameters and safety considerations required for this synthesis, please refer to the standardized guide below.
- Mix cyanuric acid, epichlorohydrin, quaternary ammonium salt catalyst, and water, then heat to 80-120°C for 4-6 hours to form the intermediate.
- Add caustic soda to the reaction system at 15-20°C for cyclization, maintaining a molar ratio of 1: 3 to 1:3.5, followed by pressure filtration to remove solid salts.
- Purify the crude product via vacuum distillation and high-vacuum thin-film evaporation, followed by crystallization, centrifugation, cryogenic pulverization, and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced TGIC manufacturing process represents a strategic opportunity to secure a more reliable and cost-effective supply of critical electronic chemicals. By eliminating the inefficiencies associated with traditional water-washing and multi-stage stripping methods, the new process significantly streamlines the production workflow, reducing the overall processing time and energy consumption required to achieve electronic grade specifications. The ability to consistently produce material with purity levels exceeding 99% means that downstream customers in the electronics sector can reduce their own quality control burdens and rejection rates, creating a value chain that is more resilient to market fluctuations and quality disputes. Furthermore, the robust nature of the phase transfer catalytic system allows for greater flexibility in raw material sourcing, as the process is less sensitive to minor variations in feedstock quality compared to older, more fragile synthetic routes.
- Cost Reduction in Manufacturing: The implementation of high-vacuum thin-film evaporation and cryogenic pulverization creates substantial operational efficiencies that translate directly into lower manufacturing costs. By recovering unreacted epichlorohydrin more effectively through thin-film technology, the process minimizes raw material waste, while the cryogenic grinding technique prevents product loss due to caking and wall adhesion, ensuring higher overall yields. Additionally, the elimination of complex water-washing sequences reduces the volume of wastewater generated, lowering the costs associated with effluent treatment and environmental compliance, which is a significant factor in the total cost of ownership for chemical manufacturing facilities.
- Enhanced Supply Chain Reliability: The scalability of this synthesis method offers a distinct advantage in maintaining continuous supply lines for high-volume electronic manufacturing. Because the reaction conditions are tightly controlled and the purification steps are designed for continuous operation via thin-film evaporation, the process is highly amenable to scale-up from pilot batches to multi-ton annual production capacities without sacrificing product consistency. This reliability ensures that buyers can depend on steady deliveries of electronic grade TGIC, mitigating the risks of production stoppages caused by quality failures or batch-to-batch variability that often plague suppliers using outdated technology.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process offers a cleaner production profile that aligns with increasingly strict global sustainability standards. The thorough removal of chlorine and the efficient recycling of solvents mean that the final product meets rigorous international specifications for halogen content, facilitating easier registration and approval in regulated markets like the EU and North America. Moreover, the reduced generation of saline wastewater and the minimized thermal degradation of products contribute to a smaller environmental footprint, making this method a future-proof choice for companies aiming to enhance their corporate social responsibility profiles while maintaining competitive production costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of electronic grade TGIC, derived directly from the specific innovations and data presented in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this material into their formulations or for procurement specialists assessing the capability of potential suppliers. The answers provided reflect the specific advantages of the phase transfer catalysis and thin-film evaporation methodology, highlighting why this approach is superior to conventional synthesis routes for high-end electronic applications.
Q: How does this method achieve electronic grade purity compared to traditional methods?
A: Traditional methods struggle with water control during cyclization, leading to side reactions and lower purity (around 92%). This novel method utilizes a specific phase transfer catalyst system and high-vacuum thin-film evaporation to effectively remove impurities and unreacted epichlorohydrin, pushing purity above 99%.
Q: What is the role of cryogenic pulverization in the final processing step?
A: Standard pulverization generates friction heat that can cause the TGIC product to melt and cake on equipment walls, leading to material loss. Cryogenic pulverization at 0°C or below prevents this thermal aggregation, ensuring high recovery rates and consistent particle size distribution.
Q: Why is thin-film evaporation critical for TGIC production?
A: TGIC is thermally sensitive. Traditional kettle distillation exposes the material to heat for extended periods, causing decomposition. Thin-film evaporation reduces the heating time to mere seconds, significantly minimizing thermal degradation and preserving product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Electronic Grade Triglycidyl Isocyanurate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to electronic grade materials requires a partner with deep technical expertise and a proven track record in scaling complex chemical pathways. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated requirements of the TGIC synthesis process, such as precise temperature control and thin-film evaporation, are executed with absolute precision. We maintain stringent purity specifications across all our production lines, supported by rigorous QC labs equipped to analyze trace impurities and epoxy equivalents, guaranteeing that every batch of TGIC we supply meets the exacting standards required for electronic inks and solder resist applications.
We invite you to collaborate with us to explore how this advanced manufacturing technology can optimize your supply chain and reduce your overall material costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our electronic grade TGIC can enhance the performance and reliability of your final electronic products.
