Advanced Synthesis of Crisaborole Polymer Impurity A for Pharmaceutical Quality Control
Advanced Synthesis of Crisaborole Polymer Impurity A for Pharmaceutical Quality Control
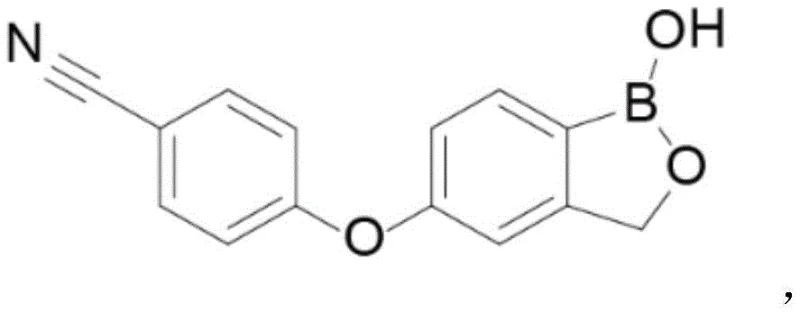
The pharmaceutical industry demands rigorous quality control standards, particularly for complex small molecule drugs like Crisaborole, a non-hormonal topical anti-inflammatory agent. Patent CN115215765A introduces a groundbreaking preparation method for Crisaborole Polymer Impurity A, a critical related substance that must be monitored to ensure drug safety and efficacy. This technical insight report analyzes the novel synthetic pathway disclosed in the patent, highlighting its superiority over conventional methods in terms of yield, purity, and operational safety. By leveraging mild acidic conditions and specific solvent systems, this innovation addresses the longstanding challenge of sourcing high-purity impurity reference standards, thereby enabling more robust quality assurance protocols for global pharmaceutical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of boron-containing impurities has been fraught with difficulties due to the sensitivity of the boron atom to harsh reaction conditions. Traditional approaches often relied on strong mineral acids, such as concentrated hydrochloric acid, in organic solvents like tetrahydrofuran (THF) to induce hydrolysis and subsequent polymerization. As illustrated in prior art synthesis routes, these aggressive conditions frequently lead to uncontrolled degradation of the molecular scaffold, resulting in a complex mixture of by-products rather than the desired specific polymer impurity. The comparative data indicates that such conventional methods suffer from abysmal conversion rates, with yields often plummeting to approximately 22.8% and purity levels struggling to exceed 85%. This inefficiency not only drives up the cost of goods but also complicates the downstream purification process, making it economically unviable for the large-scale production of reference standards required for regulatory compliance.
The Novel Approach
In stark contrast, the methodology outlined in patent CN115215765A employs a sophisticated yet operationally simple strategy utilizing dilute phosphoric acid in an acetonitrile medium. This novel approach fundamentally shifts the reaction paradigm from aggressive degradation to controlled polymerization. By meticulously adjusting the acidity to a mere 0.085wt% phosphoric acid concentration, the process creates an environment that selectively promotes the formation of the target dimeric or polymeric structure without destroying the sensitive nitrile and ether functionalities present in the molecule. The result is a dramatic enhancement in process efficiency, with reported yields surpassing 75% and purity levels consistently exceeding 97%. This significant improvement transforms the production of Crisaborole Polymer Impurity A from a laboratory curiosity into a commercially viable process, ensuring a steady supply of high-quality reference materials for the global supply chain.
Mechanistic Insights into Acidic Hydrolysis and Polymerization
The core of this technological breakthrough lies in the precise manipulation of the hydrolysis kinetics of the pinacol boronic ester group. Under the optimized conditions of 20-30°C, the dilute phosphoric acid acts as a gentle catalyst, facilitating the cleavage of the boron-oxygen bonds in the pinacol ester moiety. Unlike strong acids that might protonate the nitrile group or cleave the ether linkage, the weak acid environment allows for a controlled generation of the boronic acid intermediate, which subsequently undergoes condensation to form the stable benzoxaborole ring system found in the polymer impurity. This mechanistic pathway minimizes the formation of random oligomers and ensures that the reaction proceeds predominantly towards the specific structural isomer defined as Impurity A. The use of acetonitrile as a co-solvent further stabilizes the transition states and ensures homogeneous mixing of the organic substrate with the aqueous acid phase, thereby maximizing the interfacial contact area and reaction rate.
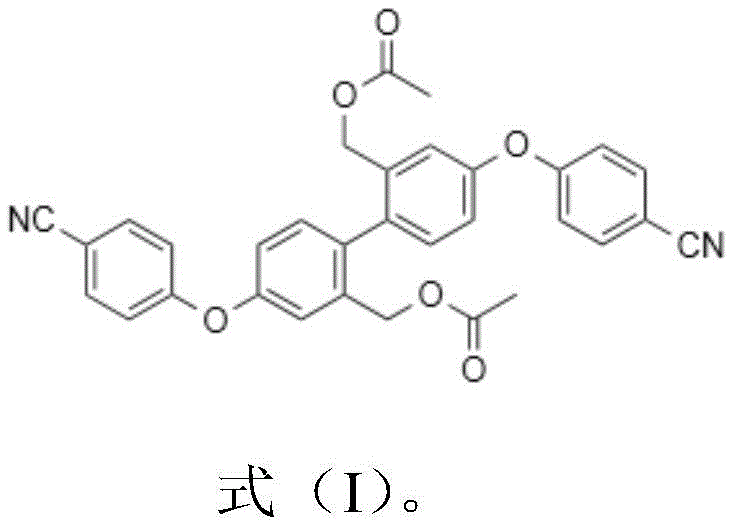
Furthermore, the purification mechanism leverages the distinct polarity differences between the target polymer impurity and the unreacted starting material or other degradation products. The protocol specifies a gradient elution strategy using petroleum ether and ethyl acetate, which effectively separates the target compound based on its specific adsorption characteristics on silica gel. The initial wash with a 10:1 ratio removes non-polar impurities, while the subsequent 5:1 elution selectively recovers the target impurity. This fine-tuned chromatographic separation is critical for achieving the requisite 97% purity, as it removes trace amounts of the starting pinacol ester and any mono-hydrolyzed intermediates. The structural integrity of the final product is confirmed through rigorous spectroscopic analysis, ensuring that the synthesized material is chemically identical to the impurity formed during the actual drug manufacturing process, thus validating its utility as a reference standard.
How to Synthesize Crisaborole Polymer Impurity A Efficiently
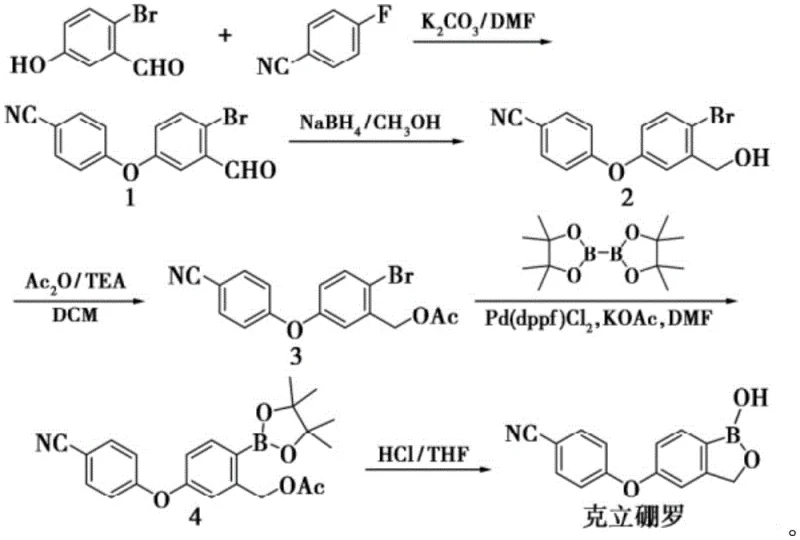
The synthesis protocol described in the patent offers a streamlined three-step procedure that is amenable to both laboratory and pilot-scale operations. The process begins with the dissolution of the key starting material, 2-acetoxymethyl-4-(4-cyanophenoxy) phenylboronic acid pinacol ester, in acetonitrile, followed by the controlled addition of the dilute phosphoric acid solution. The reaction is allowed to proceed at ambient temperature for a duration of 12 to 18 hours, ensuring complete conversion while minimizing thermal stress on the molecule. Following the reaction, the workup involves a straightforward extraction and concentration sequence, culminating in a purification step that guarantees high purity. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Dissolve 2-acetoxymethyl-4-(4-cyanophenoxy) phenylboronic acid pinacol ester in acetonitrile and add dilute phosphoric acid solution.
- Stir the reaction mixture at 20-30°C for 12-18 hours to facilitate polymerization under controlled acidic conditions.
- Extract the product with ethyl acetate, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers substantial strategic advantages over legacy methods. The shift away from hazardous concentrated acids and volatile solvents like THF towards safer, more common reagents like dilute phosphoric acid and acetonitrile significantly reduces the regulatory burden and safety costs associated with chemical handling and waste disposal. This simplification of the reagent profile enhances supply chain resilience, as the raw materials are commodity chemicals with stable availability and pricing, mitigating the risk of production delays caused by specialized reagent shortages. Furthermore, the dramatic increase in yield implies a more efficient utilization of raw materials, directly translating to lower unit costs for the production of these critical reference standards.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents, combined with the substantial increase in reaction yield, drives a significant reduction in the overall cost of manufacturing. By avoiding the need for complex remediation of harsh acid waste and reducing the volume of starting material required per unit of product, manufacturers can achieve a leaner cost structure. The simplified purification process also reduces solvent consumption and labor hours, contributing to further operational savings without compromising on the quality of the final output.
- Enhanced Supply Chain Reliability: The reliance on widely available, non-proprietary reagents ensures a robust and reliable supply chain. Unlike processes dependent on specialized catalysts or custom-synthesized intermediates, this method utilizes off-the-shelf chemicals that are easily sourced from multiple vendors globally. This diversification of supply sources minimizes the risk of bottlenecks and ensures consistent production schedules, which is critical for maintaining the continuous availability of quality control standards required by pharmaceutical regulators.
- Scalability and Environmental Compliance: The mild reaction conditions, operating at near-ambient temperatures and atmospheric pressure, make this process inherently scalable and safe for large-scale production. The reduced generation of hazardous waste aligns with modern green chemistry principles and environmental regulations, facilitating easier permitting and compliance in various jurisdictions. This environmental compatibility not only future-proofs the manufacturing process against tightening regulations but also enhances the corporate sustainability profile of the supplier.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of Crisaborole Polymer Impurity A. These answers are derived directly from the experimental data and technical specifications provided in patent CN115215765A, offering clarity on the feasibility and benefits of this new method for industry stakeholders.
Q: What is the purity level achievable with this new synthesis method?
A: The patented method achieves a purity of greater than 97%, specifically reaching 97.25% in optimized examples, making it suitable for use as a reference standard.
Q: How does this method compare to traditional hydrolysis techniques?
A: Unlike traditional methods using concentrated hydrochloric acid which yield only ~22%, this mild phosphoric acid approach improves yields to over 75% while significantly reducing side reactions.
Q: Is this process scalable for industrial production of reference materials?
A: Yes, the process utilizes common reagents like acetonitrile and dilute phosphoric acid at ambient temperatures, ensuring high safety and ease of scale-up for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Crisaborole Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity impurity standards play in the development and quality control of life-saving medications like Crisaborole. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your demand for reference materials with speed and precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for pharmaceutical intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your quality control objectives and drive efficiency in your operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
