Advanced Diamine Monomer Technology Enabling High-Modulus Colorless Polyimide For Flexible Electronics
The rapid evolution of flexible display technology has created an urgent demand for substrate materials that combine exceptional optical clarity with robust thermal and mechanical performance. Patent CN112110827B introduces a breakthrough diamine monomer specifically engineered to address the longstanding trade-off between transparency and heat resistance in polyimide films. This novel chemical structure incorporates a unique arrangement of trifluoromethyl and carboxyl groups, which effectively suppresses the formation of charge transfer complexes that typically cause yellowing in aromatic polyimides. By leveraging this advanced molecular design, manufacturers can now produce colorless transparent films capable of withstanding the rigorous processing conditions required for next-generation smartphones and wearable devices. The strategic inclusion of a third reactive carboxyl point significantly enhances intermolecular crosslinking density, resulting in a material that maintains dimensional stability even under extreme thermal stress.
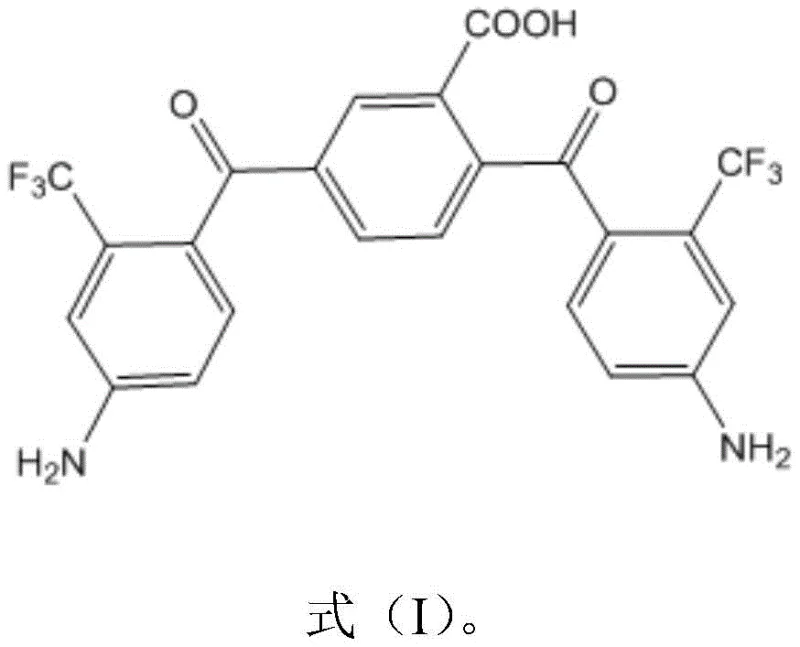
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional aromatic polyimides have long been plagued by inherent limitations that restrict their application in high-end optoelectronic devices, primarily due to their tendency to form intense brownish-yellow colors. This discoloration arises from strong intramolecular and intermolecular charge transfer interactions within the polymer backbone, which drastically reduce visible light transmittance and compromise the visual quality of flexible displays. While previous attempts to mitigate this issue involved introducing fluorine atoms or alicyclic structures, these modifications often came at the expense of thermal stability and mechanical modulus. Many existing solutions fail to simultaneously achieve a glass transition temperature above 350°C and a high modulus, making them unsuitable for components that require high-temperature processing steps. Furthermore, some prior art methods rely on complex multi-step syntheses or expensive monomers that drive up production costs and complicate supply chain logistics for large-scale manufacturing operations.
The Novel Approach
The innovative synthesis route disclosed in the patent data overcomes these historical barriers by utilizing a specialized diamine structure that balances electronic effects with steric hindrance to prevent coloration without sacrificing thermal performance. This new approach employs a Grignard metallization reaction followed by a copper-catalyzed coupling process, which allows for the precise construction of the molecular architecture under relatively mild conditions. The introduction of the carboxyl group serves as a critical third reactive site, enabling enhanced crosslinking during the subsequent polyimide formation which directly contributes to the observed increase in film modulus. Unlike conventional methods that might require high-temperature imidization or harsh chemical treatments, this pathway facilitates the production of high-quality polyamic acid precursors that can be converted into films with superior optical and mechanical properties. The result is a material solution that meets the stringent requirements of modern flexible electronics while offering a more streamlined and cost-effective production workflow.
Mechanistic Insights into CuCN-Catalyzed Coupling and Grignard Metallization
The core of this synthetic achievement lies in the controlled formation of the organomagnesium intermediate, known as Substance A, which acts as the nucleophilic partner in the subsequent coupling reaction. The process begins with the metallization of 2-chloro-5-bromoethyl benzoate using magnesium metal in an anhydrous ether solvent, initiated by a small amount of iodine to activate the metal surface. This step requires careful control of the addition rate to manage the exothermic nature of the Grignard formation, ensuring that the reaction proceeds smoothly at room temperature without runaway thermal events. The resulting di-grignard species is highly reactive and must be handled under inert conditions to prevent quenching by moisture or oxygen, which would lead to significant yield losses and impurity formation. The stability of this intermediate is crucial for the success of the overall synthesis, as it dictates the efficiency of the carbon-carbon bond formation in the following step.
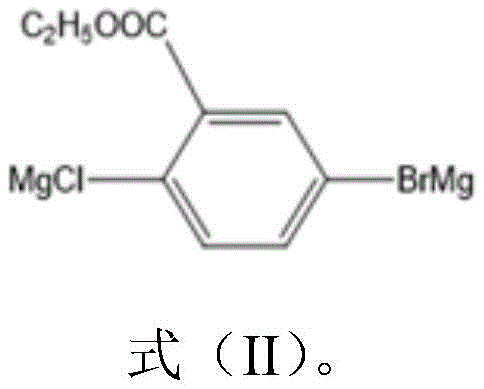
Following the generation of the Grignard reagent, the synthesis proceeds through a copper-catalyzed coupling with 4-amino-3-trifluoromethylbenzoyl chloride, a transformation that constructs the central ketone linkage of the diamine backbone. Cuprous cyanide serves as the catalyst in this step, facilitating the cross-coupling reaction in tetrahydrofuran solvent at room temperature over an extended period to ensure complete conversion. The presence of the trifluoromethyl group on the electrophile enhances the reactivity of the acid chloride while simultaneously contributing to the final polymer's optical clarity by reducing electron density interactions. After the coupling is complete, the reaction mixture undergoes acidification with aqueous hydrochloric acid to decompose any remaining organometallic species and protonate the amine groups. Final purification involves extraction and silica gel chromatography, which effectively removes metal residues and side products, ensuring the high purity required for electronic grade applications.
How to Synthesize High-Performance Diamine Monomer Efficiently
Implementing this synthesis route in a production environment requires strict adherence to the specified reaction parameters to ensure consistent quality and yield of the target diamine monomer. The process begins with the preparation of the Grignard reagent, where the ratio of magnesium to initiator and the dropwise addition of the halide solution must be meticulously monitored to maintain the reaction temperature within the optimal range. Once the intermediate is formed, it is transferred to the coupling vessel containing the acid chloride and catalyst, where the mixture is stirred for a prolonged duration to maximize conversion before quenching. Detailed standardized operating procedures regarding solvent drying, inert gas purging, and workup protocols are essential to minimize variability and ensure that the final product meets the stringent specifications for polyimide precursor applications.
- Prepare the Grignard reagent by reacting 2-chloro-5-bromoethyl benzoate with magnesium metal in anhydrous ether using iodine as an initiator.
- React the resulting organomagnesium intermediate with 4-amino-3-trifluoromethylbenzoyl chloride in the presence of a cuprous cyanide catalyst.
- Perform acidification and purification via silica gel chromatography to isolate the final diamine product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers significant strategic advantages by simplifying the manufacturing process and utilizing readily available raw materials. The ability to conduct key reaction steps at room temperature eliminates the need for energy-intensive heating or cooling systems, which translates into substantial operational cost savings and a reduced carbon footprint for the manufacturing facility. Furthermore, the starting materials such as 2-chloro-5-bromobenzoic acid derivatives and trifluoromethyl benzoyl chlorides are commercially accessible, reducing the risk of supply bottlenecks that often plague specialty chemical production. The robustness of the synthesis pathway also implies a higher tolerance for minor variations in process conditions, leading to improved batch-to-b consistency and reliability for long-term supply contracts.
- Cost Reduction in Manufacturing: The elimination of complex high-temperature processing steps and the use of efficient copper catalysis significantly lower the overall energy consumption and equipment wear associated with production. By avoiding the need for expensive transition metal removal steps often required in palladium-catalyzed processes, the downstream purification costs are drastically simplified, leading to a more economical final product. The high yield and selectivity of the reaction minimize waste generation, further contributing to cost efficiency by reducing the volume of hazardous waste that requires treatment and disposal. These factors combine to create a highly competitive cost structure that allows for aggressive pricing strategies without compromising on profit margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard solvents like ether and tetrahydrofuran ensures that the supply chain is resilient against market fluctuations and geopolitical disruptions. Since the synthesis does not depend on rare or proprietary catalysts that are subject to strict export controls or limited global capacity, procurement teams can secure raw materials with greater confidence and shorter lead times. The scalability of the room temperature reactions means that production capacity can be ramped up quickly to meet surges in demand from the consumer electronics sector without requiring major capital investment in new reactor infrastructure. This flexibility is critical for maintaining continuity of supply in the fast-paced flexible display market.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by operating under mild conditions and generating fewer byproducts compared to traditional high-energy synthesis routes. The use of standard extraction and chromatography techniques for purification aligns with existing environmental compliance frameworks, making it easier to obtain necessary regulatory approvals for new manufacturing sites. The potential for solvent recovery and recycling in the ether and THF steps further enhances the environmental profile of the operation, appealing to end-customers who prioritize sustainability in their supply chain. This alignment with environmental standards future-proofs the production capability against increasingly stringent global regulations on chemical manufacturing emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and performance of this novel diamine monomer in polyimide production. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation to provide accurate guidance for R&D and procurement decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this material into existing product lines and for assessing the potential return on investment for adopting this new technology.
Q: What are the thermal properties of the polyimide derived from this diamine?
A: The polyimide film prepared from this diamine exhibits a glass transition temperature of at least 370°C and a modulus exceeding 4.3 GPa, ensuring exceptional thermal stability.
Q: How does this diamine improve optical transparency compared to traditional aromatic polyimides?
A: By incorporating specific fluorine and carbonyl groups while managing charge transfer complexes, the resulting film achieves colorless transparency suitable for flexible display substrates.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the synthesis utilizes room temperature reactions and commercially available starting materials, facilitating straightforward scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyimide Diamine Monomer Supplier
NINGBO INNO PHARMCHEM stands ready to support your transition to next-generation flexible display materials by leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team possesses deep expertise in handling sensitive organometallic reactions and ensuring stringent purity specifications required for electronic grade chemicals. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the high performance benchmarks set by patent CN112110827B, guaranteeing that the diamine monomer you receive will deliver the expected thermal and optical properties in your final polyimide films.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific production volume and quality requirements. By engaging with us early in your development cycle, you can secure specific COA data and route feasibility assessments that will accelerate your time to market for new flexible electronic products. Let us partner with you to transform this innovative patent technology into a commercial reality that drives value for your organization.
