Advanced Synthesis of N-[4-[(4-Hydroxyanthraquinone-1-yl) Amino] Benzyl] Acetamide for Industrial Dye Applications
The global demand for high-performance solvent dyes, particularly those utilized in engineering plastics and polyester spinning, necessitates manufacturing processes that guarantee exceptional purity and consistent chromatic properties. Patent CN102050757B, published in September 2013, introduces a transformative methodology for synthesizing N-[4-[(4-hydroxyanthraquinone-1-yl) amino] benzyl] acetamide, commonly known as Solvent Blue 19, which addresses critical deficiencies in prior art technologies. This technical disclosure outlines a robust two-step synthetic pathway that leverages hydrochloric acid catalysis within a butanol-water solvent system to achieve product purities ranging from 95% to 99%. By shifting away from traditional boron-based catalysts and scarce chlorinated starting materials, this innovation provides a scalable solution for producing dye intermediates with minimal ash content and superior compatibility with PET resin matrices. For industrial stakeholders, this represents a pivotal advancement in reliable solvent dye intermediate supplier capabilities, ensuring that downstream textile and plastic applications meet rigorous quality standards without the risk of filter pressure spikes or fiber breakage during spinning operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing landscape for anthraquinone-based solvent dyes has been plagued by two distinct synthetic routes, both of which suffer from significant technical and economic drawbacks that hinder large-scale efficiency. The first conventional method relies on the condensation of 1,4-dihydroxyanthraquinone with p-aminoacetanilide using boric acid as a catalyst, a process that frequently yields products with inconsistent content ranging widely from 79% to 97% and generates substantial quantities of difficult-to-remove byproducts. A major failure point in this legacy technology is the elevated ash content, often exceeding 0.5%, which arises from the complex post-reaction treatments required to remove residual 1,4-dihydroxyanthraquinone using multiple hot water washes that inadvertently form sodium salts. These impurities lead to unstable dye shades and poor compatibility with polyester resins, causing severe operational issues such as high filter pressure and filament breakage during the spinning process. The second traditional approach utilizes 1-hydroxy-4-chloroanthraquinone as a starting material, but this route is fundamentally flawed due to the lack of large-scale production availability for the chlorinated precursor and its prohibitively high market price. Consequently, these conventional methodologies fail to offer the suitability for industrialized production required by modern chemical supply chains, creating a bottleneck for manufacturers seeking cost reduction in dye manufacturing.
The Novel Approach
In stark contrast to these legacy systems, the novel approach detailed in the patent employs a sophisticated condensation reaction between 1,4-dihydroxyanthraquinone, its corresponding leuco body, and p-phenylenediamine (Ursol D) within a specifically optimized butanol-water medium. This innovative strategy utilizes hydrochloric acid as a highly effective catalyst, operating at a controlled temperature range of 95°C to 105°C for a duration of 10 to 16 hours to ensure complete conversion while minimizing thermal degradation. The process is distinguished by its precise stoichiometric control, maintaining a molar ratio of 1,4-dihydroxyanthraquinone to its leuco compound between 3:1 and 4:1, which is critical for driving the reaction to completion and suppressing the formation of bis-acetamido byproducts. Following the condensation phase, the protocol incorporates a streamlined workup procedure involving water addition, distillation to recover butanol, and a targeted alkaline treatment that effectively removes unreacted quinones without generating excessive salt waste. The subsequent acylation step, conducted with acetic anhydride in an acetic acid medium at 110°C to 112°C, finalizes the molecular structure with high fidelity, resulting in a product with ash content reduced to ≤0.2% and exceptional color stability suitable for high-end polyester applications.
Mechanistic Insights into HCl-Catalyzed Condensation and Acylation
The core chemical transformation in this synthesis relies on an electrophilic aromatic substitution mechanism facilitated by the protonation of the anthraquinone carbonyl groups by hydrochloric acid, which enhances the electrophilicity of the quinone ring system. The inclusion of the 1,4-dihydroxyanthraquinone leuco body serves a dual purpose: it acts as a reducing agent to maintain the stability of the reaction environment and improves the solubility of the reactants in the biphasic butanol-water system, thereby facilitating better mass transfer between the organic and aqueous phases. The nucleophilic attack by the amine group of p-phenylenediamine on the activated anthraquinone ring is carefully modulated by the reaction temperature, where deviations outside the 95°C to 105°C window can lead to either sluggish kinetics or the generation of poly-condensed impurities that compromise the final dye shade. This delicate balance ensures that the mono-substituted product is favored over di-substituted side products, which is essential for maintaining the specific hue characteristics required for Solvent Blue 19 specifications.
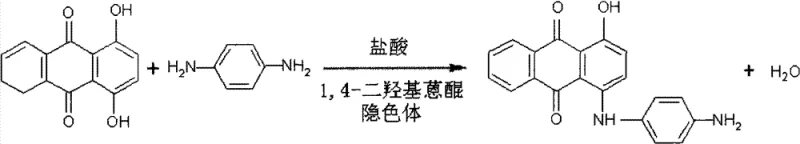
Impurity control is rigorously managed through the specific post-reaction processing steps, particularly the alkaline treatment with dilute sodium hydroxide solution at elevated temperatures of 90°C to 95°C. This step is mechanistically designed to convert any residual 1,4-dihydroxyanthraquinone into its water-soluble sodium salt form, which can be easily washed away, thus reducing the residual starting material to less than 0.5% in the final isolated solid. Furthermore, the choice of acetic acid as the solvent for the acylation step prevents the hydrolysis of the newly formed amine bond while providing a homogeneous medium for the acetic anhydride to react efficiently with the free amine group of the condensation intermediate. The result is a crystalline product with a narrow particle size distribution and low metal ion content, which directly translates to improved rheological properties when the dye is incorporated into polymer melts. For R&D teams, understanding these mechanistic nuances is vital for replicating the high-purity profiles described in the patent and for troubleshooting any potential deviations during the commercial scale-up of complex dye intermediates.
How to Synthesize N-[4-[(4-Hydroxyanthraquinone-1-yl) Amino] Benzyl] Acetamide Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction parameters and sequential processing steps to achieve the reported yields and purity levels consistently. The process begins with the preparation of the butanol-water solvent system, followed by the controlled addition of the anthraquinone derivatives and the amine source under acidic conditions, necessitating precise temperature monitoring to avoid exothermic runaways. Operators must ensure that the distillation of butanol is conducted thoroughly before the filtration step to maximize solvent recovery and minimize environmental waste, aligning with green chemistry principles. The subsequent acylation reaction demands careful control of the acetic anhydride dosage, typically 0.4 to 0.6 times the weight of the condensation material, to prevent over-acetylation while ensuring complete conversion of the amine functionality. Detailed standardized synthetic steps see the guide below for exact operational protocols.
- Conduct condensation of 1,4-dihydroxyanthraquinone and its leuco body with p-phenylenediamine in a butanol-water medium using hydrochloric acid catalyst at 95-105°C.
- Perform post-reaction workup including water addition, distillation, filtration, and alkaline treatment to remove unreacted starting materials and reduce ash content.
- Execute acylation of the condensed intermediate with acetic anhydride in an acetic acid medium at 110-112°C to finalize the product structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented methodology offers substantial advantages by fundamentally altering the raw material cost structure and simplifying the supply chain logistics associated with anthraquinone dye production. The shift from using p-aminoacetanilide in traditional methods to utilizing p-phenylenediamine (Ursol D) followed by acetylation leverages the lower market price and reduced unit consumption of the diamine, resulting in a more economically viable production model. Additionally, the elimination of boric acid catalysts removes the need for expensive and complex heavy metal removal processes, further streamlining the manufacturing workflow and reducing the overall cost of goods sold. This process optimization allows manufacturers to offer high-purity dye intermediates at a more competitive price point without sacrificing quality, addressing the constant pressure on procurement managers to reduce expenses in volatile chemical markets.
- Cost Reduction in Manufacturing: The replacement of scarce and expensive 1-hydroxy-4-chloroanthraquinone with readily available 1,4-dihydroxyanthraquinone and its leuco derivative significantly lowers the raw material entry cost for production facilities. By avoiding the use of specialized chlorinated precursors that lack large-scale production capacity, manufacturers can secure a stable supply of inputs at predictable prices, insulating the supply chain from market fluctuations associated with niche chemicals. Furthermore, the high conversion efficiency of the condensation reaction minimizes the loss of valuable anthraquinone feedstock, ensuring that the maximum amount of raw material is converted into saleable product rather than waste. The simplified purification process, which avoids multiple extensive hot water washes, also reduces utility consumption in terms of steam and water, contributing to lower operational expenditures.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as hydrochloric acid, butanol, acetic acid, and acetic anhydride ensures that the production process is not dependent on single-source suppliers or geopolitically sensitive raw materials. This diversification of the supply base enhances the resilience of the manufacturing operation, reducing the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without catastrophic failure, further supports consistent output rates, allowing supply chain planners to forecast delivery timelines with greater accuracy. This reliability is crucial for downstream customers in the textile and plastics industries who operate on tight just-in-time manufacturing schedules and cannot afford interruptions in their dye supply.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of standard reactor materials and common solvent systems that are well-understood in industrial chemical engineering, facilitating the transition from pilot plant to multi-ton production. The significant reduction in ash content and residual starting materials means that the final product requires less downstream cleaning, reducing the volume of wastewater generated during the application phase by the end-user. Moreover, the ability to recover and recycle butanol through distillation aligns with increasingly stringent environmental regulations regarding volatile organic compound emissions and solvent waste disposal. This environmental compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer, a key factor for multinational corporations evaluating their vendor partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology for anthraquinone derivatives. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of adopting this method for large-scale commercial production.
Q: How does the new HCl-catalyzed method improve product purity compared to traditional boric acid methods?
A: The novel hydrochloric acid-catalyzed route significantly reduces ash content to below 0.2% and limits 1,4-dihydroxyanthraquinone residues to less than 0.5%, whereas traditional boric acid methods often result in ash content greater than 0.5% and variable purity levels between 79% and 97%.
Q: Why is the ratio of 1,4-dihydroxyanthraquinone to its leuco body critical in this synthesis?
A: Maintaining a molar ratio of approximately 3.4:1 to 3.8:1 between 1,4-dihydroxyanthraquinone and its leuco compound ensures optimal reaction kinetics and thorough conversion, which minimizes byproduct formation and stabilizes the final dye shade for polyester applications.
Q: What are the primary commercial advantages of using p-phenylenediamine over p-aminoacetanilide in this process?
A: Utilizing p-phenylenediamine (Ursol D) followed by acylation offers lower raw material costs and reduced unit consumption compared to direct condensation with p-aminoacetanilide, while simultaneously allowing for better control over the final acetylation step to ensure consistent product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Solvent Blue 19 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and supply continuity in the specialty chemicals sector, which is why we have invested heavily in mastering complex synthetic pathways like the one described in CN102050757B. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global textile and polymer manufacturers without compromising on stringent purity specifications. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of N-[4-[(4-hydroxyanthraquinone-1-yl) amino] benzyl] acetamide meets the low ash and high content standards required for high-performance polyester spinning applications. Our commitment to technical excellence allows us to bridge the gap between laboratory innovation and industrial reality, delivering products that perform reliably in your most demanding formulations.
We invite you to engage with our technical procurement team to discuss how our optimized manufacturing processes can support your specific product development goals and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient synthesis routes can lower your total landed costs while improving the quality of your final dyed products. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique requirements, ensuring a partnership built on transparency, technical expertise, and mutual growth.
