Advanced Refining Technology for High-Purity Sodium p-Toluenesulfinate: A Commercial Scale-Up Perspective
Introduction to Advanced Refining Technologies
The global demand for high-purity organic intermediates continues to escalate, driven by stringent regulatory requirements in the pharmaceutical and agrochemical sectors. A pivotal development in this landscape is documented in patent CN115850128A, which outlines a novel refining method for sodium p-toluenesulfinate. This compound serves as a critical building block for synthesizing medicaments, disperse dye intermediates, and electroplating brightening agents. Traditional manufacturing processes often struggle to meet the rigorous purity standards required for sensitive applications, such as medical raw materials, where sodium salt content must be minimized to trace levels. The innovation presented in this patent addresses these challenges by introducing a sophisticated acid-base extraction strategy that fundamentally alters the purification thermodynamics, ensuring a final product purity exceeding 99% while drastically simplifying the downstream processing workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
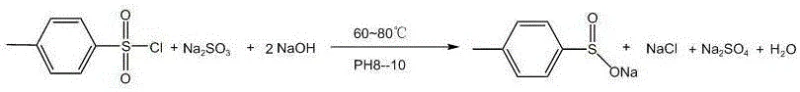
Historically, the industrial production of sodium p-toluenesulfinate has relied on an aqueous phase reaction between p-toluenesulfonyl chloride, sodium sulfite, and sodium hydroxide. While effective for generating the crude material, this method introduces significant purification bottlenecks. The reaction inherently produces substantial quantities of inorganic byproducts, specifically sodium sulfate and sodium chloride, which possess high water solubility similar to the target sulfinate salt. Consequently, separating these impurities through standard crystallization is exceptionally difficult, often necessitating repeated recrystallization cycles that erode overall process yield. Furthermore, the product isolated from aqueous solutions typically exists as a trihydrate, containing three molecules of crystal water per formula unit. Removing this structural water requires aggressive thermal treatment, often involving vacuum drying at temperatures exceeding 110°C, which imposes a heavy energy burden and increases the risk of thermal degradation.
The Novel Approach
In stark contrast to the legacy aqueous processes, the methodology described in patent CN115850128A employs a strategic phase-transfer purification technique. Instead of attempting to purify the salt directly from a complex brine, the process first converts the crude sodium p-toluenesulfinate into its corresponding free acid form, p-toluenesulfinic acid. This transformation is crucial because the free acid exhibits markedly different solubility characteristics compared to its salt counterparts. By exploiting the slight solubility of p-toluenesulfinic acid in water, the process allows for the efficient washing away of highly soluble inorganic contaminants like sodium chloride and sodium sulfate. Following this purification, the acid is reconverted to the sodium salt in an anhydrous alcoholic environment. This approach not only eliminates the formation of crystal hydrates but also ensures that the final precipitation occurs in a medium where inorganic salts are insoluble, resulting in a product of exceptional chemical integrity without the need for energy-intensive dehydration steps.
Mechanistic Insights into Acid-Base Extraction and Neutralization
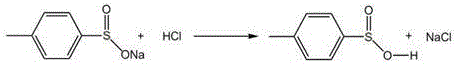
The core of this refining technology lies in the precise manipulation of acid-base equilibria and solubility profiles. The first critical stage involves the acidification of the crude sulfinate solution using hydrochloric acid. As the pH is lowered, the equilibrium shifts towards the formation of p-toluenesulfinic acid. Unlike the ionic sulfinate species, the protonated acid form is less polar and exhibits limited solubility in the aqueous phase, prompting it to crystallize out of the solution. This physical separation is the key to purity; while the target acid precipitates, the unwanted inorganic salts (NaCl and Na2SO4) remain dissolved in the supernatant liquor. Subsequent washing and centrifugation steps effectively strip these impurities from the solid cake, yielding a refined p-toluenesulfinic acid intermediate with a purity greater than 98% and negligible sodium salt content.
Following the isolation of the purified acid, the second mechanistic pillar is the anhydrous neutralization step. The refined p-toluenesulfinic acid is dissolved in absolute methanol or ethanol, creating a homogeneous organic phase. A solution of sodium alkoxide (either sodium methoxide or sodium ethoxide) is then introduced dropwise. This reaction regenerates the sodium p-toluenesulfinate salt; however, unlike the aqueous synthesis, this reaction occurs in an alcohol medium where the target sodium salt is virtually insoluble. As the neutralization proceeds, the sodium p-toluenesulfinate precipitates immediately as a fine, anhydrous powder. Because the reaction byproduct is simply the corresponding alcohol (methanol or ethanol), and the inorganic salts were removed in the previous step, the precipitating solid is chemically pure. This mechanism effectively decouples the formation of the product from the generation of inorganic waste, ensuring a clean crystal lattice free from occluded impurities.
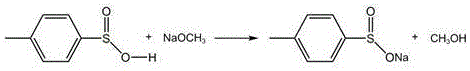
How to Synthesize Sodium p-Toluenesulfinate Efficiently
Implementing this refining protocol requires careful control of reaction parameters to maximize yield and purity. The process begins with the dissolution of crude material followed by precise pH control during acidification to ensure complete conversion to the free acid without excessive acid usage. The subsequent neutralization must be conducted under inert atmosphere protection, typically nitrogen, to prevent oxidation of the sensitive sulfinic acid moiety. Temperature control is also vital; cooling the acidification mixture below 30°C promotes optimal crystallization of the acid, while maintaining mild conditions during neutralization ensures the formation of uniform particles suitable for rapid filtration. For a detailed breakdown of the specific operational parameters, reagent ratios, and equipment specifications required to execute this synthesis successfully, please refer to the standardized guide below.
- Acidify crude sodium p-toluenesulfinate with hydrochloric acid to precipitate p-toluenesulfinic acid, then wash and centrifuge to remove water-soluble salts.
- Dissolve the refined acid in anhydrous alcohol and neutralize with sodium alkoxide solution to precipitate the anhydrous sodium salt.
- Perform flash evaporation drying at 80-82°C to obtain the final high-purity product with minimal residual solvent.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this refining methodology offers profound advantages for procurement managers and supply chain directors seeking to optimize their sourcing strategies for fine chemical intermediates. The most significant impact is observed in the realm of cost reduction in manufacturing. By eliminating the need for multiple recrystallization cycles, which are notoriously material-intensive and labor-heavy, the process significantly reduces the loss of valuable product during purification. Furthermore, the transition from a hydrated product requiring high-temperature vacuum drying to an anhydrous product that can be dried at mild temperatures (80-82°C) results in substantial energy savings. The removal of the high-temperature dehydration step not only lowers utility costs but also extends the lifespan of drying equipment and reduces the carbon footprint of the manufacturing operation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the drastic simplification of the purification train. Traditional methods often suffer from low recovery rates due to the solubility of the product in the mother liquor during repeated washing steps. In this novel approach, the insolubility of the final product in the alcoholic medium ensures near-quantitative recovery during centrifugation. Additionally, the alcohol mother liquor generated during the process is essentially pure and can be recycled directly for the preparation of sodium alkoxide solutions or reused as a solvent, creating a closed-loop system that minimizes raw material consumption and waste disposal costs.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that are prone to variability and batch failures. The robustness of this acid-base extraction method enhances reliability by providing a more forgiving operating window. The ability to use hydrochloric acid, a commodity chemical often available as a byproduct from upstream chlorination processes, ensures a stable and cost-effective supply of reagents. Moreover, the simplified workflow reduces the total batch cycle time, allowing manufacturers to respond more agilely to fluctuations in market demand and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production is frequently hindered by waste management challenges. This refining method aligns perfectly with green chemistry principles by minimizing aqueous waste streams laden with organic contaminants. Since the inorganic salts are separated in the aqueous acidification step and the organic solvent is recovered in the neutralization step, the effluent load is significantly reduced. This facilitates easier compliance with environmental regulations and lowers the capital expenditure required for wastewater treatment infrastructure, making the process highly scalable for commercial production volumes ranging from hundreds of kilograms to hundreds of metric tons annually.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this refining technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of how this method overcomes traditional limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this high-purity intermediate into their own synthesis pipelines.
Q: Why is the conventional aqueous synthesis method insufficient for high-purity applications?
A: Conventional methods produce sodium p-toluenesulfinate trihydrate alongside soluble byproducts like sodium sulfate and chloride. These impurities are difficult to separate via recrystallization due to similar solubilities, often requiring multiple cycles that reduce yield and necessitate high-energy vacuum drying above 110°C.
Q: How does the acidification step improve purity?
A: By converting the sulfinate salt into p-toluenesulfinic acid using hydrochloric acid, the process leverages the acid's slight solubility in water. This allows water-soluble inorganic salts (NaCl, Na2SO4) to be washed away effectively before the final product is regenerated.
Q: What are the advantages of using sodium alkoxide for neutralization?
A: Using sodium methoxide or ethoxide in an anhydrous alcohol environment prevents the formation of crystal water. The resulting sodium p-toluenesulfinate is insoluble in the alcohol medium, allowing for easy separation via centrifugation without co-precipitating inorganic salts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sodium p-Toluenesulfinate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of your final API or specialty chemical is only as good as the intermediates you start with. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated refining techniques described in patent CN115850128A can be seamlessly translated into industrial reality. We are committed to delivering sodium p-toluenesulfinate that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the precise temperature controls and inert atmosphere conditions required for this process, guaranteeing a consistent supply of high-performance intermediates for your most demanding applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall cost of goods. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project milestones and quality standards.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
