Scalable Synthesis of Benzimidazole Derivatives via Composite Solid Catalysis for Industrial Applications
Scalable Synthesis of Benzimidazole Derivatives via Composite Solid Catalysis for Industrial Applications
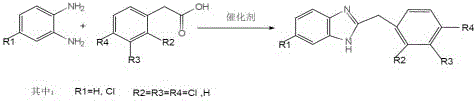
The pharmaceutical and electronic chemical industries are constantly seeking robust, scalable methodologies for constructing heterocyclic scaffolds, and the technology disclosed in patent CN109535080B represents a significant leap forward in the efficient manufacturing of benzimidazole derivatives. This specific intellectual property outlines a refined synthetic protocol that utilizes o-phenylenediamine or its derivatives reacting with phenylacetic acid derivatives under the influence of a specialized composite solid catalyst system. Unlike legacy processes that rely on corrosive liquid acids or generate substantial oxidative byproducts, this innovation leverages a heterogeneous catalytic environment combined with high-boiling organic solvents to achieve superior reaction kinetics and product quality. The method operates within a moderate temperature range of 100-130°C over a duration of 4-8 hours, facilitating a condensation-cyclization sequence that is both economically viable and environmentally compliant for large-scale production facilities. By integrating a water-separation mechanism directly into the reaction setup, the process effectively shifts the chemical equilibrium towards the desired product, thereby maximizing throughput while minimizing the formation of difficult-to-remove impurities that often plague traditional batch syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of benzimidazole cores has been fraught with significant technical and operational challenges that hinder cost-effective manufacturing and supply chain stability. The first conventional pathway typically involves the condensation of o-phenylenediamine with carboxylic acids under the harsh influence of strong mineral acids like hydrochloric acid or polyphosphoric acid, which necessitates extremely high reaction temperatures and places severe corrosive demands on reactor equipment, leading to frequent maintenance downtime and safety hazards. Alternatively, the second common route employs aldehyde compounds and oxidants to induce cyclization, a method that, while operationally simple, suffers from poor atom economy and generates a complex mixture of oxidative byproducts that are notoriously difficult to separate from the target molecule. Furthermore, emerging techniques such as microwave-assisted synthesis, although rapid on a laboratory scale, have failed to translate effectively to industrial continuous flow or large batch reactors due to penetration depth limitations and energy inefficiency. These traditional approaches often result in yields hovering around 70% with products exhibiting poor color profiles, requiring extensive and costly downstream purification steps such as column chromatography or multiple recrystallizations to meet the stringent purity specifications required by pharmaceutical clients.
The Novel Approach
In stark contrast to these legacy limitations, the novel approach detailed in the patent introduces a transformative strategy utilizing a composite solid catalyst system, specifically combinations like Zn/Al2O3 or Zn/B2O3, which fundamentally alters the reaction landscape for benzimidazole synthesis. This methodology replaces corrosive liquid acids with a reusable solid catalyst that not only provides the necessary acidic sites for cyclization but also incorporates zinc powder to act as an in-situ antioxidant, preventing the oxidative degradation of the sensitive diamine starting material. The process utilizes high-boiling solvents such as xylene or toluene, which allow the reaction to proceed at elevated temperatures of 100-130°C while enabling the continuous removal of water via a Dean-Stark type water separator, thus driving the reversible condensation reaction to completion. This strategic combination results in a dramatic improvement in product quality, evidenced by excellent color and yields consistently exceeding 80%, while simultaneously simplifying the workup procedure through straightforward filtration and solvent recovery. The elimination of heavy metal contaminants and the ability to recycle the bulk solvent make this route exceptionally attractive for a reliable benzimidazole derivative supplier aiming to reduce environmental footprint and operational costs.
Mechanistic Insights into Composite Solid Catalyst Cyclization
The mechanistic pathway facilitated by the composite solid catalyst, specifically the Zn/Al2O3 or Zn/B2O3 systems, represents a sophisticated departure from traditional homogeneous acid catalysis that warrants deep technical analysis for R&D optimization. In this novel architecture, the zinc component serves a dual critical function: primarily, it acts as a sacrificial reductant that effectively suppresses the oxidative degradation of the sensitive o-phenylenediamine starting material, which is notoriously prone to forming dark-colored polymeric tars under aerobic heating conditions. Simultaneously, the metal oxide support, whether alumina or boron trioxide, provides the necessary Lewis acidic sites to activate the carboxylic acid carbonyl group, promoting the nucleophilic attack by the amine nitrogen without the excessive protonation that leads to substrate decomposition in strong mineral acid media. This synergistic interaction ensures that the cyclization proceeds through a clean condensation pathway, minimizing the formation of oligomeric impurities that typically plague benzimidazole syntheses and necessitate costly chromatographic purification steps in later stages. The solid nature of the catalyst also allows for easy separation post-reaction, preventing catalyst residues from contaminating the final API intermediate and ensuring compliance with strict heavy metal limits.
Furthermore, the thermodynamic management of the reaction through the use of high-boiling solvents and water separation plays a pivotal role in the observed yield enhancement and impurity profile control. By employing solvents like xylene or toluene, the system maintains a liquid phase at temperatures sufficient to overcome the activation energy barrier for cyclization while remaining below the decomposition threshold of the reactants. The integration of a water separator allows for the azeotropic removal of water generated during the amide formation and subsequent dehydration steps, effectively shifting the equilibrium position according to Le Chatelier's principle towards the formation of the benzimidazole ring. This continuous removal of the byproduct water prevents the hydrolysis of the intermediate amide back to the starting materials, a common issue in closed-system reactions, thereby locking in the conversion efficiency. Additionally, the use of ammonia water for neutralization in the post-treatment phase ensures that any unreacted acid is converted into water-soluble salts that are easily washed away, leaving behind a high-purity organic phase ready for final crystallization.
How to Synthesize 2-Benzylbenzimidazole Efficiently
The practical execution of this synthesis route is designed for seamless integration into existing multipurpose chemical reactors, requiring only standard modifications such as the installation of a water separator to achieve optimal results. The process begins with the precise charging of o-phenylenediamine and phenylacetic acid derivatives in a molar ratio ranging from 1:1 to 1:3, preferably 1:1.2, alongside the composite solid catalyst at a mass ratio of 1:0.01 to 1:0.15 relative to the diamine. Once the high-boiling solvent is added, the mixture is heated to reflux, typically between 110°C and 130°C, and maintained for 4-8 hours to ensure complete conversion while monitoring water collection to gauge reaction progress. Following the reaction, the solvent is recovered via reduced pressure distillation for reuse, and the crude product undergoes a mild workup involving ethanol dissolution, ammonia neutralization, and activated carbon decolorization to yield the final crystalline material. The detailed standardized operating procedures, including specific stirring rates, cooling profiles, and filtration parameters required for GMP-compliant manufacturing, are outlined in the technical guide below.
- Charge o-phenylenediamine derivatives and phenylacetic acid derivatives into a reactor with a composite solid catalyst such as Zn/Al2O3 and a high-boiling solvent like xylene.
- Heat the mixture to 100-130°C for 4-8 hours under reflux with a water separator to continuously remove reaction-generated water and drive equilibrium.
- Recover solvent via distillation, dissolve the residue in ethanol, neutralize with ammonia water, decolorize with activated carbon, and recrystallize to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology translates directly into tangible operational efficiencies and risk mitigation strategies that enhance the overall resilience of the supply network. The shift from corrosive liquid acids to a stable solid catalyst system drastically reduces the wear and tear on reactor vessels and piping, leading to significantly extended equipment lifecycles and reduced capital expenditure on maintenance and replacement parts. Moreover, the ability to recover and recycle the majority of the high-boiling organic solvent through simple distillation creates a closed-loop material flow that minimizes raw material consumption and lowers the volume of hazardous waste requiring disposal. This streamlined approach not only simplifies the regulatory compliance burden associated with waste treatment but also stabilizes the production schedule by eliminating the bottlenecks often caused by complex purification sequences in traditional methods.
- Cost Reduction in Manufacturing: The implementation of this composite catalyst system drives down manufacturing costs through multiple mechanisms, primarily by eliminating the need for expensive and hazardous strong acid reagents and the associated neutralization waste streams. The solid catalyst can potentially be regenerated or reused, further amortizing the cost of goods sold over multiple batches, while the high selectivity of the reaction reduces the loss of valuable starting materials to side reactions. Additionally, the simplified workup procedure, which avoids complex extraction or chromatography steps, reduces labor hours and utility consumption, resulting in substantial cost savings that can be passed down to the customer or reinvested into capacity expansion. The overall process efficiency ensures that the cost reduction in pharmaceutical intermediates manufacturing is achieved without compromising the quality or purity of the final benzimidazole derivative product.
- Enhanced Supply Chain Reliability: By utilizing readily available and stable raw materials such as o-phenylenediamine derivatives and phenylacetic acid, the supply chain becomes less vulnerable to the volatility often seen with specialized reagents or unstable oxidants. The robustness of the reaction conditions, which tolerate slight variations in temperature and mixing without significant yield penalties, ensures consistent batch-to-batch quality, thereby reducing the risk of production delays caused by out-of-specification results. This reliability is crucial for reducing lead time for high-purity benzimidazole derivatives, allowing manufacturers to respond more agilely to fluctuating market demands and urgent customer orders. The simplified logistics of handling solid catalysts and common organic solvents also streamline inventory management and reduce the safety risks associated with transporting corrosive liquids.
- Scalability and Environmental Compliance: The transition from laboratory to commercial scale-up of complex benzimidazole derivatives is significantly de-risked by the heterogeneous nature of the catalysis, which avoids the heat transfer limitations and mixing issues common in highly exothermic liquid acid reactions. The process inherently generates less hazardous waste, as the primary byproduct is water which is separated and removed, and the solvent is recycled, aligning perfectly with modern green chemistry principles and stringent environmental regulations. This environmental friendliness facilitates easier permitting for new production lines and ensures long-term operational continuity in regions with strict emission controls. The scalability is further supported by the use of standard industrial unit operations like distillation and filtration, making it easy to replicate the process across different manufacturing sites globally.
Frequently Asked Questions (FAQ)
To assist our technical partners and potential clients in evaluating the feasibility of this synthesis route for their specific applications, we have compiled a set of frequently asked questions based on the detailed experimental data and technical disclosures within the patent literature. These inquiries address critical aspects regarding catalyst selection, solvent optimization, and the specific advantages of the water-separation technique employed in this method. Understanding these nuances is essential for R&D teams planning to integrate this technology into their existing pipelines or for procurement specialists assessing the long-term viability of this supply source. The answers provided below are derived strictly from the validated experimental examples and theoretical framework established in the patent documentation.
Q: What specific catalyst system is utilized in this novel synthesis method?
A: The process employs a composite solid catalyst system, specifically utilizing combinations such as Zn/Al2O3 or Zn/B2O3, where zinc powder inhibits oxidation and the metal oxide provides acidic catalytic sites.
Q: How does this method improve yield compared to traditional strong acid catalysis?
A: By using high-boiling solvents like xylene or toluene coupled with a water separator, the reaction equilibrium is driven forward, increasing yields from typical levels of 70% to over 80% while improving product color.
Q: Is the solvent recyclable in this industrial process?
A: Yes, the high-boiling organic solvents such as toluene or xylene can be largely recovered through reduced pressure distillation after the reaction, significantly reducing raw material costs and waste discharge.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Benzylbenzimidazole Supplier
As a premier CDMO and manufacturer, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to leverage advanced synthetic routes like the one described in CN109535080B to deliver superior value to our global clientele. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full industrial manufacturing is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. By adopting this innovative composite catalyst technology, we can offer our partners a supply of benzimidazole derivatives that is not only cost-effective but also characterized by exceptional consistency and low impurity profiles suitable for sensitive pharmaceutical applications.
We invite you to engage with our technical team to discuss how this optimized synthesis method can be tailored to your specific project requirements and volume needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis that details the potential economic benefits of switching to this greener, more efficient manufacturing process. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to serve as your trusted long-term partner in the supply of high-value chemical intermediates. Let us collaborate to drive innovation and efficiency in your supply chain today.
